No products in the cart.
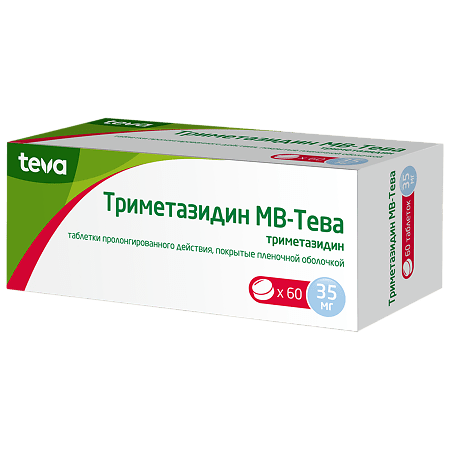
Trimetazidine MB-Teva, 35 mg 60 pcs
€13.64 €11.37
Description
Pharmacotherapeutic group: antihypoxant
ATX code: C01EB15
Pharmacological properties
Pharmacodynamics
Trimetazidine prevents intracellular decrease in adenosine triphosphatase (ATP) activity under conditions of ischemia and hypoxia, preserving energy metabolism and ensuring cell homeostasis by ensuring normal functioning of cell membrane ion channels and transmembrane sodium-potassium flow. It inhibits beta-oxidation of fatty acids by selectively blocking the enzyme 3-ketoacyl-CoA-thiolase, which enhances glucose oxidation. Cells in ischemia require less oxygen for energy in the process of glucose oxidation than in the process of beta-oxidation of fatty acids.
The switch of cellular energy metabolism from fatty acid oxidation to glucose oxidation underlies the pharmacological effects of trimetazidine. Under experimental conditions it has been shown that the drug:
- maintains the energy metabolism of the heart and neurosensory tissues under ischemia;
- reduces the severity of intracellular acidosis and changes in transmembrane ion flux occurring during ischemia;
- reduces the size of myocardial damage;
- does not adversely affect hemodynamic parameters.
In patients with ischemic heart disease, trimetazidine acts as a metabolic agent, maintaining sufficient intracellular high-energy phosphate activity in the myocardium. The anti-ischemic effect is achieved without affecting hemodynamics.
In patients with stenocardia trimetazidine:
- increases coronary reserve, thereby delaying the onset of exercise-induced ischemia beginning on day 15 of therapy;
- limits exercise-induced blood pressure fluctuations without significant changes in heart rate;
- reduces the frequency of angina attacks and the need for short-acting nitroglycerin;
- improves left ventricular contractile function in patients with coronary dysfunction.
Pharmacokinetics
When taken orally, trimetazidine is rapidly absorbed from the gastrointestinal (GI) tract, with maximum plasma concentration reached in an average of 5 hours. For more than 24 hours, the plasma concentration of trimetazidine is maintained at levels greater than or equal to 75% of maximum.
The equilibrium state of the drug concentration in blood is reached after 60 hours. Food intake has no effect on the pharmacokinetic properties of trimetazidine. The volume of distribution is 4.8 l/kg. The binding to plasma proteins is low and in vitro is 16%.
Trimetazidine is mainly excreted by the kidneys, mainly unchanged. When administered orally at a dose of 35 mg, the half-life in young healthy volunteers is on average 7 hours; in patients over 65 years of age, it is 12 hours.
The total renal clearance of trimetazidine directly correlates with creatinine clearance (CK), hepatic clearance decreases with age.
Patients over 75 years
In patients over 75 years of age, increased plasma exposure to trimetazidine may be observed as a result of age-related decline in renal function. No differences have been found regarding the safety of the drug in patients over 75 years of age compared to the general population.
Patients with impaired renal function
. Plasma exposure to trimetazidine was increased approximately 2.4-fold in patients with moderate renal dysfunction (CK 30-60 mL/min), and approximately 4-fold in patients with severe renal dysfunction (CK less than 30 mL/min) compared with healthy volunteers with normal renal function.
There were no findings regarding the safety of the drug in patients with impaired renal function compared to the general population.
Application in children and adolescents
The pharmacokinetics of trimetazidine in children and adolescents less than 18 years have not been studied.
Indications
Indications
Dizziness, Hearing impairment, Impaired spatial orientation, Vascular eye disease, Visual impairment, Tinnitus, Angina pectoris Long-term therapy for coronary heart disease: prevention of stable angina attacks as monotherapy or as part of combination therapy.
Active ingredient
Active ingredient
Trimetazidine
Composition
Composition
1 tablet contains: active ingredient trimetazidine dihydrochloride 35.00 mg; excipients (core): hypromellose 90.00 mg, calcium hydrophosphate dihydrate 141.00 mg, magnesium stearate 3.00 mg, colloidal silica 1.00 mg; excipients (coating): water gloss pink* 7.00 mg. *- Hypromellose 60.00%, stearic acid 5.00%, macrogol-6000 3.00%, glycerol 5.00%, titanium dioxide 26.75%, iron oxide red dye 0.25%.
How to take, the dosage
How to take, the dosage
Overly, with meals.
1 tablet (35 mg) 2 times a day (morning and evening). Tablets should be taken whole, without chewing, with water.
The duration of treatment is decided by the doctor. The result of the treatment is evaluated after three months. If the treatment is not effective, Trimetazidine MB-Teva should be discontinued.
Patients with renal impairment
In patients with moderate renal impairment (CK 30-60 ml/min), the recommended dose is 35 mg (1 tablet) in the morning with breakfast.
Elderly patients over 75 years
Elderly patients may have increased exposure to trimetazidine due to age-related decreased renal function. In patients with moderate renal impairment (CKD 30-60 ml/min), the recommended dose is 35 mg (1 tablet) in the morning with breakfast.
Caution should be exercised when adjusting the dose in elderly patients.
Interaction
Interaction
No cases of drug interactions have been described. Trimetazidine may be used concomitantly with heparin, low-molecular-weight heparin, indirect-acting anticoagulants, drugs used for lipid metabolism disorders, acetylsalicylic acid, beta-adrenoblockers, “slow” calcium channel blockers, cardiac glycosides.
Special Instructions
Special Instructions
If one or more doses of Trimetazidine MV-Teva are missed, the higher dose should not be taken at the next dose.
Trimetazidine MB-Teva is not intended to control angina attacks, nor is it indicated as an initial treatment for unstable angina or myocardial infarction. It should not be taken before hospitalization or in the first few days of hospitalization.
In case of an attack of angina, coronary pathology should be reassessed and the treatment used should be adjusted (drug treatment and possibly revascularization).
The drug Trimetazidine MB-Teva may cause or worsen the symptoms of parkinsonism (tremor, akinesia, increased muscle tone). Patients should be monitored regularly, especially in the elderly. In doubtful cases, the patient should be referred to a neurologist for appropriate examination.
The development of movement disorders, such as parkinsonian symptoms, restless legs, tremor, wobbly walking, may lead to withdrawal of trimetazidine.
The incidence of movement disorders is low, reversible, and resolves after discontinuation of the drug. If symptoms of parkinsonism persist more than 4 months after discontinuation of the drug, a neurologist should be consulted.
If unsteady walking or low muscle tone, especially when using antihypertensive therapy, the patient may fall (see side effects).
Trimetazidine should be administered with caution in patients who may have increased exposure to it:
- in moderate renal dysfunction (see section “Dosage”).
- in elderly patients over 75 years of age (see “Administration and Doses”);
- in severe hepatic impairment (see “Caution”).
Influence on driving and operating ability
Because of the possible development of dizziness and other side effects, caution should be exercised when driving vehicles and engaging in other activities requiring increased concentration and rapid psychomotor reactions.
Synopsis
Synopsis
Round biconvex pink film-coated tablets. A cross section of the tablets shows a white or almost white core.
Contraindications
Contraindications
- high sensitivity to any of the ingredients of the drug;
- severe renal impairment (CKR less than 30 ml/min);
- Parkinson’s disease, Parkinsonian symptoms, tremor, restless legs syndrome and other symptoms associated with movement disorders;
- pregnancy;
- breastfeeding period;
- children under 18 years of age (efficacy and safety not established).
Cautions
Patients with severe hepatic impairment, moderate renal impairment, and elderly patients over 75 years of age (increased exposure to trimetazidine may occur).
Side effects
Side effects
The following criteria are used to assess the incidence of side effects: very common (≥1/10); common (≥1/100, < 1/10); infrequent (≥1/1000, < 1/100); rare (≥1/10000, < 1/1000); very rare (< 1/10000), frequency unknown – based on available data it is not possible to estimate the frequency.
Blood and lymphatic system disorders: frequency unknown- agranulocytosis, thrombocytopenia, thrombocytopenic purpura.
Nervous system disorders: frequent – dizziness, headache; frequency unknown – symptoms of parkinsonism (tremor, akinesia, increased tone), “wobbly” gait, restless legs syndrome, other related movement disorders (usually reversible after discontinuation of therapy), sleep disorders (insomnia, somnolence).
Hearing and labyrinth disorders: frequency unknown- Vertigo.
Cardiac disorders: frequently – palpitations, extrasystole, tachycardia.
vascular disorders: frequently – arterial hypotension, orthostatic hypotension, which may be accompanied by general weakness, dizziness or loss of balance (falling), especially when concomitant use of hypotensive drugs, “flushes” of blood to the face.
Gastrointestinal tract disorders: frequent – abdominal pain, diarrhea, dyspepsia, nausea, vomiting; frequency unknown – constipation.
Liver and biliary tract disorders: frequency unknown – hepatitis.
Dermatological and subcutaneous tissue disorders: frequent – skin rash, itching, urticaria; frequency unknown – acute generalized eczematous pustulosis, angioedema.
General disorders and disorders at the site of administration: often – asthenia.
Overdose
Overdose
No cases of drug overdose have been reported. In case of overdose symptomatic treatment should be administered.
Pregnancy use
Pregnancy use
There are no data on the use of trimetazidine in pregnant women. Animal studies have shown no teratogenic effects. The potential risk of use in pregnant women is unknown. Trimetazidine MB-Teva is contraindicated during pregnancy. It is unknown whether trimetazidine is excreted with the breast milk, therefore if it is necessary to use Trimetazidine MV-Teva during lactation, breastfeeding should be stopped. No effect of trimetazidine on fertility has been established in animal studies of reproductive toxicity.
Similarities
Similarities
Predysin, Deprenorm MB, Trimectal MB, Trimetazidine, Antisthen MB, Preductal OD , Trimetazidine MB, Deprenorm OD
Additional information
| Weight | 0.035 kg |
|---|---|
| Shelf life | 3 years. Do not use after the expiration date. |
| Conditions of storage | Store at a temperature not exceeding 25°C. Keep out of reach of children! |
| Manufacturer | Merkle GmbH, Germany |
| Medication form | sustained release tablets |
| Brand | Merkle GmbH |
Related products
Buy Trimetazidine MB-Teva, 35 mg 60 pcs with delivery to USA, UK, Europe and over 120 other countries.