No products in the cart.
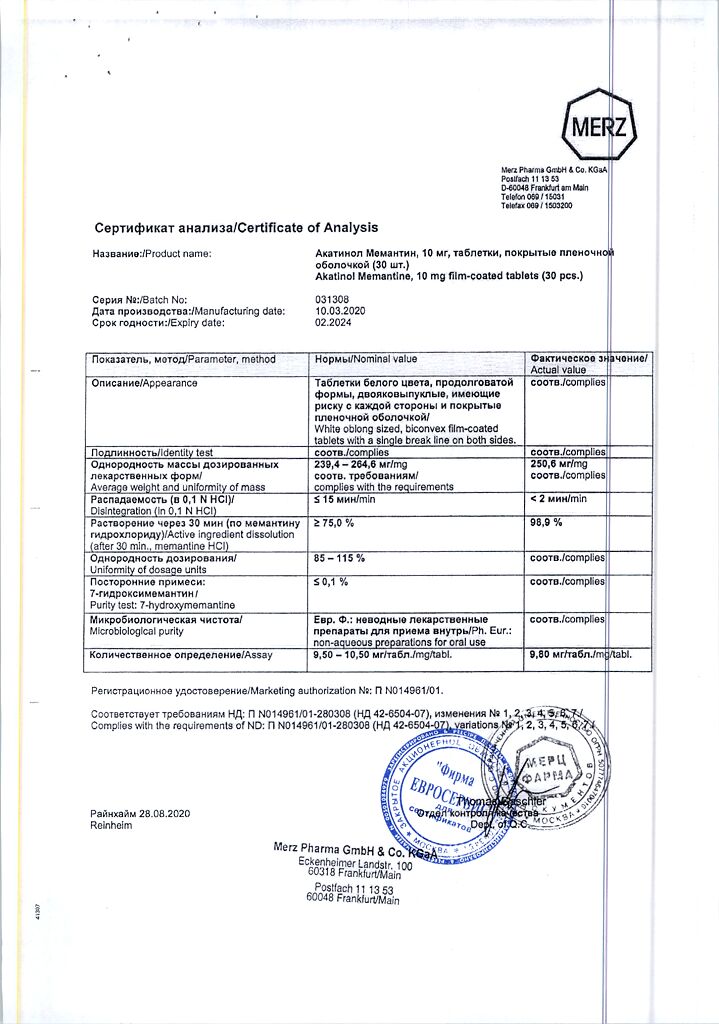
Akatinol Memantine, 10 mg 30 pcs
€45.95 €38.29
Description
Akatinol Memantine is a myorelaxant of central action.
Being a non-competitive antagonist of N-methyl-D-aspartate (NMDA)-receptors it has modulating effect on glutamatergic system.
It regulates ion transport, blocks calcium channels, normalizes membrane potential, improves nerve impulse transmission, improves cognitive processes, memory and learning ability, and increases daily activity.
Indications
Indications
Acquired dementia, Attention and memory impairment, Concussion and other brain injuries, Alzheimer’s disease, Stroke aftermath, Alcoholism
- Dementia of Alzheimer’s type, vascular dementia, mixed dementia of all degrees of severity.
- Decreased memory, concentration and learning ability.
- Cerebral and spinal spastic syndrome, including due to craniocerebral injury, multiple sclerosis, stroke.
.
Active ingredient
Active ingredient
Memantine
Composition
Composition
Active substance:
Memantine hydrochloride 10 mg;
Ancillary substances:
Lactose;
MCC;
Colloidal silicon dioxide;
talc;
magnesium stearate:
methacrylic acid copolymer;
sodium lauryl sulfate;
polysorbate 80;
triacetin;
simethicone emulsion;
talt.
How to take, the dosage
How to take, the dosage
Akatinol Memantine is taken orally,with meals. Dosing regimen is set individually. It is recommended to start treatment with prescribing the least effective doses.
In adults with dementia during the 1st week of therapy – in a dose of 5 mg/day, the 2nd week – in a dose of 10 mg/day, the 3rd week – in a dose of 15-20 mg/day. If necessary, further weekly dose increase by 10 mg to reach a daily dose of 30 mg.
The optimal dose is reached gradually, increasing the dose each week.
Interaction
Interaction
Concomitant use with L-dopa drugs, dopamine agonists, anticholinergic agents may increase their effect.
Concomitant use with barbiturates, neuroleptics may decrease the effect of the latter. Co-administration may change (increase or decrease) the effect of dantrolene or baclofen; therefore the doses of the drugs should be selected individually. Concomitant administration with amantadine, ketamine and dexamethorphan should be avoided.
Possible increase in plasma levels of cimetidine, procainamide, quinidine, quinine and nicotine when concomitantly administered with memantine. There may be a decrease in levels of hydrochlorothiazide when concomitantly taken with memantine.
Special Instructions
Special Instructions
Patients with moderate to severe dementia with Alzheimer’s disease usually have impaired ability to drive vehicles and operate complex machinery.
In addition, memantine may cause changes in reaction speed, so patients treated as outpatients should use extra caution when driving or operating machinery.
Contraindications
Contraindications
- Individual hypersensitivity to the drug;
- explicit renal dysfunction;
- breastfeeding;
- children under 18 years of age (due to insufficient data).
With caution:
- thyrotoxicosis;
- epilepsy;
- convulsions (including.
- myocardial infarction
- heart failure.
Side effects
Side effects
Adverse reactions are classified according to clinical manifestations (according to the involvement of certain organ systems) and by frequency of occurrence: Very common – ≥1/10; common – ≥1/100-
In the body as a whole – general adverse reactions Frequent Headache
Rarely Fatigue Infections
Rarely Fungal infections
Mental disorders Frequently Sleepiness
/p>
Rarely Confusion
Rarely Hallucinations
Frequency not determined
Psychotic reactions
Cardiovascular disorders
Rarely Hypertension
/p>
Rarely Venous Thrombosis/Thromboembolism
Gastrointestinal Disorders
Often Constipation
Rarely Nausea, Vomiting
Frequency not determined Pancreatitis
Disorders of the central and peripheral nervous systembr>
Often Dizziness
Rarely Gait disturbance
Very rarely Seizures
/p>
Hallucinations have been observed mainly in patients with Alzheimer’s disease at the stage of severe dementia.
There have been anecdotal reports of these adverse reactions when the drug is used in clinical practice (data obtained after the drug was commercially available).
Overdose
Overdose
Symptoms: increased severity of side effects.
Treatment: gastric lavage, administration of activated charcoal, symptomatic therapy.
Pregnancy use
Pregnancy use
It is contraindicated in pregnancy. Breastfeeding should be stopped during treatment.
Similarities
Similarities
Noogeron, Maruksa, Memantal, Memaneirin, Memantine
Additional information
| Weight | 0.018 kg |
|---|---|
| Shelf life | 4 years |
| Conditions of storage | At a temperature not exceeding 25 °C |
| Manufacturer | Rottendorf Pharma GmbH, Germany |
| Medication form | pills |
| Brand | Rottendorf Pharma GmbH |
Other forms…
Related products
Buy Akatinol Memantine, 10 mg 30 pcs with delivery to USA, UK, Europe and over 120 other countries.