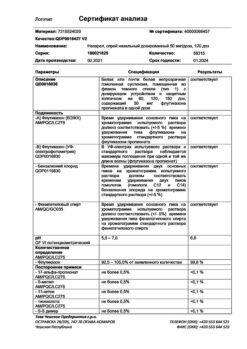
-
×

-
×


-
×


-
×


-
×


-
×

Subtotal: €185.30