No products in the cart.
Tetraderm, cream 15 g
€20.68 €17.23
Description
Pharmacological action – anti-inflammatory, antibacterial, wound healing, antifungal, glucocorticoid.
Indications
Indications
Eczema, Skin fungus, Neurodermatitis, Dermatitis, Dermatosis, Skin infections, Skin itching Treatment of dermatoses of inflammatory genesis with concomitant bacterial and mycotic infection or high likelihood of secondary infection (simple and allergic dermatitis, atopic dermatitis (incl. diffuse atopic dermatitis, atopic dermatitis, atopic dermatitis).diffuse neurodermitis), limited neurodermitis, eczema, dermatomycoses (dermatophytosis, candidiasis, lichen planus), especially when localized in the groin area and large skin folds; simple chronic lichen (limited neurodermitis).
Active ingredient
Active ingredient
Gentamicin, Dexpanthenol, Mometasone, Econazole
Composition
Composition
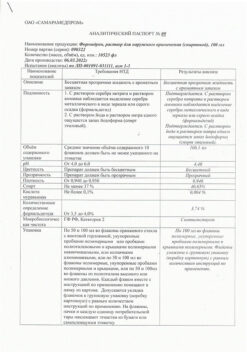
The cream for external use is white or almost white color, homogeneous.
1 g of mometasone furoate 0.5 mg,
gentamicin sulfate 1 mg,
econazole nitrate 10 mg,
dexpanthenol 50 mg.
Associates:
Interaction
Interaction
Studies of interaction of Tetraderm with other drugs have not been conducted, but it is not recommended to use it simultaneously with other drugs for external use.
Directions for use
Directions for use
Externally. The cream is applied to the affected skin areas in a thin layer, gently rubbing, 2 times a day until a positive clinical result. The duration of treatment is individual; it depends on the size and localization of the lesion and severity of the disease and is usually 1-2 weeks.
In foot dermatomycosis the average duration of treatment is 2-4 weeks. It is not recommended to use Tetraderm® for more than 4 weeks. If after the treatment no improvement occurs or new symptoms appear it is necessary to consult a physician.
Use the drug only according to the way of application and in such doses as specified in the instructions.
If necessary, please consult a doctor before using the drug.
Special Instructions
Special Instructions
Tetraderm is not intended for use in ophthalmology. Do not allow the drug to get in the eyes and on the periorbital area. Tetraderm cream is not recommended for use on the face and scalp. It is not recommended to use the drug under occlusive dressings, except in cases where it is necessary. It should not be used to treat varicose trophic ulcers of the lower leg and open wounds.
Some body regions (inguinal folds, armpits and perianal area) are more prone to the risk of occurrence of striae, therefore the duration of application of the drug to these areas of the body should be limited.
If irritation or signs of hypersensitivity are observed during the use of Tetraderm, the treatment should be stopped and the patient should undergo another therapy. Any side effects similar to those observed with systemic GCS including inhibition of adrenal cortex function may also be observed with external application of GCS. Systemic absorption of topical GCS may increase with prolonged use, treatment of large body surfaces or use of occlusive dressings.
In these cases side effects typical for systemic GCS may develop. It should be taken into consideration that GCS may change the manifestation of some skin diseases which may complicate the diagnosis. In addition, the use of GCS may cause delay in wound healing.
In case of prolonged GCS therapy abrupt discontinuation of the therapy may lead to the development of ricochet syndrome, which is manifested as dermatitis with intense reddening of the skin and burning sensation. Therefore, after a long course of treatment Tetraderm should be withdrawn gradually.
Systemic absorption of gentamicin when used externally may increase if applied to large areas of skin, especially during long-term treatment or in the presence of skin lesions.
During prolonged use of Tetraderm on large surfaces of skin with compromised integrity there is a potential possibility of absorption of gentamicin and accordingly the development of ototoxicity symptoms and other adverse events characteristic of gentamicin in its systemic use. Cross-allergic reactions to antibiotics from the group of aminoglycosides may develop.
With prolonged external use of gentamicin the growth of insensitive microflora, including fungi, may be observed. In this case, as well as in case of development of irritation, hypersensitivity reactions and superinfection, the treatment should be stopped and appropriate therapy should be prescribed.
Influence on the ability to drive vehicles and mechanisms.
Contraindications
Contraindications
Hypersensitivity to dexpanthenol, econazole, gentamicin, mometazone or any of the components that make up the drug; skin postvaccination reactions; skin tuberculosis; cutaneous syphilis; varicella; herpes simplex; use in open wounds; pink acne; perioral dermatitis; age under 18 years (effectiveness and safety is not established).
With caution: pregnancy and breastfeeding; use on large areas of skin; long-term use; use in violation of the integrity of the skin; use of occlusive dressings.
Side effects
Side effects
Classification of side effect frequency according to WHO recommendations: very common – ≥1/10; common – from ≥1/100 to < 1/10; infrequent – from ≥1/1000 to < 1/100; rare – from ≥1/10000 to < 1/1000; very rare – < 1/10000, including some reports; frequency is unknown – according to available data it is impossible to determine the frequency of occurrence.
Skin side: rare – skin irritation, dry skin, skin burning sensation, itching, acne, skin atrophy (thinning and loss of elasticity), hypertrichosis (excessive hair growth), hypopigmentation (brightening), perioral dermatitis (rash or irritation around the mouth), allergic contact dermatitis, skin maceration (softening of the upper layers of skin), stretch marks (stretch marks), sweating (skin irritation due to increased sweating), papule and/or pustule formation (rash); frequency is unknown – skin hyperemia (redness), urticaria (blisters).
Nervous system disorders: frequency is unknown – paresthesia (tingling sensation and numbness).
Overdose
Overdose
Symptoms: With prolonged use of topical GCS at high doses, suppression of adrenal function with the development of secondary adrenal insufficiency and symptoms of hypercorticism, including Icenko-Cushing’s syndrome may occur.
A single overdose of gentamicin is not accompanied by the appearance of any symptoms. Prolonged use or use in doses exceeding the recommended ones may lead to significant growth of insensitive microflora, including fungal, in the lesion area.
Pregnancy use
Pregnancy use
Controlled studies of Tetraderm in pregnancy have not been conducted.
Use in pregnancy is possible only in cases where the anticipated benefits to the mother outweigh the potential risk to the fetus.
Additional information
| Weight | 0.030 kg |
|---|---|
| Shelf life | 2 years |
| Conditions of storage | At a temperature not exceeding 25 °C. Keep out of reach of children. |
| Manufacturer | Vertex, Russia |
| Medication form | exterior cream |
| Brand | Vertex |
Related products
Buy Tetraderm, cream 15 g with delivery to USA, UK, Europe and over 120 other countries.