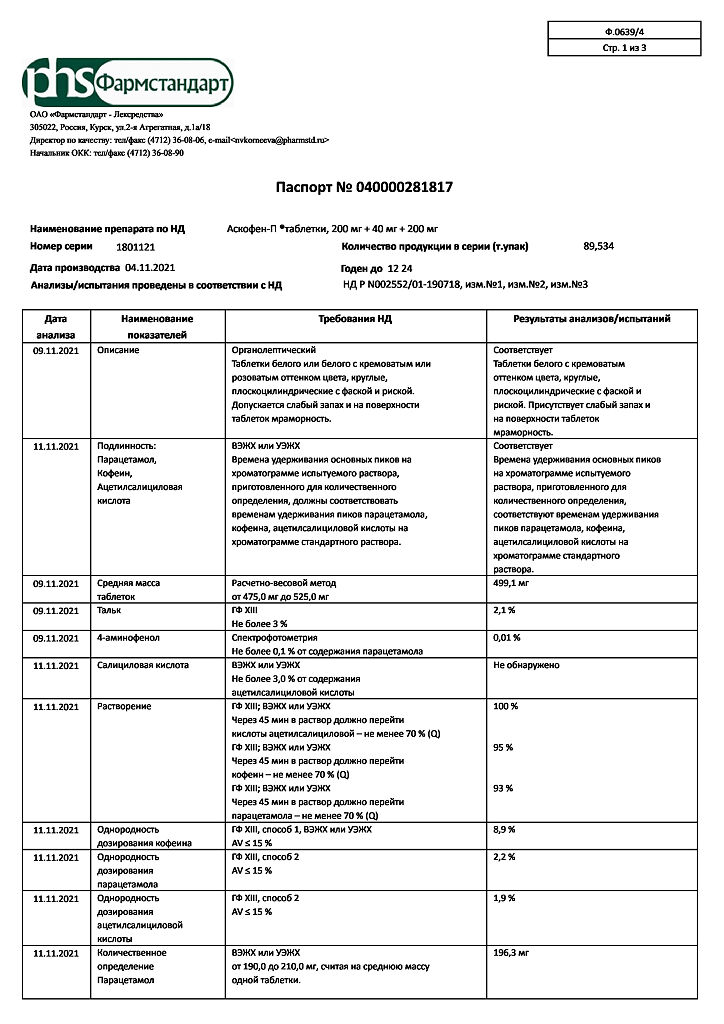
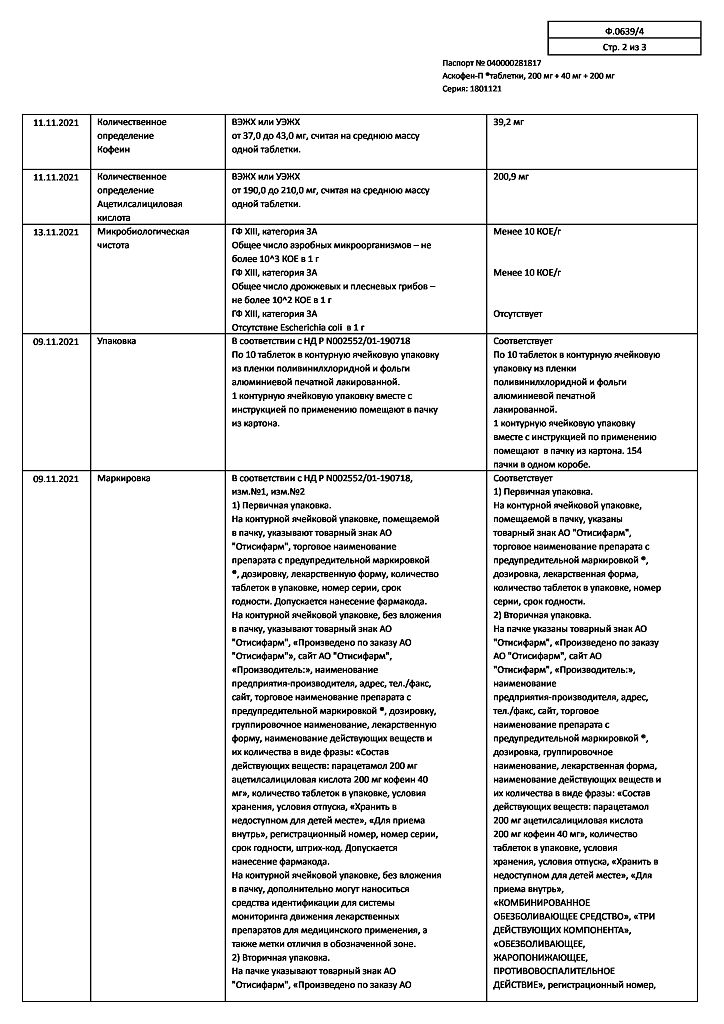
No products in the cart.
Ascophen-P, tablets 10 pcs
€3.28 €2.98
Description
Pharmacotherapeutic group: analgesic combined (non-steroidal anti-inflammatory agent + analgesic non-narcotic agent + psychostimulant)
ATX code: N02BA71
Pharmacological properties
Pharmacodynamics:
Askophen-P® is a combination drug, the action of which is determined by the components in its composition.
Acetylsalicylic acid (ASA) has antipyretic and anti-inflammatory effects, reduces pain, especially caused by inflammation, as well as inhibits thrombosis, improves microcirculation in the inflammation focus. The mechanism of action of ASA is associated with inhibition of cyclooxygenases 1 and 2, which regulate the synthesis of prostaglandins.
Caffeine increases reflex excitability of the spinal cord, excites respiratory and vasomotor centers, dilates blood vessels of skeletal muscles, brain, heart and kidneys, reduces platelet aggregation; it reduces sleepiness and feeling of fatigue and increases mental and physical performance.
In this combination, the low-dose caffeine has almost no simulative effect on the central nervous system, increases the tone of cerebral blood vessels and helps to accelerate blood flow.
Paracetamol has antipyretic, analgesic and weak anti-inflammatory effects, due to its effect on the thermoregulation center in the hypothalamus and a weak ability to inhibit the synthesis of prostaglandins in peripheral tissues.
Pharmacokinetics
Acetylsalicylic acid (ASA): absorption is complete when taken orally. During absorption it undergoes presystemic elimination in the intestinal wall and systemic elimination in the liver by deacetylation.
The resorbed part is rapidly hydrolyzed by cholinesterases and albumin esterase, so the half-life is not more than 15-20 minutes. Binding with plasma proteins (albumin) is 75-90%. Time to reach maximum concentration (Tmax) – 2 hours. It is metabolized mainly in liver and is eliminated by active secretion in renal tubules as salicylate (60%) and its metabolites.
The elimination rate depends on the dose: at low doses the elimination half-life is 2-3 hours, with increasing doses it can increase to 15-30 hours.
Paracetamol: absorption is high, Tmax is reached after 0.5-2 hours and is 5-20 mcg/ml, binding to plasma proteins is 15%. It penetrates through the blood-brain barrier. It is metabolized in liver (90-95%) with formation of active and inactive metabolites with glutathione and CYP2E1.
In case of glutathione deficiency these metabolites can block the enzyme systems of hepatocytes and cause their necrosis. The elimination half-life is 1-4 hours. It is excreted by kidneys as metabolites. Only 3% is eliminated unchanged. In elderly patients the drug clearance is decreased and the half-life is increased.
Caffeine: is completely and quickly absorbed in the gastrointestinal tract, Tmax is reached after 50-75 minutes and is 1.58-1.76 mg/l. It is rapidly distributed in all organs and tissues of the body, easily penetrates through the blood-brain barrier and the placenta. Binding with blood proteins is 25-36%.
More than 90% is metabolized in the liver. In adults about 80% of caffeine dose is metabolized to paraxanthine, about 10% to theobromine and about 4% to theophylline. The half-life in adults is 3.9-5.3 hours (sometimes up to 10 hours) Excretion of caffeine and its metabolites is done by kidneys (1-2% of adults excrete unchanged).
Indications
Indications
Back pain, Neuralgia, Colds, Headache, Joint pain (arthralgia), Myalgia (muscle pain), Neuritis, Flu, Pain, Fever, Migraine, Painful menstruation (algodysmenorrhea) Pain syndrome of mild to moderate intensity (various genesis): headache, migraine, toothache, neuralgia, myalgia, arthralgia, algodysmenorrhea.
Fever syndrome: in acute respiratory diseases, influenza.
Active ingredient
Active ingredient
Acetylsalicylic acid, Caffeine, Paracetamol
Composition
Composition
The active ingredients:
Paracetamol – 200.00 mg,
Acetylsalicylic acid – 200.00 mg,
Caffeine anhydrous, in terms of dry substance – 40.00 mg;
Auxiliary substances: potato starch – 45.60 mg, povidone K-17 (polyvinylpyrrolidone low molecular weight medical) – 2.40 mg, stearic acid – 2.50 mg, talc – 6.40 mg, calcium stearate – 1.10 mg, silicone emulsion KE 10-12 – 1.25mg, vaseline oil – 0.75 mg.
How to take, the dosage
How to take, the dosage
The drug should not be taken more than 5 days as an analgesic and more than 3 days as an antipyretic (without a physician’s prescription and supervision).
Over the mouth (with or after meals, drinking plenty of water with each dose).
In case of headache, the recommended dose is 1-2 tablets, in case of severe headache, the next dose after 4-6 hours.
In case of migraine, the recommended dose is 2 tablets at the appearance of symptoms, if necessary, the next dose in 4-6 hours. For treatment of headache and migraine the drug is used not more than 4 days.
In pain syndrome 1-2 tablets; the average daily dose is 3-4 tablets, the maximum daily dose is 8 tablets.
The course of treatment is not more than 3-5 days.
Elderly (over 65 years)
In elderly patients, especially those with low body weight, caution should be exercised.
Patients with hepatic and renal impairment
The effect of hepatic or renal impairment on the pharmacokinetics of the drug has not been studied.
Given the mechanism of action of acetylsalicylic acid and paracetamol, their use may aggravate renal and hepatic impairment. In this regard, the drug is contraindicated in patients with severe hepatic or renal failure (see section “Contraindications”), and in patients with mild to moderate hepatic and renal failure it should be used with caution (see section “Caution”).
Interaction
Interaction
Acetylsalicylic acid
Other nonsteroidal anti-inflammatory drugs: Increased damaging effect on the gastrointestinal (GI) mucosa, increased risk of gastrointestinal bleeding. If concomitant use is necessary, it is recommended to use gastroprotectors for prevention of NSAID-induced gastrointestinal ulcers, so concomitant use is not recommended.
Glucocorticosteroids: Increased damaging effect on the gastrointestinal mucosa, increased risk of gastrointestinal bleeding. If simultaneous use is necessary, it is recommended to use gastroprotective agents, especially in patients older than 65 years old; therefore simultaneous use is not recommended.
Peroral anticoagulants (e.g. coumarin derivatives): Acetylsalicylic acid (ASA) may potentiate the effect of anticoagulants. Clinical and laboratory monitoring of bleeding time and prothrombin time is necessary. Simultaneous use is not recommended.
Trombolytics: Increased risk of bleeding. The use of ASA in patients within the first 24 hours after acute stroke is not recommended. Simultaneous use is not recommended.
Heparin: Increased risk of bleeding. Clinical and laboratory monitoring of bleeding time is required. Simultaneous use is not recommended.
T Platelet aggregation inhibitors (ticlopidine, paracetamol, clopidogrel, cilostazol): Increased risk of bleeding. Clinical and laboratory monitoring of bleeding time is required. Simultaneous use is not recommended.
Selective serotonin reuptake inhibitors (SSRIs): Concomitant use may affect blood clotting or platelet function, resulting in an increased risk of bleeding in general and gastrointestinal bleeding in particular, so concomitant use is not recommended.
Phenytoin: ASA increases the plasma concentration of phenytoin, which requires monitoring.
Valproic acid: ASA disrupts binding to plasma proteins and therefore may increase its toxicity. Monitoring of plasma concentrations of valproic acid is necessary.
Aldosterone antagonists (spironolactone, canrenoate): ASA may decrease their activity due to impaired sodium excretion, proper control of blood pressure is necessary.
Petrous diuretics (e.g., furosemide): ASA may decrease their activity due to impaired glomerular filtration caused by inhibition of prostaglandin synthesis in kidneys. Concomitant use of nonsteroidal anti-inflammatory drugs (NSAIDs) may lead to acute renal failure, especially in dehydrated patients. If diuretics are used concomitantly with ASA, adequate rehydration of the patient and monitoring of renal function and blood pressure are necessary, especially at the beginning of treatment with diuretics.
Hypotensive agents (ACE inhibitors, angiotensin II receptor antagonists, slow calcium channel blockers): ASA may reduce their activity due to inhibition of prostaglandin synthesis in the kidneys.
Simultaneous use can lead to acute renal failure in elderly or dehydrated patients. If diuretics are used concomitantly with ASA, adequate rehydration of the patient and monitoring of renal function and blood pressure are necessary. Bleeding time should be monitored when concomitant use with verapamil.
The uricosuric drugs (e.g., probenecid, sulfinpyrazone): ASA may decrease their activity due to inhibition of tubular reabsorption leading to high plasma concentration of ASA.
Methotrexate ≤ 15 mg/week: ASA, like all NSAIDs, reduces tubular secretion of methotrexate, increasing its plasma concentration and thus its toxicity. Therefore, concomitant use of NSAIDs in patients receiving high doses of methotrexate is not recommended (see section “Contraindications”).
In patients receiving low doses of methotrexate, the risk of interaction between methotrexate and NSAIDs should also be considered, especially if renal function is impaired. If combined therapy is necessary, it is necessary to monitor general blood count, liver function and renal function, especially in the first days of treatment.
Sulfonylurea derivatives and insulin: Asc increases their hypoglycemic effect, so a decrease in the dose of hypoglycemic drugs may be required when taking a high dose of salicylates. It is recommended to monitor blood glucose levels more frequently.
Alcohol: Increases the risk of gastrointestinal bleeding, concomitant use should be avoided.
Paracetamol
Hepatic microsomal enzyme inducers or potentially hepatotoxic substances (e.g., alcohol, rifampicin, isoniazid, hypnotics and antiepileptics including phenobarbital, phenytoin and carbamazepine): Increased toxicity of paracetamol, which may lead to liver damage even with non-toxic doses of paracetamol, so liver function should be monitored. Simultaneous use is not recommended.
Chloramphenicol: Paracetamol may increase the risk of increased concentration of chloramphenicol. Simultaneous use is not recommended.
Zidovudine: Paracetamol may increase the tendency to develop neutropenia, and therefore hematologic parameters should be controlled. Simultaneous use is possible only with the approval of the physician.
Probenecid:Probenecid decreases the clearance of paracetamol, which requires reducing the dose of paracetamol. Simultaneous use is not recommended.
Indirect anticoagulants: Repeated administration of paracetamol for more than one week increases the anticoagulant effect. Episodic administration of paracetamol has no significant effect.
Propantelin and other drugs that slow gastric evacuation: Decreases the rate of absorption of paracetamol, which may delay or reduce the rapid relief of pain.
Methoclopramide and other drugs that speed up gastric evacuation: Increases the absorption rate of paracetamol and therefore the effectiveness and onset of analgesic action.
Colestyramine: Decreases the rate of absorption of paracetamol, so if maximum analgesia is needed, colestyramine is not taken until 1 hour after taking paracetamol.
Caffeine
Sleeping pills (e.g., benzodiazepines, barbiturates, H1-histamine receptor blockers): Concomitant use may decrease the sleeping pills or decrease the anticonvulsant effect of barbiturates, so concomitant use is not recommended. If simultaneous use is necessary, it is reasonable to take the combination in the morning.
Lithium: Caffeine withdrawal may increase the plasma concentration of lithium because caffeine increases renal clearance of lithium, so a reduction in the lithium dose may be required when caffeine is withdrawn. Simultaneous use is not recommended.
Disulfiram: Patients on disulfiram treatment should be cautioned to avoid using caffeine to avoid the risk of worsening alcohol withdrawal syndrome, due to the cardiovascular and central nervous system stimulatory effects of caffeine.
Ephedrine-like substances: Increased risk of drug dependence. Simultaneous use is not recommended.
Sympathomimetics or levothyroxine: By mutual potentiation may increase the chronotropic effect. Simultaneous use is not recommended.
Theophylline:Simultaneous use decreases the excretion of theophylline.
Quinolone antibacterials, enoxacin and pipemic acid, terbinafine, cimetidine, fluvoxamine and oral contraceptives: Increased caffeine half-life due to inhibition of liver cytochrome P450, so patients with liver dysfunction, heart rhythm disorders and latent epilepsy should avoid using caffeine.
Nicotine, phenytoin and phenylpropanolamine: Decreases the terminal half-life of caffeine.
Clozapine: Caffeine increases the serum concentration of clozapine, probably through both pharmacokinetic and pharmacodynamic mechanisms. Monitoring of the serum concentration of clozapine is necessary. Simultaneous use is not recommended.
Special Instructions
Special Instructions
General
This medication should not be taken concomitantly with medications containing ASA or paracetamol.
Like other migraine medications, caution should be used to rule out other potentially serious neurological disorders before starting treatment of suspected migraine in patients who have not previously been diagnosed with migraine, or in those patients who have migraine with atypical symptoms.
If patients experience vomiting during >20% of migraine attacks or require bed rest during >50% of attacks, the drug should not be used.
If a migraine does not resolve after taking the first two pills of the drug, medical help should be sought.
The drug should not be used if for at least the last three months the patient has had more than 10 headache episodes in a month. In this case, the headache should be suspected due to overuse of the medication and treatment should be discontinued. In addition, patients should seek medical attention. Caution should be exercised in patients with risk factors for dehydration, such as vomiting, diarrhea, or before or after major surgery.
The drug may mask signs and symptoms of infection because of its pharmacodynamic properties.
Due to the acetylsalicylic acid content of the drug
The drug should be used with caution in patients with gout, impaired renal or hepatic function, dehydration, uncontrolled arterial hypertension, glucose-6-phosphate dehydrogenase deficiency and diabetes.
Because of inhibition of platelet aggregation by ASA, the drug may lead to prolongation of bleeding time during and after surgical interventions (including minor ones, such as tooth extraction).
The drug should not be used concomitantly with anticoagulants and other drugs which disturb blood clotting without a physician’s supervision (see section “Interaction with other medicinal products”). Patients with blood clotting disorders should be closely monitored. Caution should be exercised in case of meto- or menorrhagia.
If a patient develops bleeding or gastrointestinal ulceration while taking the drug, it should be stopped immediately. At any time during treatment with any NSAID, potentially fatal bleeding, ulceration, and gastrointestinal perforations with or without a history of precursors and severe gastrointestinal complications may occur.
These complications tend to be more severe in older patients. Alcohol, glucocorticosteroids and NSAIDs may increase the risk of gastrointestinal bleeding (see section “Interaction with other medicinal products”). The drug may contribute to the development of bronchospasm and exacerbation of bronchial asthma (including bronchial asthma due to intolerance of analgesics) or other hypersensitivity reactions.
Risk factors include bronchial asthma, seasonal allergic rhinitis, nasal polyposis, chronic obstructive pulmonary disease, and chronic airway infections (particularly those associated with symptoms characteristic of allergic rhinitis). These events can also occur in patients with allergic reactions (e.g., skin reactions, including pruritus and urticaria) to other substances.
In these patients, special caution is recommended.
Children under 18 years of age should not be prescribed medicines containing acetylsalicylic acid as an antipyretic because they can increase the risk of Reye’s syndrome if there is a viral infection.
The symptoms of Reye syndrome include hyperpyrexia, prolonged vomiting, metabolic acidosis, nervous system and mental disorders, hepatomegaly and liver dysfunction, acute encephalopathy, respiratory distress, seizures, and coma.
ASK may distort the results of laboratory tests of thyroid function due to false positive low concentrations of levothyroxine (T4) and triiodothyronine (T3) (see section “Interaction with other medicines”).
Because of the paracetamol in the drug
Patients with impaired renal or hepatic function or alcohol dependence should use caution when prescribing the drug.
The risk of paracetamol poisoning is increased in patients taking other potentially hepatotoxic drugs or drugs that induce microsomal liver enzymes (e.g., rifampicin, isoniazid, chloramphenicol, sedatives and anticonvulsants, including phenobarbital, phenytoin and carbamazepine).
Patients with a history of alcoholism are at special risk of liver damage (see section “Interaction with other medicinal products”).
The drug may cause serious skin reactions, such as acute generalized episanthematous pustulosis, Stevens-Johnson syndrome, and toxic epidermal necrolysis, which can be fatal.
Patients should be informed about signs of serious skin reactions. The drug should be discontinued at the first signs of skin reactions or any other signs of hypersensitivity.
Due to the caffeine content of the drug
The drug should be prescribed with caution in patients with gout, hyperthyroidism, and arrhythmia.
Caffeine containing foods should be limited when using the product, as caffeine overdose may lead to nervousness, irritability, insomnia, and, in some cases, palpitations.
Impact on laboratory studies
High doses of ASA can distort the results of a number of clinical and biochemical laboratory tests.
The use of paracetamol may affect the results of uric acid determination by the phosphoric-tungstic acid method and the glycemiciglucose oxidase/peroxidase method.
Caffeine can reverse the effects of dipyridamole on myocardial blood flow, thereby distorting the results of this study. Caffeine should be withheld from the patient for 8-12 hours during the study.
Influence on driving and operating machinery
During the use of the drug, refrain from driving vehicles and operating other mechanisms, as the caffeine contained in the drug may cause impairment of concentration and reaction rate, and acetylsalicylic acid, also contained in the drug, may cause dizziness in rare cases.
Contraindications
Contraindications
– Gastric and duodenal ulcer in the acute phase;
– History of gastrointestinal bleeding or perforation, peptic ulcer;
– Severe renal or hepatic failure;
– Hemorrhagic diathesis, hypocoagulation (including hemophilia, hypoprothrombinemia);
– Surgical interventions accompanied by major bleeding;
– Pregnancy, breastfeeding period;
– Deficiency of glucose-6-phosphate dehydrogenase;
– Glaucoma;
– Hypersensitivity to components of the drug;
– Complete and incomplete combination of bronchial asthma, recurrent polyposis of the nose and sinuses and intolerance of ASA or other non-steroidal anti-inflammatory drugs (including history);
– Children under 15 years of age as an analgesic, with febrile syndrome under 18 years of age;
– Irritability, sleep disorders;
– Organic diseases of the cardiovascular system (including.ч. acute myocardial infarction, atherosclerosis;
– Severe arterial hypertension;
– Portal hypertension;
– Chronic heart failure NYHA functional class III-IV;
– Simultaneous administration of methotrexate more than 15 mg per week;
– Avitaminosis K;
– Hypoproteinemia.
With caution
. Gout, hyperuricemia, peptic ulcer, chronic obstructive pulmonary disease, hay fever, nasal polyposis, drug allergy, concomitant administration of methotrexate at a dose less than 15 mg/week, concomitant therapy with anticoagulants, nonsteroidal anti-inflammatory drugs, glucocorticosteroids, antiaggregants, selective serotonin reuptake inhibitors.
Mild to moderate renal failure, mild to moderate hepatic failure with elevated transaminases, benign hyperbilirubinemia (including Gilbert syndrome, alcoholic liver damage), alcoholism, epilepsy and proneness to seizures, old age, gout, arterial hypertension, chronic heart failure class I-II according to NYHA, coronary heart disease, cerebrovascular diseases, peripheral arterial disease, smoking.
Side effects
Side effects
The frequency of adverse drug reactions is classified according to the World Health Organization recommendations:
very common (≥ 1/10), common (≥ 1/100 to < 1/10), infrequent (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000), frequency unknown (cannot estimate frequency based on available data).
Infections and invasions:
Rarely, pharyngitis.
Metabolic and nutritional disorders:
Rarely, anorexia (decreased appetite).
Mental disorders:
often- nervousness;
infrequently- insomnia;
rarely- anxiety, euphoric mood, inner tension.
Nervous system disorders:
often- dizziness;
infrequent- tremor, paresthesias, headache;
rare- taste disorder, attention disorder, amnesia, impaired coordination of movement, hyperesthesia, pain in the paranasal sinuses.
Blood disorders:
rarely – decreased platelet aggregation, hypocoagulation, hemorrhagic syndrome (nasal bleeding, bleeding gums, purpura, etc.).
Disorders of the immune system:
frequency unknown-allergic reactions: skin rash, Quincke’s edema, bronchospasm, erythema multiforme (including Stevens-Johnson syndrome), toxic epidermal necrolysis (Lyell syndrome), Reye syndrome in children (metabolic acidosis, nervous system and mental disorders, vomiting, liver function disorders).
Visual disorders:
Rare visual disturbances.
Hearing disorders:
infrequent- tinnitus;
frequency unknown – deafness.
Cardiac disorders:
infrequent – tachycardia, arrhythmia.
Respiratory system, chest and mediastinum disorders:
Rare nasal bleeding, hypoventilation, rhinorrhea.
Disorders of the digestive system:
often-nausea, abdominal discomfort;
infrequently-dry mouth, vomiting, diarrhea, erosive-ulcerative gastrointestinal lesions;
rare- belching, flatulence, dysphagia, mouth paresthesias, increased salivation;
frequent unknown- gastralgia, gastrointestinal bleeding.
Hepatic and biliary tract disorders:
infrequent – hepatotoxicity;
frequency unknown – hepatic failure.
Skin and subcutaneous tissue disorders:
seldom – hyperhidrosis, itching, urticaria.
Musculoskeletal and connective tissue disorders:
seldom – musculoskeletal stiffness, neck pain, back pain, muscle cramps.
Renal and urinary tract disorders:
infrequent- nephrotoxicity;
frequency unknown- renal failure, renal damage with papillary necrosis.
General disorders and disorders at the site of administration:
infrequent- fatigue, increased excitability;
rarely- asthenia, heaviness in the chest.
There are no data about intensification or expansion of the spectrum of adverse effects of individual components when they are used in combination according to the instructions for medical use.
If you have side effects mentioned in the instructions, or if they worsen, or if you notice any other side effects not mentioned in the instructions, inform your physician.
Overdose
Overdose
Acetylsalicylic acid
In mild intoxication – dizziness, tinnitus, deafness, increased sweating, nausea, vomiting, headache and confusion. Occurs at plasma concentrations of 150-300 mcg/mL. Treatment is dose reduction or withdrawal of therapy.
At concentrations above 300 µg/ml, more severe intoxication occurs, manifested by hyperventilation, fever, anxiety, ketoacidosis, respiratory alkalosis and metabolic acidosis. Central nervous system depression may lead to coma, and cardiovascular collapse and respiratory failure may also occur.
The highest risk of chronic intoxication is seen in children and the elderly when more than 100 mg/kg/day is taken for several days.
Treatment
If ingestion of more than 120 mg/kg of salicylates is suspected, multiple oral activated charcoal injections are given within the last hour.
If more than 120 mg/kg of salicylates are ingested, their plasma concentration should be determined, although it is impossible to predict their severity based on this figure alone, clinical and biochemical parameters must also be considered.
If plasma concentrations exceed 500 µg/ml (350 µg/ml for children younger than 5 years), intravenous sodium bicarbonate administration effectively removes salicylates from plasma. If plasma concentrations exceed 700 µg/ml (lower concentrations in children and the elderly) or in severe metabolic acidosis, hemodialysis or hemoperfusion is the therapy of choice.
Paracetamol overdose
. In overdose, intoxication is possible, especially in elderly patients, children, patients with liver disease (caused by chronic alcoholism), in patients with nutritional disorders, as well as in patients taking microsomal liver enzyme inducers, which may lead to fulminant hepatitis, liver failure, cholestatic hepatitis, cytolytic hepatitis, in the above cases – sometimes with fatal outcome.
The clinical picture of acute overdose develops within 24 hours after taking paracetamol.
Symptoms: gastrointestinal disorders (nausea, vomiting, decreased appetite, feeling of discomfort in the abdomen and (or) abdominal pain), pale skin. When administered to adults 7.5 g or more or children more than 140 mg/kg at a time, cytolysis of hepatocytes occurs with complete and irreversible liver necrosis, the development of liver failure, metabolic acidosis and encephalopathy, which can lead to coma and death.
12-48 hours after administration of paracetamol an increase in the activity of microsomal liver enzymes, lactate dehydrogenase, bilirubin concentration and prothrombin decrease is noted. Clinical symptoms of liver damage appear 2 days after overdose of the drug and reach their maximum on the 4th-6th day.
Treatment
Hospitalization immediately.
The quantification of plasma paracetamol prior to treatment as soon as possible after overdose; Administration of SH-group donors and precursors of glutathione synthesis – methionine and acetylcysteine – most effective in the first 8 hours.
The need for additional therapeutic measures (further administration of methionine, IV administration of acetylcysteine) is determined depending on the concentration of paracetamol in the blood, as well as on the time elapsed after its administration.
Symptomatic treatment.
Laboratory studies of microsomal liver enzyme activity should be performed at the beginning of treatment and every 24 h thereafter.
In most cases, microsomal liver enzyme activity normalizes within 1-2 weeks. In very severe cases, liver transplantation may be necessary.
Caffeine
. Common symptoms include gastralgia, agitation, delirium, anxiety, nervousness, restlessness, insomnia, mental agitation, muscle twitching, confusion, seizures, dehydration, rapid urination, hyperthermia, headache, increased tactile or pain sensitivity, nausea and vomiting (sometimes with blood), tinnitus.
In severe overdose, hyperglycemia may occur. Cardiac disorders are manifested by tachycardia and arrhythmia.
The treatment is dose reduction or caffeine withdrawal.
Pregnancy use
Pregnancy use
Ascophen-P® is contraindicated in pregnancy and during breast-feeding.
Similarities
Similarities
Coffil-plus, Citramon Ultra, Citramon, Citramon P, Citramon-ExtraCap, Ascophen ULTRA, Brustrio
Additional information
| Weight | 0.010 kg |
|---|---|
| Shelf life | 3 years. Do not use after the expiration date printed on the package. |
| Conditions of storage | In a dry, light-protected place at a temperature not exceeding 25 °C |
| Manufacturer | Pharmstandard-Leksredstva, Russia |
| Medication form | pills |
| Brand | Pharmstandard-Leksredstva |
Other forms…
Related products
Buy Ascophen-P, tablets 10 pcs with delivery to USA, UK, Europe and over 120 other countries.