No products in the cart.
Trihydrosol for oral administration 9,45g, 20 pcs.
€1.00
Out of stock
(E-mail when Stock is available)
Description
Oral rehydration and prevention of dehydration in mild to moderate dehydration due to acute diarrhea, in order to replenish water and electrolyte losses.
Active ingredient
Active ingredient
Dextrose, Potassium chloride, Sodium chloride, Sodium citrate
Composition
Composition
Composition per bag:
Potassium chloride – 1.25 g,
Sodium chloride – 1.75 g,
dextrose – 5.00 g, <
Sodium citrate dihydrate – 1.45 g [in terms of sodium citrate – 1.27 g].
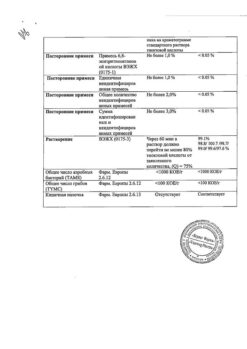
The solution obtained by dissolving Trihydrosol and® , contains the following concentrations of active ingredients:
| Potassium chloride | . 33.5 mmol/L |
| Sodium chloride | 59.8 mmol/L |
| Sodium citrate dihydrate | 9.8 mmol/l |
| Dextrose | 55.5 mmol/l |
| Na + | 89.5 mmol/l |
| K + | 34 mmol/l |
| C l – | 94 mmol/l |
| Citrate | 9.8 mmol/l |
How to take, the dosage
How to take, the dosage
Ingestion. Before use, dissolve the contents of the bag in 0.5 liters of freshly boiled water at room temperature. The prepared solution should be kept in a cool place in a refrigerator (at +2 – +8°C) and used within 24 hours. No other ingredients should be added to the solution so as not to impair the action of the drug.
Before starting treatment, the patient should be weighed to assess weight loss and the degree of dehydration.
It is recommended to avoid eating foods rich in fats and simple carbohydrates during treatment with the drug. In order to prevent dehydration, the use of Trihydrosol ® should be started as soon as diarrhea has started. Usually the drug should not be taken more than 3-4 days, the treatment is stopped with the end of diarrhea.
If a patient has nausea or vomiting, it is recommended that the solution be given chilled, in small portions.
Rehydration. For rehydration, Trihydrosol ® is taken during the first 6 to 10 hours in an amount that is twice the weight loss caused by the diarrhea. For example, if the weight loss is 400 g, the volume of Trihydrosolide ® solution is 800 ml. During this phase the use of other fluids is not required.
The follow-up therapy. If diarrhea persists, after correction of dehydration, it is advisable to administer Trihydrosol ® or water for 24 hours according to the following scheme:
| Body weight (kg) | Total amount of fluid required (L) | Trihydrosol ® (ml) | Water (ml) | Other fluids (ml) |
| 40 – 49 | 2.10 | 900 | 540 | 660 |
| 50 – 59 | 2.30 | 1000 | 600 | 700 |
| 60 – 69 | 2.50 | 1100 | 660 | 740 |
| 70 – 79 | 2.70 | 1200 | 720 | 780 |
| 80 – 89 | 3.20 | 1400 | 800 | 1,000 |
| 90 – 99 | 3.60 | 1500 | 900 | 1200 |
| 100 or more | 4.00 | 1700 | 1000 | 1300 |
Special Instructions
Special Instructions
In children, other solutions with lower sodium concentration and osmolarity should be used. Severe dehydration (weight loss > 10%, anuria) should be stopped using intravenous rehydration products, after which Trigidrosol® can be used.
Recommended doses of the drug should not be exceeded if the patient’s need for additional electrolyte administration is not confirmed by laboratory tests. Food may be given immediately after rehydration. In case of vomiting it is necessary to wait 10 minutes after the end of the vomiting attacks and give the solution to drink slowly in small sips.
Patients with dehydration developed against the background of renal insufficiency, diabetes or other chronic diseases accompanied by acid-base, electrolyte balance or carbohydrate exchange disorders require careful monitoring during therapy with Trihydrosol®.
The patient should consult a physician if during the use of Trihydrosol® the following situations occur:
Slow speech, rapid fatigue, drowsiness, stupor;
- Body temperature rises above 39°C;
- The discharge of urine stops;
- Liquid bloody stools appear;
- Diarrhea lasts more than 2 days;
- Diarrhea suddenly stops, severe abdominal pain appears;
- If home treatment is unsuccessful or impossible.
Trihydrosol ® does not affect the ability to drive vehicles and engage in other potentially dangerous activities that require increased concentration and rapid psychomotor reactions.
Contraindications
Contraindications
Unconsciousness. Intestinal obstruction.
Side effects
Side effects
The risk of hypernatremia and hyperhydration in patients with normal renal function is low. Vomiting may occur if the drug is taken quickly.
Overdose
Overdose
If a large volume or highly concentrated solution of Trihydrosol® is administered (if the solution is not prepared properly), hypernatremia may occur.
Additional information
| Weight | 0.250 kg |
|---|---|
| Shelf life | 2 years. |
| Conditions of storage | In the dry, protected from light at a temperature not exceeding 25 ° C. Keep out of reach of children. |
| Manufacturer | Marbiopharm, Russia |
| Brand | Marbiopharm |
Related products
Buy Trihydrosol for oral administration 9,45g, 20 pcs. with delivery to USA, UK, Europe and over 120 other countries.