No products in the cart.
Description
Pharmgroup:
an antiviral agent.
Pharmic action:
The antiviral drug is a synthetic analog of thymidine nucleoside.
In infected cells containing viral thymidine kinase is phosphorylated and converted into acyclovir monophosphate. Under the influence of guanylate cyclase acyclovir monophosphate is converted into diphosphate and under the influence of several cellular enzymes into triphosphate. High selectivity and low toxicity for humans is caused by the absence of necessary enzyme for formation of acyclovir triphosphate in intact cells of macroorganism.
Aciclovir triphosphate, “being built” into the DNA synthesized by the virus, blocks virus multiplication. Specificity and very high selectivity of action are also due to its preferential accumulation in cells affected by the herpes virus.
It is highly active against Herpes simplex viruses of types 1 and 2; viruses causing chicken pox and shingles (Varicella zoster); Epstein-Barr virus (types of viruses are listed in ascending order of MAC value of acyclovir).
It is moderately active against CMV.
In case of herpes it prevents forming of new rash elements, reduces the possibility of cutaneous dissemination and visceral complications, accelerates crusts forming, reduces pain in the acute phase of herpes zoster. It has an immunostimulatory effect.
Indications
Indications
Treatment of primary and recurrent severe genital herpes (including in patients with impaired immunity) – orally and parenterally;
prevention of frequently recurrent (6 or more cases per year) genital herpes (including in patients with impaired immunity) – orally;
treatment of primary and recurrent herpes simplex with damage to the skin and mucous membranes, caused by the Herpes simplex virus types 1 and 2, in patients with impaired immunity – orally, parenterally;
prevention of herpes simplex in patients with impaired immunity (including after transplantation and taking immunosuppressive drugs, HIV-infected patients, during chemotherapy) – orally;
encephalitis caused by the Herpes simplex virus types 1 and 2 – parenterally;
treatment of herpes zoster caused by the Varicella zoster virus, in adults – orally; in patients with impaired immunity and with generalized herpes zoster in patients with unimpaired immunity – parenterally;
prevention of herpes zoster caused by the Varicella zoster virus, after the initial period of using acyclovir for parenteral use in all patients with impaired immunity (including after transplantation and when taking immunosuppressive drugs, HIV-infected patients, during chemotherapy) – orally;
herpes zoster with eye damage – orally, parenterally;
generalized infection in newborns caused by the Herpes simplex virus – parenterally;
chicken pox in patients with unimpaired immunity within 24 hours after the appearance of a typical rash – orally, in patients with impaired immunity – parenterally.
Pharmacological effect
Pharmacological effect
Pharmaceutical group:
antiviral agent.
Pharmaceutical action:
The antiviral drug is a synthetic analogue of thymidine nucleoside.
In infected cells containing viral thymidine kinase, phosphorylation occurs and is converted to acyclovir monophosphate. Under the influence of acyclovir guanylate cyclase, monophosphate is converted into diphosphate and, under the action of several cellular enzymes, into triphosphate. High selectivity of action and low toxicity to humans are due to the absence of the necessary enzyme for the formation of acyclovir triphosphate in intact cells of the macroorganism.
Acyclovir triphosphate, “integrating” into the DNA synthesized by the virus, blocks the reproduction of the virus. The specificity and very high selectivity of the action are also due to its predominant accumulation in cells affected by the herpes virus.
Highly active against Herpes simplex virus types 1 and 2; the virus that causes chickenpox and herpes zoster (Varicella zoster); Epstein-Barr virus (types of viruses are indicated in increasing order of the MIC of acyclovir).
Moderately active against CMV.
In case of herpes, it prevents the formation of new rash elements, reduces the likelihood of skin dissemination and visceral complications, accelerates the formation of crusts, and reduces pain in the acute phase of herpes zoster. Has an immunostimulating effect.
Special instructions
Special instructions
Acyclovir should be prescribed with caution to patients with impaired renal function and elderly patients due to an increase in the half-life of acyclovir.
When using the drug, it is necessary to ensure the supply of a sufficient amount of fluid.
The earlier it is started, the higher the effectiveness of treatment when using the ointment.
In patients with immunodeficiency, with multiple repeated courses of treatment, viral resistance to acyclovir sometimes develops.
When taking the drug, kidney function should be monitored (blood urea and plasma creatinine levels).
Active ingredient
Active ingredient
Acyclovir
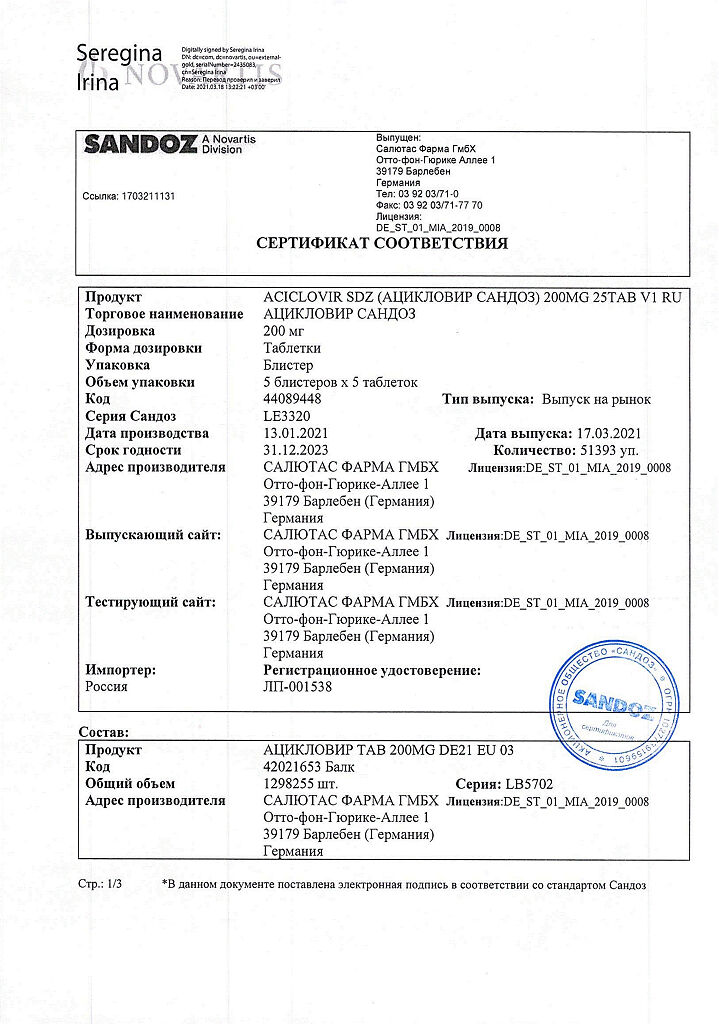
Composition
Composition
1 tablet contains:
active ingredient:
acyclovir 200 mg.
Pregnancy
Pregnancy
Use during pregnancy is only possible if the expected benefit to the mother outweighs the potential risk to the fetus.
Contraindications
Contraindications
Hypersensitivity (including to valacyclovir), children under 3 years of age (for solid dosage forms).
With caution. Pregnancy, lactation period.
Side Effects
Side Effects
From the digestive system:
nausea, vomiting, diarrhea, abdominal pain; rarely – a reversible increase in bilirubin content and the activity of liver enzymes.
Interaction
Interaction
When used simultaneously with probenecid, the average half-life of acyclovir increases and the clearance of acyclovir decreases.
When taken simultaneously with nephrotoxic drugs, the risk of developing renal dysfunction increases.
Overdose
Overdose
Ingestion of 20 g of acyclovir has been reported. Symptoms: agitation, coma, convulsions, lethargy. Precipitation of acyclovir in the renal tubules is possible if its concentration exceeds the solubility in the renal tubules (2.5 mg/ml).
Overdose with parenteral administration (with bolus administration, or use in high doses, or in patients whose water and electrolyte balance is not properly controlled): increased urea nitrogen, hypercreatininemia, impaired renal function, lethargy, convulsions, coma.
Treatment: maintenance of vital functions, hemodialysis.
Storage conditions
Storage conditions
Store in a dry place, protected from light, out of reach of children, at a temperature not exceeding 25°C.
Manufacturer
Manufacturer
Salutas Pharma GmbH, Germany
Additional information
| Conditions of storage | Store in a dry, protected from light, out of the reach of children, at a temperature not exceeding 25 ° C. |
|---|---|
| Manufacturer | Salutas Pharma GmbH, Germany |
| Medication form | pills |
| Brand | Salutas Pharma GmbH |
Other forms…
Related products
Buy Aciclovir Sandoz, tablets 200 mg 25 pcs with delivery to USA, UK, Europe and over 120 other countries.