No products in the cart.
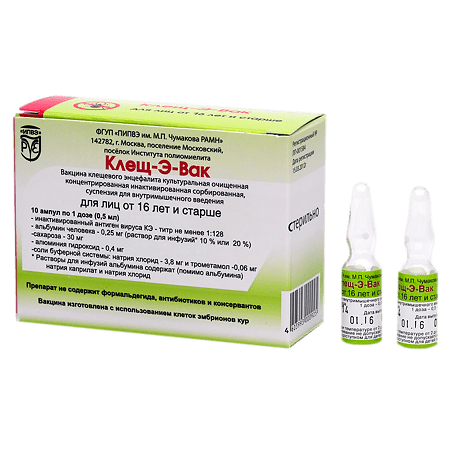
Tick-E-Vac, 0.5 ml/dose 0.5 ml 10 pcs
€1.00
Out of stock
(E-mail when Stock is available)
Description
Specific prophylaxis of tick-borne encephalitis for persons aged 16 years and older in a dose of 0.5 ml and for children from 1 to 16 years in a dose of 0.25 ml;
The immunization of donors in order to obtain specific immunoglobulin.
Contingents subject to specific prophylaxis:
1. Population living in areas enzootic for tick-borne encephalitis.
2. Persons arriving in these areas who perform the following work:
– agricultural, hydromelioration, construction, excavation and relocation of soil, procurement, fishing, geological, surveying, expeditionary, deratization and disinfestation.
– on logging, clearing and improvement of forests, recreation and recreation areas.
3. Persons visiting areas endemic for tick-borne encephalitis for recreation, tourism, work in summer cottages and garden plots.
4 Persons working with live cultures of tick-borne encephalitis pathogen.
Active ingredient
Active ingredient
Vaccine to prevent tick-borne encephalitis
Composition
Composition
One vaccine dose for persons 16 years of age and older (0.5 ml) contains:
How to take, the dosage
How to take, the dosage
The drug is injected intramuscularly into the deltoid muscle of the shoulder.
1. Preventive vaccination.
1.1. Scheduled vaccination.
Initial course of vaccination consists of two intramuscular injections of 1 dose at intervals of 1-7 months. One vaccine dose is: for persons of 16 years old and over – 0.5 ml; for children from 1 to 16 years old – 0.25 ml.
Vaccinations can be given throughout the year, including the epizootic season. No earlier than two weeks after the second vaccination is allowed to visit a CE center during the epidemic season.
The optimal interval between the first and second vaccinations is 5-7 months (autumn – spring).
1.2. Emergency vaccination.
Emergency vaccination can be carried out on epidemic indications. In this case the vaccine shall be administered twice with two-week intervals in 0.5 ml dose to persons aged 16 years and older; 0.25 ml dose to children aged 1-16 years.
Visiting a CE center during epidemic season is allowed not earlier than 2 weeks after the second vaccination.
The first revaccination for both regimens is done once in 1 year after the initial course of vaccination with 0.5 ml dose for persons 16 years and older and with 0.25 ml dose for children from 1 to 16 years old.
Subsequent distant revaccinations are carried out every three years, once in the age dosage.
The general vaccination schedule is presented in the table:
The general vaccination schedule is presented in the table:
Type of vaccination | Primary vaccination | First revaccination | Subsequent revaccinations | ||
First | Second | ||||
| | day 0 vaccination /p> | 1-7 months after the first vaccination | Every 3 years | ||
Emergency | 2 weeks after the first vaccination | ||||
Dose for persons 16 years and older | 0.5 ml | 0.5 ml | 0.5 ml | 0.5 ml | |
Dose for children from 1 year to 16 years | 0.25 ml | 0.25 ml | 0.25 ml | 0.25 ml | |
2. Vaccination of donors.
The course of vaccination is two intramuscular injections of 0.5 ml with an interval of 5-7 months or three injections of 0.5 ml with an interval of 3-5 weeks between vaccinations. The first scheme provides the best immunizing effect.
Revaccination is a single dose of 0.5 ml in 6-12 months. First blood sampling from donors should be carried out 14-30 days after vaccination.
After vaccination in some cases local and general reactions may develop.
Interaction
Interaction
Vaccination against tick-borne encephalitis is allowed simultaneously (on the same day) with other vaccinations with inactivated vaccines of the National calendar of preventive vaccinations and calendar of preventive vaccinations for epidemic indications (except for anti-rabies).
Special Instructions
Special Instructions
Vaccinations are carried out with strict adherence to asepsis and antisepsis rules. The room must be equipped with anti-shock and anti-allergic therapy.
Before opening the ampoule a visual inspection must be carried out. The drug in ampoules with broken integrity, labeling, foreign inclusions, large unbreakable conglomerates, expired shelf life, violation of temperature conditions of storage or transportation shall not be suitable.
Immediately prior to injection the vaccine in an ampoule shall be shaken until a homogeneous suspension is obtained. The drug is injected immediately after opening the ampoule intramuscularly into the deltoid muscle of the shoulder.
The vaccinations administered shall be recorded in the established record forms indicating the name of the drug, date of vaccination, dose, series number, manufacturer, reaction to vaccination.
The drug cannot be administered intravenously!
Inoculation of children and adults with chronic diseases in the acute stage is carried out not earlier than 1 month after recovery (remission).
The vaccine is not used in children under 1 year of age.
Impact on the ability to drive vehicles and machines:
The marked general reactions to administration of the vaccine (significant fever, severe headache) are contraindications to driving vehicles and machinery.
Contraindications
Contraindications
Acute infectious and non-infectious diseases, chronic diseases in the acute stage – vaccination shall be carried out not earlier than 1 month after recovery (remission);
2. Severe allergic reactions in the history; bronchial asthma; autoimmune diseases.
3. history of allergic reactions to components of the vaccine.
4. severe reaction (fever over 40 ° C; injection site – swelling, hyperemia over 8 cm in diameter) or complications of the previous vaccine dose.
5. Children under 1 year.
When vaccinating donors, consider the contraindications listed above, as well as contraindications relating to the selection of donors.
In each case of disease not contained in this list of contraindications, the vaccination is carried out on the approval of the doctor, based on the health of the vaccinated and the risk of tick-borne encephalitis. In order to identify contraindications the doctor (paramedic) conducts an interview and examination of the vaccinated person on the day of vaccination with mandatory thermometry.
Side effects
Side effects
After administration of the vaccine in some cases local and general reactions may develop.
In assessing the adverse reactions of the drug were based on the following data in terms of frequency:
very common >10%;
common, 1 to 10%;
occasional, 0.1 to 1%;
rare, 0.01 to 0.1%;
very rare, <0.01%, including isolated cases.
Overdose
Overdose
No cases of overdose have been identified.
Additional information
| Weight | 0.032 kg |
|---|---|
| Shelf life | 2 years.Preparation with expired shelf life cannot be used. |
| Conditions of storage | The drug is stored and transported at a temperature of 2 to 8 ° C. |
| Manufacturer | FSUE Chumakov Institute, Russia |
| Medication form | suspension |
| Brand | FSUE Chumakov Institute |
Related products
Buy Tick-E-Vac, 0.5 ml/dose 0.5 ml 10 pcs with delivery to USA, UK, Europe and over 120 other countries.