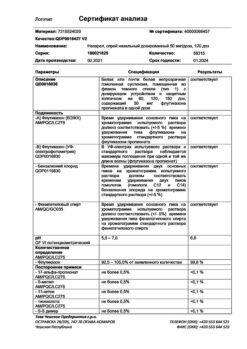
-
×


-
×

-
×


-
×

-
×

-
×


-
×


-
×


-
×

-
×


Subtotal: €275.53