No products in the cart.
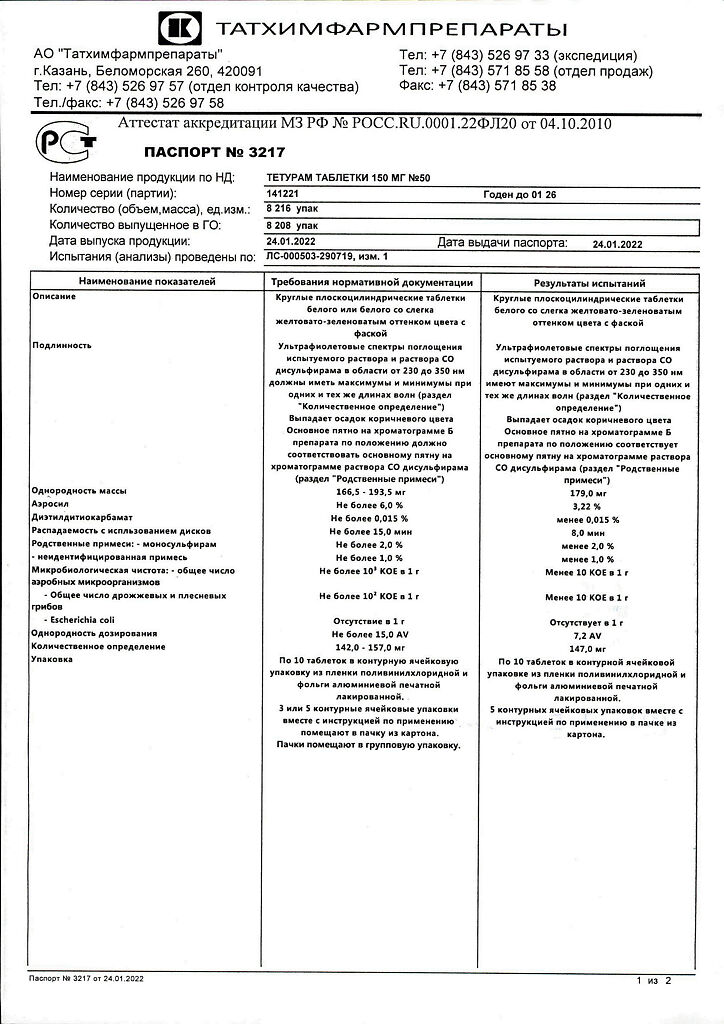
Teturam, tablets 150 mg 50 pcs
€9.73 €8.11
EAN: 4604060991511
SKU: 86060
Categories: Medicine, Neurology and Psychiatry, Sedatives and hypnotics
Description
The action of this drug is based on the blockade of acetaldehyde dehydrogenase, which is involved in the metabolism of ethyl alcohol.
This leads to a significant increase in the concentration of the metabolite of ethyl alcohol – acetaldehyde, which causes negative feelings (blood rush to the face, nausea, vomiting, tachycardia, lowering of blood pressure (BP), etc.) that make drinking alcohol after taking Teturam extremely unpleasant.
This leads to a conditioned reflex aversion to the taste and smell of alcoholic beverages.
Indications
Indications
Prevention and treatment of relapses in chronic alcoholism.
As a detoxifying agent in chronic nickel poisoning.
Active ingredient
Active ingredient
Composition
Composition
1 tablet contains teturam 150 mg
How to take, the dosage
How to take, the dosage
The treatment is prescribed after careful examination of the patient and warning about the consequences and complications.
The drug is taken orally at 150-500 mg 2 times a day according to an individual regimen.
In 7-10 days the tetramalcohol test is carried out (20-30 ml of 40% vodka after taking 500 mg of the preparation), in case of poor reaction the dose of alcohol is increased by 10-20 ml (maximum dose of vodka is 100-120 ml).
The test is repeated in 1-2 days in the hospital and in 3-5 days on an outpatient basis, with correction of the doses of alcohol and/or the drug as necessary.
Afterward, a maintenance dose of 150-200 mg per day may be used for 1-3 years.
Interaction
Interaction
Contraindicated combinations
Alcohol: intolerance reaction (hot flashes, erythema, vomiting, tachycardia). Avoid taking alcoholic beverages and medicines containing alcohol.
Indesirable combinations
Isoniazid: behavioral and coordination disorders.
Nitro-5-imidazoles (metronidazole, ordinazole, secnidazole, tinidazole): delirium disorders, confusion.
Phenytoin.: significant and rapid rise in plasma levels of phenytoin with toxic symptoms (suppression of its metabolism).
If the combination cannot be avoided, clinical observation and monitoring of plasma concentrations of the drug in entrance and after treatment with teturam should be performed.
Combinations requiring caution
Warfarin (and other oral anticoagulants): increased effect of oral anticoagulants and risk of bleeding (reduced breakdown of warfarin in the liver). More frequent monitoring of warfarin concentrations and adjustment of anticoagulant dose within 8 days of Tetum withdrawal is recommended.
Theophylline: Teturam inhibits theophylline metabolism. As a result, the dose of theophylline should be adjusted (reduced dosage), depending on clinical symptoms and plasma concentrations of the drug.
Benzodiazepines: teturam may potentiate the sedative effect of benzodiazepines by inhibiting their oxidative metabolism (especially chlordiazepoxide and diazepam). Benzodiazepine dosage should be adjusted according to clinical manifestations.
Tricyclic antidepressants: possible exacerbation of alcohol intolerance reactions (especially if patients take alcoholic beverages while taking teturam).
Special Instructions
Special Instructions
Patients should be warned about the risk of alcohol intolerance reactions.
Teturam should be taken with caution in patients with renal insufficiency or hypothyroidism, especially at the risk of possible combination with alcohol.
Contraindications
Contraindications
Hypersensitivity, thyrotoxicosis, cardiovascular diseases in decompensation stage (including severely pronounced cardiosclerosis, atherosclerosis of cerebral vessels, pre- and postinfarction states, aortic aneurysm, coronary insufficiency, arterial hypertension stage II-III, CHF), pulmonary tuberculosis with hemoptysis, bronchial asthma, COPD, marked pulmonary emphysema, erosive lesions of the mucous membrane of the gastrointestinal tract, peptic ulcer and 12 duodenal ulcer (acute stage), gastrointestinal bleeding, kidney disease, hepatic insufficiency, diabetes, epilepsy, neuropsychiatric diseases, CNS infectious diseases, polyneuropathy, neuritis of the auditory and ocular nerves, glaucoma, malignant tumors, pregnancy, lactation.
With caution.
Age older than 60 years, peptic ulcer disease (in remission stage), endarteritis, residual effects of stroke, psychosis on taking disulfiram in past history.
Side effects
Side effects
“Metallic” taste in the mouth, unpleasant odor (due to carbon disulfide) in patients with colostomy, hepatitis, polyneuropathy of the lower extremities, neuropsychiatric disorders, memory loss, disorientation in time and space, asthenia, headache, skin allergic reactions.
Overdose
Overdose
Symptoms: the teturam-ethanol combination may cause depression of consciousness up to coma, cardiovascular collapse, neurological complications.
Treatment: symptomatic.
Similarities
Similarities
Additional information
| Weight | 0.037 kg |
|---|---|
| Shelf life | 4 years |
| Conditions of storage | In a light-protected place at a temperature not exceeding 25 °C. |
| Manufacturer | Tatkhimpharmpreparaty, Russia |
| Medication form | pills |
| Brand | Tatkhimpharmpreparaty |
Other forms…
Related products
Buy Teturam, tablets 150 mg 50 pcs with delivery to USA, UK, Europe and over 120 other countries.