No products in the cart.
Description
Pharmacological group: antifungal agent
ATX code: D01AE22
Pharmacological properties
Pharmacodynamics
/p>
Naftifin is an antifungal agent for external use, belonging to the class of allilamines. The mechanism of action is associated with inhibition of squalene-2,3-epoxidase, which leads to a decrease in the formation of ergosterol, which is part of the cell wall of the fungus. It is active against dermatophytes such as Trichophyton, Epidermophyton and Microsporum, molds (Aspergillus spp.), yeasts (Candida spp., Pityrosporum) and other fungi (such as Sporothrix schenckii). Against dermatophytes and aspergillus naphthifin acts fungicidal. Against yeast fungi the drug exhibits fungicidal or fungistatic activity depending on the strain of the microorganism. It has antibacterial activity against Gram-positive and Gram-negative microorganisms that can cause secondary bacterial infections. It has anti-inflammatory action, which contributes to the rapid disappearance of symptoms of inflammation, especially itching.
Pharmacokinetics
When applied externally, naphthifin quickly penetrates the skin, creating sustained antifungal concentrations in its various layers, making once-daily use possible.
Indications
Indications
fungal infections of the skin and skin folds (tinea corporis, tinea inguinalis);
interdigital mycoses (tinea manum, tinea pedum);
fungal nail infections (onychomycosis);
skin candidiasis;
versicolor (pityriasis versicolor);
ringworm (with or without accompanying itching).
Pharmacological effect
Pharmacological effect
Pharmacological group: antifungal agent
ATX code: D01AE22
Pharmacological properties
Pharmacodynamics
Naftifine is an antifungal agent for external use, belonging to the class of allylamines. The mechanism of action is associated with inhibition of squalene-2,3-epoxidase, which leads to a decrease in the formation of ergosterol, which is part of the cell wall of the fungus. Active against dermatophytes such as Trichophyton, Epidermophyton and Microsporum, molds (Aspergillus spp.), yeasts (Candida spp., Pityrosporum) and other fungi (for example, Sporothrix schenckii). Naftifine has a fungicidal effect against dermatophytes and aspergillus. Against yeast fungi, the drug exhibits fungicidal or fungistatic activity depending on the strain of the microorganism. It has antibacterial activity against gram-positive and gram-negative microorganisms that can cause secondary bacterial infections. It has an anti-inflammatory effect, which contributes to the rapid disappearance of symptoms of inflammation, especially itching.
Pharmacokinetics
When used externally, naftifine quickly penetrates the skin, creating stable antifungal concentrations in its various layers, which makes it possible to use it once a day.
Special instructions
Special instructions
The drug Mizol® Evalar is not intended for use in ophthalmology. The drug should not come into contact with the eyes.
To achieve a therapeutic effect, a course of treatment is required.
Mizol® Evalar is effective in the treatment of mycoses affecting areas of the skin with hyperkeratosis, as well as in areas of hair growth.
Propylene glycol contained in the solution may cause skin irritation; If side effects occur, you should stop using the drug and consult a doctor.
Impact on the ability to drive vehicles and machinery
Mizol® Evalar does not affect the performance of potentially hazardous activities that require increased concentration and speed of psychomotor reactions.
Active ingredient
Active ingredient
Naftifin
Composition
Composition
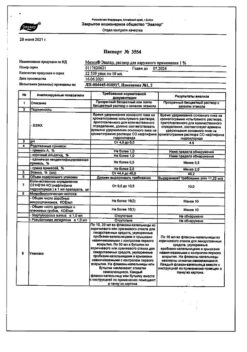
1 ml of solution contains:
Active ingredient: naftifine hydrochloride – 10.00 mg
Excipients: propylene glycol – 50.00 mg; ethanol (ethyl alcohol) 95% – 400.00 mg; purified water – 475.00 mg.
Pregnancy
Pregnancy
The use of Mizol® Evalar during pregnancy and breastfeeding is contraindicated (the safety and effectiveness of naftifine in this category of patients has not been studied).
Contraindications
Contraindications
Hypersensitivity to naftifine or propylene glycol.
Pregnancy and breastfeeding (safety and effectiveness have not been established).
Application of the drug to the wound surface is contraindicated.
With caution
Children’s age (experience of clinical use is limited).
Side Effects
Side Effects
In some cases, local reactions may be observed: dry skin, skin hyperemia and burning. Side effects are reversible and do not require discontinuation of treatment.
If you experience the side effects listed in the instructions or they get worse, or you notice any other side effects not listed in the instructions, tell your doctor.
Interaction
Interaction
Interaction with other drugs has not been studied.
Overdose
Overdose
When using the drug in accordance with the instructions for use, an overdose is unlikely. In case of accidental ingestion, gastric lavage and symptomatic therapy are indicated.
Storage conditions
Storage conditions
At a temperature not exceeding 25 °C. Keep out of the reach of children.
Shelf life
Shelf life
3 years. Once opened, use within 6 months. Do not use after expiration date.
Manufacturer
Manufacturer
Evalar CJSC, Russia
Additional information
| Shelf life | 3 years. After opening, use within 6 months. Do not use after the expiration date. |
|---|---|
| Conditions of storage | At a temperature not higher than 25 ° C. Keep out of reach of children. |
| Manufacturer | Evalar, Russia |
| Medication form | solution for external use |
| Brand | Evalar |
Other forms…
Related products
Buy Mizol Evalar, 1% 50 ml with delivery to USA, UK, Europe and over 120 other countries.