No products in the cart.
Trombital Forte, 150 mg+30.39 mg 30 pcs
€1.00
Out of stock
(E-mail when Stock is available)
Description
Creases aggregation, platelet adhesion and thrombosis by inhibiting the synthesis of thromboxane A2 in platelets. Antiplatelet effect lasts for 7 days after a single dose (more pronounced in men than in women).
Acetylsalicylic acid reduces lethality and risk of myocardial infarction in unstable angina, it is effective in primary prevention of cardiovascular diseases, especially in men over 40 years old and in secondary prevention of myocardial infarction.
Suppresses prothrombin synthesis in the liver and increases prothrombin time. Increases the fibrinolytic activity of the blood plasma and reduces the concentration of vitamin K-dependent clotting factors (II, VII, IX, X). Increases the risk of hemorrhagic complications during surgical interventions, increases the risk of bleeding during anticoagulant therapy.
Acetylsalicylic acid in high doses also has anti-inflammatory, analgesic and antipyretic effects.
In high doses, acetylsalicylic acid stimulates excretion of uric acid (disrupts its reabsorption in the renal tubules).
Blockade of cyclooxygenase-1 in gastric mucosa leads to inhibition of gastroprotective prostaglandins, which may cause mucosal ulceration and subsequent bleeding.
Magnesium hydroxide, which is part of the drug Trombital Forte, protects the gastrointestinal mucosa from the effects of acetylsalicylic acid.
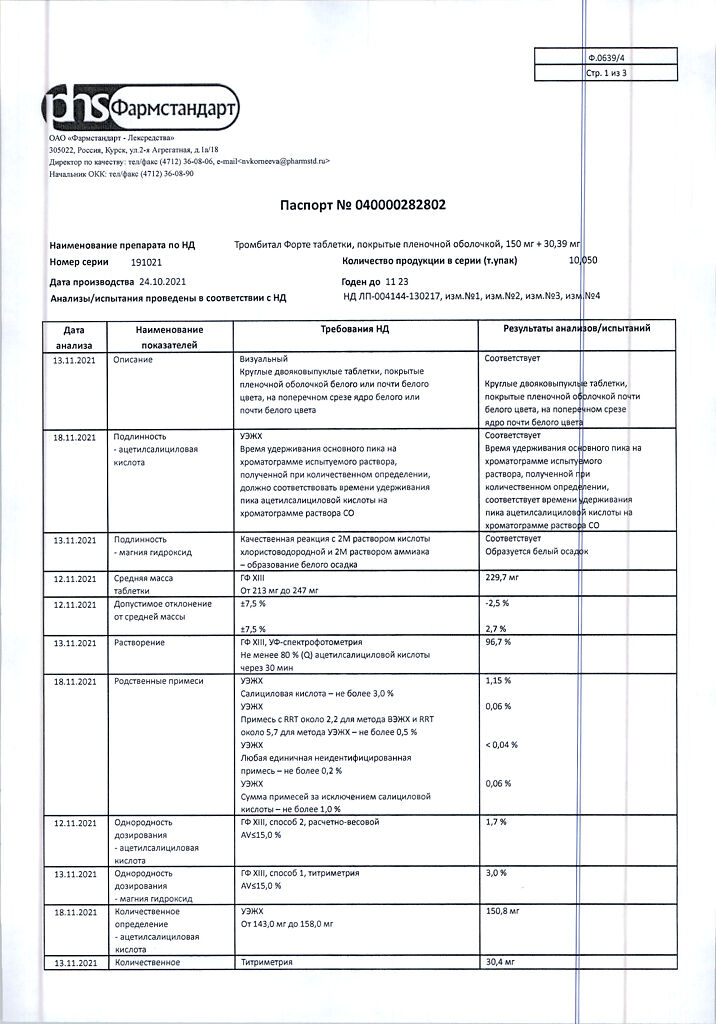
Indications
Indications
Prevention of heart attacks and strokes, Thrombosis Prevention
Primary prevention of cardiovascular diseases such as thrombosis and acute heart failure in the presence of risk factors (e.g., diabetes, hyperlipidemia, arterial hypertension, obesity, smoking, old age).
Prevention of cardiovascular disease: recurrent myocardial infarction, blood vessel thrombosis.
Prevention of thromboembolism after surgical interventions on blood vessels (e.g., aorto-coronary bypass surgery, percutaneous transluminal coronary angioplasty, etc.).
Unstable angina (including suspected acute myocardial infarction).
Active ingredient
Active ingredient
Acetylsalicylic acid, Magnesium hydroxide
Composition
Composition
Active ingredients:
acetylsalicylic acid – 150.00 mg, magnesium
hydroxide – 30.39 mg.
Auxiliary substances:
microcrystalline cellulose – 24.15 mg,
corn starch – 19.00 mg,
potato starch – 4.00 mg,
magnesium stearate – 0.30 mg.
Shell:
hypromellose (hydroxypropyl methylcellulose 15 cPs) – 1.20 mg,
macrogol (polyglycol 4000) – 0.24 mg,
talc – 0.72 mg.
How to take, the dosage
How to take, the dosage
The drug is taken orally with water. The tablet can be swallowed whole, chewed or crushed.
The drug is intended for long-term use. The duration of treatment with Trombital Forte is determined by the physician. One tablet of Trombital Forte contains 150 mg of acetylsalicylic acid.
If necessary, the daily dose of acetylsalicylic acid may be reduced to 75 mg, at the discretion of the physician (see section “Special information”). Primary prevention of cardiovascular diseases, such as thrombosis and acute heart failure in the presence of risk factors (e.g., diabetes mellitus, hyperlipidemia, arterial hypertension, obesity, smoking, old age) – 1 tablet once daily.
Prevention of cardiovascular diseases: repeated myocardial infarction, blood vessel thrombosis – 1 tablet once a day.
Prophylaxis of thromboembolism after surgical interventions on blood vessels (for example, aorto-coronary bypass surgery, percutaneous transluminal coronary angioplasty and others) – 1 tablet once a day.
Unstable angina pectoris (including suspected acute myocardial infarction) – 1 tablet once daily.
In unstable angina with suspected acute myocardial infarction, the first tablet should be chewed for faster absorption.
The drug should only be used according to the indication, route of administration, and dosage listed in the directions.
Interaction
Interaction
Interaction with other drugs
Concomitant use of acetylsalicylic acid increases the effect of the following drugs:
– methotrexate by reducing renal clearance and displacing it from protein binding, the combination of acetylsalicylic acid with methotrexate is accompanied by an increased frequency of side effects from the hematopoietic organs;
– narcotic analgesics, other NSAIDs;
– heparin and indirect anticoagulants due to platelet malfunction and displacement of indirect anticoagulants from bonding to plasma proteins;
– Thrombolytic, antiplatelet and anticoagulant drugs (ticlopidine);
– Digoxin due to decrease of its renal excretion;
– Hypoglycemic oral hypoglycemic agents (sulfonylurea derivatives) and insulin due to hypoglycemic properties of acetylsalicylic acid at high doses and displacement of sulfonylurea derivatives from plasma proteins;
– Valproic acid due to its displacement from plasma proteins.
Concomitant use of acetylsalicylic acid with ibuprofen reduces the cardioprotective effects of acetylsalicylic acid.
Combination of acetylsalicylic acid with anticoagulants, thrombolytics and antiaggregants is accompanied by an increased risk of bleeding.
Simultaneous use with acetylsalicylic acid increases the concentration of barbiturates and lithium salts in blood plasma.
Increasing the elimination of salicylates, systemic glucocorticosteroids weaken their effects.
Glucocorticosteroids, ethanol and ethanol-containing drugs increase the negative effects on the mucous membrane of the gastrointestinal tract and increase the risk of gastrointestinal bleeding.
Simultaneous use of acetylsalicylic acid with ethanol increases the toxic effects of ethanol on the central nervous system.
Acetylsalicylic acid weakens the effect of uricosuric drugs – benzbromaron, probenecid (reducing the uricosuric effect, due to competitive suppression of renal tubular excretion of uric acid), angiotensin-converting enzyme inhibitors (there is a dose-dependent decrease in glomerular filtration rate (GFR) as a result of inhibition of prostaglandins that have a vasodilator effect, and, accordingly, a decrease in antihypertensive effect; clinically significant decrease of GFR is noted with daily dose of acetylsalicylic acid over 160 mg), diuretics (when used together with acetylsalicylic acid in high doses, decrease of glomerular filtration rate is noted due to decrease of prostaglandin synthesis in kidneys).
Antacids and colestiramine reduce absorption of acetylsalicylic acid.
If you use the above or other medicines (including over-the-counter), consult your doctor before using the drug Trombital Forte.
Addiction
Can occur after a single large dose or when using the drug for a long time. If a single dose of acetylsalicylic acid is less than 150 mg/kg, acute poisoning is considered mild, 150-300 mg/kg – moderate severity, and when higher doses are used – severe.
Symptoms of mild to moderate overdose:
dizziness, tinnitus, hearing impairment, visual impairment, increased sweating, nausea, vomiting, headache, confusion, tachypnea, hyperventilation, respiratory alkalosis.
Treatment: provocation of vomiting, repeated administration of activated charcoal, forced alkaline diuresis, restoration of water-electrolyte balance and acid-base balance.
Symptoms of overdose from moderate to severe:
– respiratory alkalosis with compensatory metabolic acidosis;
– hyperpyrexia (extremely high body temperature);
– respiratory disorders: hyperventilation, noncardiogenic pulmonary edema, respiratory depression, asphyxia;
– disorders on the part of the cardiovascular system: cardiac rhythm disorders, decreased blood pressure, depressed cardiac activity, collapse;
– disorders of the water-electrolyte balance: dehydration, impaired renal function from oliguria to the development of renal failure, characterized by hypokalemia, hypernatriemia, hyponatremia;
– disorders of glucose metabolism: hyperglycemia, hypoglycemia (especially in children), ketoacidosis;
– tinnitus, deafness;
– gastrointestinal bleeding;
– hematological disorders: From inhibition of platelet aggregation to coagulopathy, prolongation of prothrombin time, hypoprothrombinemia;
– neurological disorders: toxic encephalopathy and depression of central nervous system function (drowsiness, confusion, coma, seizures).
Treatment: immediate hospitalization to specialized departments for emergency therapy – gastric lavage, repeated administration of activated charcoal and laxatives, alkalinization of urine (indicated with salicylate levels above 500 mg/l, is provided by intravenous infusion of sodium bicarbonate – 88 mEq in 1 L of 5% glucose solution, at a rate of 10-15 ml/kg/h), restoration of circulating blood volume and induction of diuresis (achieved by the introduction of sodium bicarbonate in the same dose and dilution, repeated 2-3 times); note that intensive fluid infusion in elderly patients may lead to pulmonary edema. The use of acetozolamide to alkalize the urine is not recommended (may cause acidemia and increase the toxic effects of salicylates). When alkaline diuresis is performed, pH values between 7.5 and 8 should be achieved. Hemodialysis is indicated when plasma levels of salicylates exceed 1000 mg/l, and in patients with chronic poisoning – 500 mg/l or lower, if indicated (refractory acidosis, progressive deterioration of condition, severe central nervous system damage, pulmonary edema and renal failure). In pulmonary edema, artificial ventilation with oxygen-enriched mixture in positive pressure mode at the end of exhalation is indicated; hyperventilation and osmotic diuresis are used to treat brain edema.
The greatest risk of developing chronic intoxication is noted in elderly persons when administered for several days over 100 mg/kg/day. In children and elderly patients the initial signs of salicylation (nausea, vomiting, tinnitus, visual disturbances, dizziness, headache, general malaise) are not always noticeable, so it is advisable to periodically determine plasma salicylate content.
Special Instructions
Special Instructions
The drug should be used as prescribed by a physician.
The tablets of the drug Trombital Forte are film-coated, have no risks and are not intended to be divided, so if the doctor recommends reducing the daily dose of acetylsalicylic acid to 75 mg, then a switch to another drug containing 75 mg of acetylsalicylic acid in 1 tablet is required.
Acetylsalicylic acid may provoke bronchospasm, as well as causing attacks of bronchial asthma and other hypersensitivity reactions.
Risk factors include a history of bronchial asthma, hay fever, nasal polyposis, chronic respiratory disease, and allergic reactions to other drugs (e.g., skin reactions, itching, urticaria).
Acetylsalicylic acid may cause bleeding of varying severity during and after surgical interventions. Several days before the planned surgical intervention, the risk of bleeding should be assessed in comparison with the risk of ischemic complications in patients taking low-dose acetylsalicylic acid.
If the risk of bleeding is significant, acetylsalicylic acid should be temporarily discontinued. The combination of acetylsalicylic acid with anticoagulants, thrombolytics and antiplatelet agents is accompanied by an increased risk of bleeding.
Acetylsalicylic acid in low doses may provoke the development of gout in predisposed patients (who have reduced excretion of uric acid). The combination of acetylsalicylic acid with methotrexate is accompanied by an increased incidence of side effects on the hematopoietic organs.
High doses of acetylsalicylic acid have hypoglycemic effects, which should be kept in mind when prescribing it for patients with diabetes who are on oral hypoglycemic agents and insulin.
When combined use of systemic glucocorticosteroids and salicylates, it should be remembered that during treatment, the concentration of salicylates in blood is decreased, and after withdrawal of systemic glucocorticosteroids an overdose of salicylates is possible.
The combination of acetylsalicylic acid with ibuprofen is not recommended in patients with increased risk of cardiovascular disease: concomitant use with ibuprofen has decreased antiplatelet effects of acetylsalicylic acid at doses up to 300 mg, which reduces the cardioprotective effects of acetylsalicylic acid.
Extending the dose of acetylsalicylic acid beyond therapeutic doses is associated with the risk of gastrointestinal bleeding.
Long-term use of low doses of acetylsalicylic acid as antiplatelet therapy requires caution in elderly patients due to the risk of gastrointestinal bleeding.
The concomitant use of acetylsalicylic acid with alcohol increases the risk of gastrointestinal mucosal damage and prolonged bleeding time.
In long-term use of the drug, periodic general blood tests and fecal occult blood tests should be performed.
Impact on the ability to drive:
At the time of treatment with acetylsalicylic acid, caution should be exercised when driving vehicles and engaging in potentially hazardous activities that require increased concentration and rapid psychomotor reactions.
Features
Features
Acetylsalicylic acid is absorbed from the gastrointestinal tract almost completely. The half-life of acetylsalicylic acid is about 15 minutes, because with the participation of enzymes acetylsalicylic acid is rapidly hydrolyzed into salicylic acid in the intestine, liver and blood plasma. The half-life of salicylic acid is about 3 hours, but it can be significantly increased by simultaneous administration of large doses of acetylsalicylic acid (more than 3.0 g) as a result of enzyme systems saturation.
The bioavailability of acetylsalicylic acid is 70%, but this value varies considerably as acetylsalicylic acid undergoes presystemic hydrolysis (gastrointestinal mucosa, liver) into salicylic acid by enzymes. The bioavailability of salicylic acid is 80-100%.
Used doses of magnesium hydroxide do not affect the bioavailability of acetylsalicylic acid.
Reduces aggregation, platelet adhesion and thrombosis by inhibiting the synthesis of thromboxane A2 in platelets. Antiplatelet effect lasts for 7 days after a single dose (more pronounced in men than in women).
Acetylsalicylic acid reduces mortality and risk of myocardial infarction in unstable angina, is effective in primary prevention of cardiovascular diseases, especially myocardial infarction in men over 40 years old and in secondary prevention of myocardial infarction.
Suppresses prothrombin synthesis in the liver and increases prothrombin time. Increases plasma fibrinolytic activity and decreases the concentration of vitamin K-dependent clotting factors (II, VII, IX, X). Increases the risk of hemorrhagic complications during surgical interventions, increases the risk of bleeding during anticoagulant therapy.
Acetylsalicylic acid in high doses also has anti-inflammatory, analgesic, antipyretic effects.
In high doses, acetylsalicylic acid stimulates excretion of uric acid (disrupts its reabsorption in the renal tubules).
Blockade of cyclooxygenase-1 in gastric mucosa leads to inhibition of gastroprotective prostaglandins, which may cause mucosal ulceration and subsequent bleeding.
Magnesium hydroxide, which is part of the drug Trombital Forte, protects the mucous membrane of the gastrointestinal tract from the effects of acetylsalicylic acid.
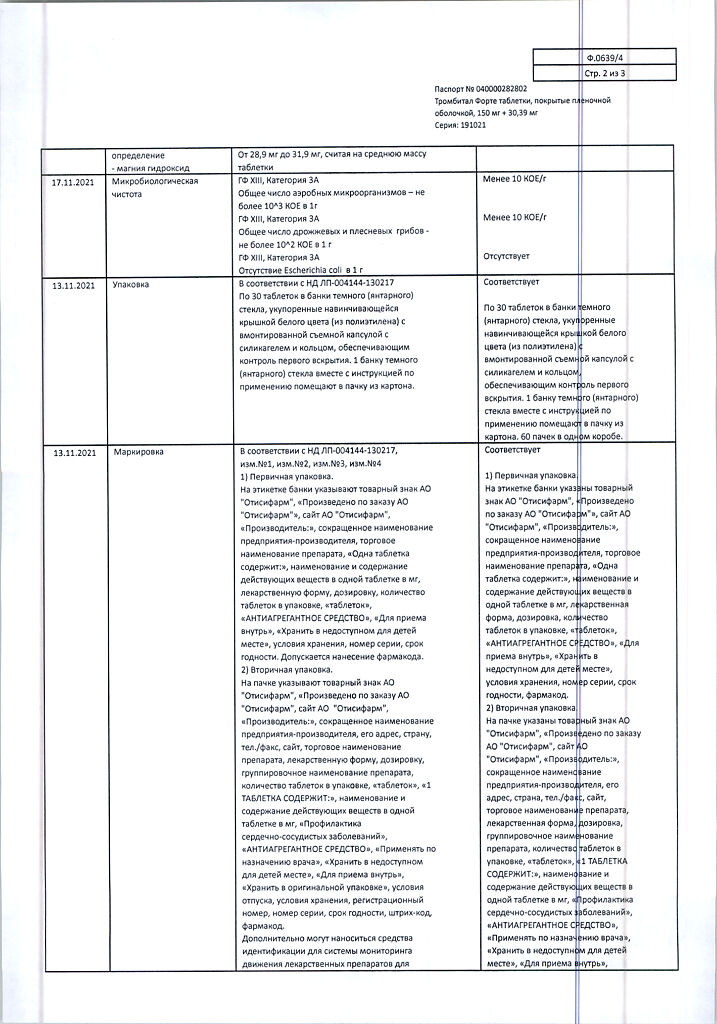
Contraindications
Contraindications
- High sensitivity to acetylsalicylic acid, excipients of the drug and other non-steroidal anti-inflammatory drugs (NSAIDs);
- Cerebral hemorrhage;
- sensitivity to bleeding (vitamin K deficiency, thrombocytopenia, hemorrhagic diathesis);
- erosive ulcerative lesions of the gastrointestinal tract (acute phase);
- gastrointestinal bleeding;
- bronchial asthma induced by taking salicylates and other NSAIDs;
- combination of bronchial asthma, recurrent nasal and paranasal sinus polyposis with intolerance to acetylsalicylic acid;
- current use of methotrexateconcomitant administration of methotrexate at a dose of 15 mg per week or more;
- pregnancy (I and III trimesters);
- lactation period;
- glucose-6-phosphate dehydrogenase deficiency;
- severe renal failure (creatinine clearance (CK) less than 30 ml/min);
- severe hepatic failure (Child-Pugh class B and C);
- chronic heart failure III and functional class IV according to the NYHA classification;
- children under 18 years of age.
.
Side effects
Side effects
The adverse events listed below are distributed by frequency of occurrence according to the following gradation: very frequently (with an incidence greater than 1/10), frequently (with an incidence of at least 1/100 but less than 1/10), infrequently (with an incidence of at least 1/1000 but less than 1/100), rarely (with an incidence of at least 1/10000 but less than 1/1000), very rarely (with an incidence less than 1/10000), including individual reports.
Blood and lymphatic system disorders: very common – increased bleeding (hematoma, nasal bleeding, bleeding gums, bleeding from the urinary tract); rare – anemia; very rare – hypoprothrombinemia, thrombocytopenia, neutropenia, aplastic anemia, eosinophilia, agranulocytosis; unknown frequency – leukopenia.
There have been reports of serious cases of bleeding, which include gastrointestinal bleeding and cerebral hemorrhage (especially in patients with hypertension who have not reached target blood pressure and/or receive concomitant therapy with anticoagulants), which in some cases may be life-threatening.
Bleeding may lead to the development of acute or chronic posthemorrhagic/iron deficiency anemia (e.g., due to hidden bleeding) with corresponding clinical and laboratory signs and symptoms (asthenia, pallor, hypoperfusion). There have been reports of cases of hemolysis and hemolytic anemia in patients with severe forms of glucose-6-phosphate dehydrogenase deficiency.
Allergic reactions: common – urticaria, Quincke’s edema; infrequent – anaphylactic reactions, including angioedema; unknown frequency – skin rash, skin itching, rhinitis, nasal mucosal edema, cardio-respiratory distress syndrome, and severe reactions, including anaphylactic shock.
Nervous system disorders: frequently – headache, insomnia; infrequently – dizziness, somnolence; rarely – tinnitus, intracerebral hemorrhage; unknown frequency – hearing loss, which may be a sign of drug overdose (see section “Overdose”).
Respiratory system, thorax and mediastinum: often – bronchospasm.
Gastrointestinal tract disorders: very often – heartburn; often – nausea, vomiting; infrequent – abdominal pain, gastric and duodenal mucous membrane ulcers, including perforative (rare), gastrointestinal bleeding; rare – increased activity of “liver” enzymes; very rare – stomatitis, esophagitis, erosive lesions of the upper gastrointestinal tract, strictures, irritable bowel syndrome, colitis; unknown frequency – decreased appetite, diarrhea.
Urinary system disorders: unknown frequency – renal dysfunction and acute renal failure.
If you experience or worsen the side effects mentioned in the instructions, or if you notice any other side effects not mentioned in the instructions, tell your doctor.
Overdose
Overdose
Can occur after a single large dose or with prolonged use of the drug. If a single dose of acetylsalicylic acid is less than 150 mg/kg, acute poisoning is considered mild, 150-300 mg/kg – moderate severity, and with the use of higher doses – severe.
Symptoms of overdose from mild to moderate severity: dizziness, tinnitus, hearing impairment, visual impairment, increased sweating, nausea, vomiting, headache, confusion, tachypnea, hyperventilation, respiratory alkalosis.
Treatment: provocation of vomiting, repeated administration of activated charcoal, forced alkaline diuresis, restoration of water-electrolyte balance and acid-base balance.
Symptoms of overdose from moderate to severe:
- respiratory alkalosis with compensatory metabolic acidosis;
- hyperpyrexia (extremely high body temperature);
- respiratory disorders: hyperventilation, noncardiogenic pulmonary edema, respiratory depression, asphyxia;
- disorders of the cardiovascular system: cardiac rhythm disorders, decreased blood pressure, depressed cardiac activity, collapse;
- disorders of the water-electrolyte balance: dehydration, impaired renal function from oliguria to the development of renal failure, characterized by hypokalemia, hypernatriemia, hyponatremia;
- disorders of glucose metabolism: hyperglycemia, hypoglycemia (especially in children), ketoacidosis;
- tinnitus, deafness;
- gastrointestinal bleeding;
- hematological disorders: From inhibition of platelet aggregation to coagulopathy, prolongation of prothrombin time, hypoprothrombinemia;
- neurological disorders: toxic encephalopathy and depression of central nervous system function (drowsiness, confusion, coma, seizures).
Treatment: immediate hospitalization to specialized departments for emergency therapy – gastric lavage, repeated administration of activated charcoal and laxatives, alkalinization of urine (indicated when salicylate levels are above 500 mg/l, is provided by intravenous infusion of sodium bicarbonate – 88 mEq in 1 L of 5% glucose solution, at a rate of 10-15 ml/kg/h), restoration of circulating blood volume and induction of diuresis (achieved by the introduction of sodium bicarbonate in the same dose and dilution, repeated 2-3 times); it should be borne in mind that intensive fluid infusion in elderly patients may lead to pulmonary edema.
The use of acetozolamide to alkalize urine is not recommended (may cause acidemia and increase the toxic effects of salicylates). When performing alkaline diuresis, it is necessary to achieve pH values between 7.5 and 8.
Hemodialysis is indicated for plasma levels of salicylates greater than 1000 mg/l, and in patients with chronic poisoning, 500 mg/l or lower, if indicated (refractory acidosis, progressive deterioration, severe central nervous system damage, pulmonary edema and renal failure).
In case of pulmonary edema artificial ventilation with oxygen-enriched mixture in positive pressure mode at the end of exhalation is indicated; hyperventilation and osmotic diuresis are used to treat brain edema. The greatest risk of developing chronic intoxication is noted in elderly people when administered over 100 mg/kg/day for several days.
In children and elderly patients the initial signs of salicylation (nausea, vomiting, tinnitus, visual disturbances, dizziness, headache, general malaise) are not always noticeable, so it is advisable to periodically determine the plasma salicylate content.
Pregnancy use
Pregnancy use
The drug is contraindicated during pregnancy (I and III trimesters); lactation period;
Similarities
Similarities
Cardiomagnil, Trombital, Trombital Forte, Fazostabil
Additional information
| Weight | 0.074 kg |
|---|---|
| Shelf life | 2 years. |
| Conditions of storage | At a temperature not higher than 25 ° C. |
| Manufacturer | Pharmstandard-Leksredstva, Russia |
| Medication form | pills |
| Brand | Pharmstandard-Leksredstva |
Other forms…
Related products
Buy Trombital Forte, 150 mg+30.39 mg 30 pcs with delivery to USA, UK, Europe and over 120 other countries.