No products in the cart.
Slabilen, drops 7.5 mg/ml 15 ml
€9.43 €8.25
Description
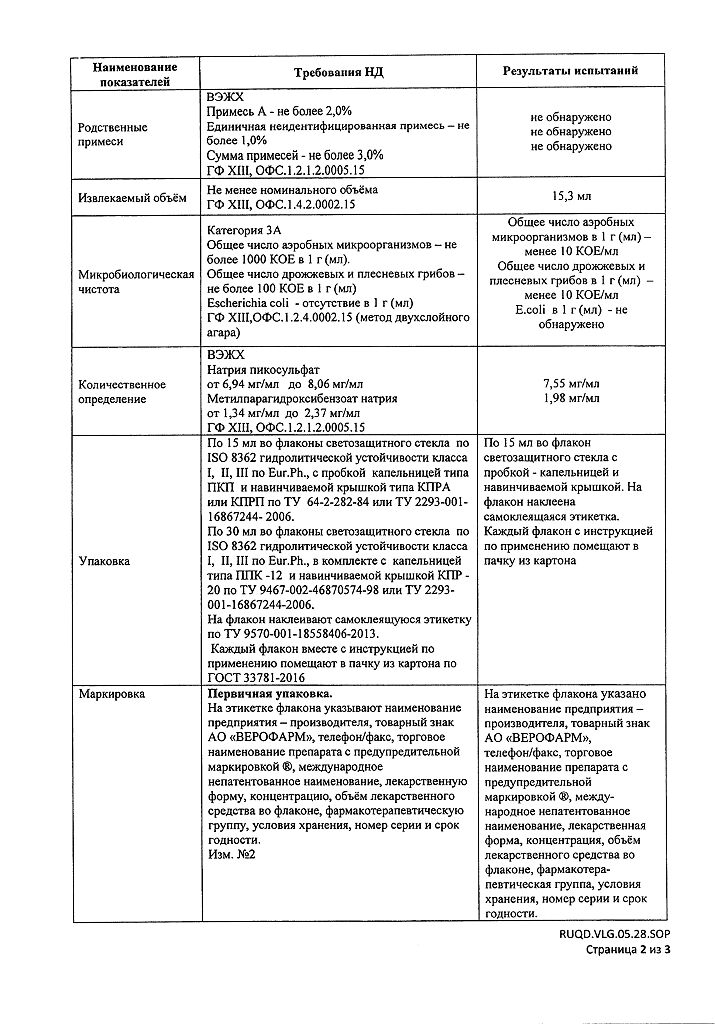
Pharmacotherapeutic group: laxative
ATC code: A06AB08
Pharmacological properties
Pharmacodynamics
Active ingredient – sodium picosulfate is a laxative of triarylmethane group. As local laxative sodium picosulfate after bacterial cleavage in large intestine has stimulating effect on mucosa of large intestine, increasing peristalsis, contributes to accumulation of water and electrolytes in large intestine. This leads to stimulation of the act of defecation, reduction of evacuation time and softening of stools. Sodium picosulfate, being a laxative acting at the level of the large intestine, stimulates the natural process of evacuation of contents from the lower parts of the gastrointestinal tract. Therefore, sodium picosulfate has no effect on digestion or absorption of caloric foods or essential nutrients in the small intestine.
Pharmacokinetics
Absorption and distribution
After oral administration sodium picosulfate enters the colon without significant absorption. Thus, enterohepatic circulation of the drug is excluded.
Biotransformation
Bacterial breakdown of sodium picosulfate occurs in the distal colon with the formation of the active metabolite bis-(p-hydroxyphenyl)-pyridyl-2-methane (BGPM) with a laxative effect.
Excretion
After cleavage, a small amount of BGPM is absorbed and then almost completely bound in the intestinal wall and liver to form inactive glucuronide. After oral administration of sodium picosulfate at a dose of 10 mg (about 10.4% of the total dose), the drug is excreted in the urine as BGPM-glucuronide in 48 hours. Excretion of sodium picosulfate in higher doses decreases.
Relationship between pharmacokinetics/pharmacodynamics
The time of laxative effect development is determined by the rate of active metabolite release (HDL) and is 6-12 hours after application (on average 10 hours).
Indications
Indications
As a laxative in the following cases:
constipation caused by atony or hypotonia of the large intestine (including elderly, bedridden patients, after surgery, childbirth, breastfeeding);
constipation caused by taking medicines;
. to regulate stool in hemorrhoids, proctitis, anal fissures (to soften the consistency of feces);
gall bladder diseases, irritable bowel syndrome with predominant constipation;
constipation caused by dysbacteriosis of the intestine, violations of the diet.
Active ingredient
Active ingredient
Sodium picosulfate
Composition
Composition
Ingredients per 1 ml:
Active ingredient:
Sodium picosulfate
in terms of anhydrous substance
7.5 mg
Excipients:
Sodium methyl parahydroxybenzoate 2.06 mg
Sorbitol (sorbitol food) 375.0 mg
Hydrochloric acid 1 M solution to pH 5.5-7.5
Purified water to 1 ml
How to take, the dosage
How to take, the dosage
It is recommended that you start with the lowest dose possible. In order to achieve regular stools, the dose may be increased to the maximum recommended dose. The maximum recommended daily dose should not be exceeded. Because the drug is not addictive, the dose may be adjusted by the patient individually: the dose may be reduced depending on individual needs, or the drug may be used on an ad hoc basis as needed.
Age | Dosage | Maximum daily dose | ||||
Children over 10 years of age and adults | 10-20 drops (5-10 mg) per day | 10 mg (20 drops) | ||||
Children 4-10 years | 5-10 drops (2.5-5 mg) per day | 5 mg (10 drops) | ||||
Children 0-4 years | 1 drop (0.5 mg of sodium picosulfate) per 2 kg of body weight per day |
The recommended dose for children under 4 years of age is 0.25 mg/kg body weight per day.
To get a laxative effect in the morning hours, the drug should be taken the night before.
The drug does not need to be dissolved in liquid.
In order to restore the natural rhythm of defecation, besides taking the laxative preparation, it is recommended to increase physical activity, to consume enough fiber in the diet (20-25 g/day), to drink enough fluids (not less than 2 liters).
Interaction
Interaction
Diuretics or glucocorticosteroids increase the risk of electrolyte imbalance (hypokalemia) when taking high doses of sodium picosulfate.
Electrolyte imbalance may increase sensitivity to cardiac glycosides.
Co-use of the drug and antibiotics may decrease the laxative effect of the drug.
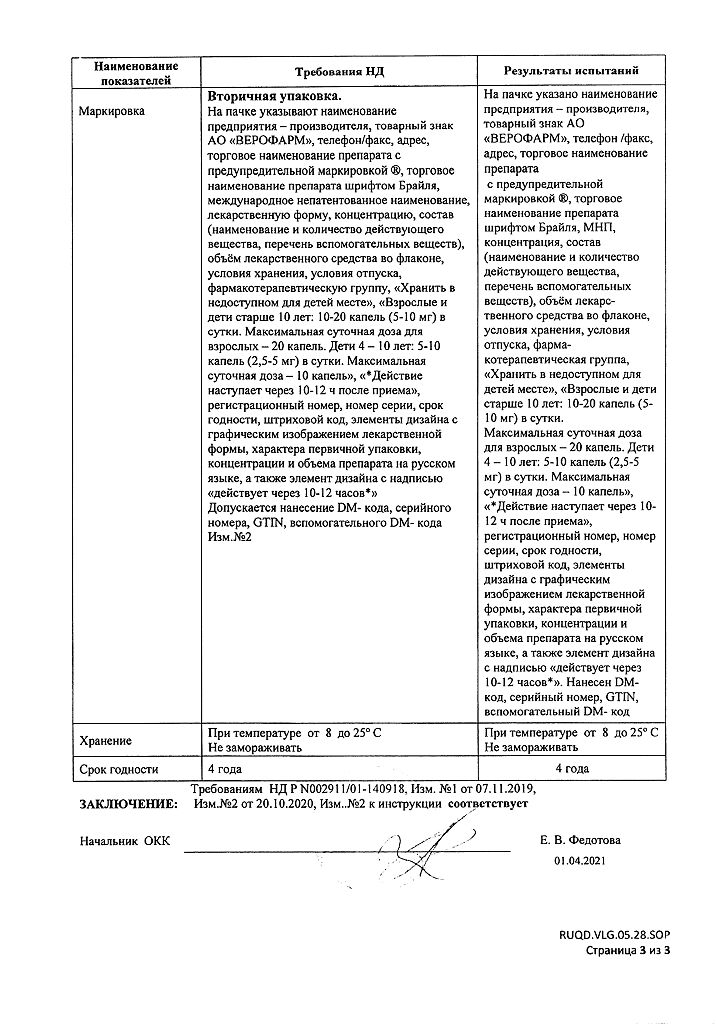
Special Instructions
Special Instructions
It is not recommended to use the drug daily for more than 10 days without medical advice.
Prolonged use of high doses of the drug may lead to fluid loss, electrolyte imbalance, hypokalemia.
1 ml of the preparation contains 0.375 g sorbitol. The maximum recommended daily dose for the treatment of children over 10 years of age and adults contains 0.5 g sorbitol, for children 4-10 years old 0.25 g sorbitol.
The drug does not taste, so it can be added to food.
Children should take the drug only by prescription.
Effect on the ability to drive vehicles, mechanisms
No special clinical studies on the effect of the drug on the ability to drive and operate machinery have been conducted. Despite this, patients should be informed that they may experience dizziness and/or fainting due to vasovagal reaction (i.e., during bowel spasm). If patients experience bowel spasm, they should refrain from performing potentially hazardous activities that require increased concentration and rapid psychomotor reactions (driving vehicles, working with moving machinery, working as a dispatcher, operator).
Contraindications
Contraindications
Hypersensitivity to sodium picosulfate or other drug components
Intestinal obstruction or obstructive bowel disease
Acute abdominal diseases or severe abdominal pain accompanied by nausea, vomiting, fever, including appendicitis
Acute inflammatory bowel disease
Severe dehydration
Fructose intolerance
Side effects
Side effects
Gastrointestinal tract disorders may include discomfort, nausea, vomiting, abdominal cramps and pain, diarrhea. The nervous system may cause dizziness and fainting. Dizziness and fainting that occur after taking the drug may be associated with vasovagal reaction (e.g., straining during defecation, abdominal cramps). Immune system hypersensitivity reactions are possible.
Skin and subcutaneous tissue reactions, such as angioedema, skin rash, itching are possible.
Overdose
Overdose
Symptoms
When taking high doses, diarrhea, dehydration, decreased blood pressure, abnormal water-electrolyte balance, hypokalemia, seizures are possible.
In addition, there are reports of cases of ischemia of the colon muscles associated with taking doses of sodium picosulfate significantly higher than those recommended for the usual treatment of constipation.
Sodium picosulfate, like other laxatives, in chronic overdose may lead to chronic diarrhea, abdominal pain, hypokalemia, secondary hyperaldosteronism, urolithiasis. Renal tubule damage, metabolic alkalosis, and muscle weakness associated with hypokalemia may develop due to chronic laxative abuse.
Treatment
Vomiting or gastric lavage may be induced to reduce absorption of the drug after ingestion. The fluid replenishment and correction of electrolyte balance as well as administration of antispasmodic drugs may be required.
Pregnancy use
Pregnancy use
There are no data on reliable and well-controlled studies in pregnant women. Long-term experience of use has shown no negative effects of sodium picosulfate on pregnancy. Administration of sodium picosulfate in the first trimester of pregnancy is contraindicated. The drug can be used in II and III trimesters of pregnancy only by medical prescription.
The active metabolite and its glucuronides are not excreted with the breast milk. Thus, the drug can be used during breast-feeding.
Studies on the effect of the drug on fertility have not been conducted. No teratogenic effects were detected during preclinical studies.
Similarities
Similarities
Regulax Picosulfate, Guttalax, Slabicap, Picodinar
Additional information
| Weight | 0.053 kg |
|---|---|
| Shelf life | 4 years. Do not use after the expiration date. |
| Conditions of storage | Store at the temperature from 8 to 25 ºC. Do not freeze. Keep out of reach of children. |
| Manufacturer | Veropharm AO, Russia |
| Medication form | oral drops |
| Brand | Veropharm AO |
Other forms…
Related products
Buy Slabilen, drops 7.5 mg/ml 15 ml with delivery to USA, UK, Europe and over 120 other countries.