No products in the cart.
Strepsils Intensive, honey-lemon tablets 24 pcs.
€11.18 €9.31
Description
Flurbiprofen is a derivative of propionic acid from the group of non-steroidal anti-inflammatory drugs (NSAIDs) and has significant analgesic, anti-inflammatory and antipyretic effect due to inhibition of cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2), with some selectivity for COX-1, resulting in reduced production of prostaglandins – mediators of pain, inflammation and hyperthermic response.
The drug has a local anesthetic and anti-inflammatory effect on the mucous membrane of the mouth and throat: it reduces swelling, difficulty in swallowing, pain and irritation in the throat.
The tablet dissolves completely in the mouth within 5-12 minutes. The soothing effect starts from the 2nd minute.
The significant reduction in the intensity of sore throat begins at 22 minutes with maximum effect after 70 minutes and lasts up to 4 hours.
Pharmacokinetics
The tablet dissolves completely in the mouth within 5-12 minutes. Absorption is high, flurbiprofen is quickly and almost completely absorbed, distributed throughout the body and largely bound to plasma proteins. Flurbiprofen is detected in blood after 5 min, maximum concentration of flurbiprofen in blood plasma (Cmax) is reached after 40-45 minutes after resorption.
Flurbiprofen can be absorbed in the oral cavity by passive diffusion. Absorption rate depends on the dosage form; maximum concentration of flurbiprofen is reached faster by resorption than by oral administration of an equivalent dose of flurbiprofen.
The elimination half-life (T1/2) is 3-6 hours. It is metabolized in liver by hydroxylation and excreted by kidneys and, to a lesser extent, with bile. Flurbiprofen is excreted with breast milk in small amounts (< 0.05 µg/ml). About 20-25 % of the oral dose of flurbiprofen is excreted unchanged.
Indications
Indications
Treatment of infectious and inflammatory diseases of the oral cavity and pharynx (sore throat, sore throat).
The drug can be used by people with diabetes (does not contain sugar).
Pharmacological effect
Pharmacological effect
Flurbiprofen is a derivative of propionic acid from the group of non-steroidal anti-inflammatory drugs (NSAIDs) and has a significant analgesic, anti-inflammatory and antipyretic effect due to the suppression of cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2), with some selectivity towards COX-1, resulting in a decrease in the production of prostaglandins – mediators of pain, inflammation and hyperthermic reaction.
The drug has a local analgesic and anti-inflammatory effect on the mucous membrane of the mouth and throat: it reduces swelling, difficulty swallowing, pain and irritation in the throat.
The tablet completely dissolves in the mouth within 5-12 minutes. The calming effect begins from the 2nd minute.
A significant reduction in the intensity of sore throat begins at 22 minutes with the maximum effect achieved after 70 minutes and lasts up to 4 hours.
Pharmacokinetics
The tablet completely dissolves in the mouth within 5-12 minutes. Absorption is high, flurbiprofen is quickly and almost completely absorbed, distributed throughout the body and is largely bound to plasma proteins. Flurbiprofen is detected in the blood within 5 minutes, the maximum concentration of flurbiprofen in the blood plasma (Cmax) is achieved 40–45 minutes after resorption.
Flurbiprofen can be absorbed from the oral cavity by passive diffusion. The rate of absorption depends on the dosage form; when absorbed, the maximum concentration of flurbiprofen is achieved faster than when an equivalent dose of flurbiprofen is taken orally.
The half-life (T1/2) is 3-6 hours. It is metabolized in the liver by hydroxylation and excreted by the kidneys and, to a lesser extent, with bile. Flurbiprofen is excreted in breast milk in small quantities (<0.05 mcg/ml). Approximately 20–25% of an oral dose of flurbiprofen is excreted unchanged.
Special instructions
Special instructions
It is recommended to take the drug for the shortest possible course and in the minimum effective dose necessary to eliminate symptoms.
Patients with diabetes should take into account that each honey-lemon lozenge contains about 2.5 g of sugar (0.21 XE) (orange lozenges do not contain sugar).
Orange lozenges contain liquid maltitol and isomalt (the maximum daily dose of which exceeds 2 g), and therefore may have a laxative effect.
When symptoms of gastropathy appear, careful monitoring is indicated, including esophagogastroduodenoscopy, a complete blood count (hemoglobin determination), and a stool test for occult blood.
If it is necessary to determine 17-ketosteroids, the drug should be discontinued 48 hours before the study.
During the treatment period, ethanol intake is not recommended.
Patients with renal or hepatic insufficiency, as well as elderly patients and patients taking diuretics, should consult a doctor before using the drug, as there is a risk of deterioration in renal function. With short-term use of the drug, the risk is negligible.
Patients with arterial hypertension, including a history of and/or chronic heart failure, should consult a doctor before using the drug, since the drug may cause fluid retention, increased blood pressure and edema.
Information for women planning pregnancy: the drug inhibits cyclooxygenase and prostaglandin synthesis and may affect ovulation, disrupting female reproductive function (reversible after discontinuation of treatment).
If irritation occurs in the oral cavity, skin rash, damage to the mucous membrane or other manifestations of an allergic reaction, you should stop using the drug and consult a doctor.
If existing symptoms worsen or new ones appear, including signs of a bacterial infection, you should immediately consult a doctor to review therapy.
Active ingredient
Active ingredient
Flurbiprofen
Composition
Composition
1 lozenge contains:
Active ingredients:
flurbiprofen 8.75 mg;
Excipients:
macrogol 300 5.47 mg,
potassium hydroxide 2.19 mg,
lemon flavor [502904 A) 3.6 mg,
levomenthol 2 mg, honey 50.4 mg,
liquid sucrose 1407 mg,
dextrose 1069 mg
Contraindications
Contraindications
Hypersensitivity to flurbiprofen or any of the components included in the drug.
A history of hypersensitivity reactions (bronchial asthma, bronchospasm, rhinitis, Quincke’s edema, urticaria, recurrent polyposis of the nose or paranasal sinuses) in response to the use of acetylsalicylic acid or other NSAIDs.
Erosive and ulcerative diseases of the gastrointestinal tract (including gastric and duodenal ulcers, Crohn’s disease, ulcerative colitis) or ulcerative bleeding in the acute phase or in history (two or more confirmed episodes of peptic ulcer or ulcerative bleeding).
A history of bleeding or perforation of a gastrointestinal ulcer caused by the use of NSAIDs.
Glucose-6-phosphate dehydrogenase deficiency, hemophilia and other bleeding disorders (including hypocoagulation), hemorrhagic diathesis.
Severe liver failure or active liver disease.
Severe renal failure (creatinine clearance <30 ml/min), confirmed hyperkalemia.
Decompensated heart failure; period after coronary artery bypass surgery.
Sucrase/isomaltase deficiency, fructose intolerance, glucose-galactose malabsorption.
Pregnancy (III trimester).
Children's age up to 12 years.
With caution
If you have the conditions listed in this section, you should consult your doctor before using the drug.
Concomitant use of other NSAIDs; a history of a single episode of gastric ulcer or gastrointestinal ulcer bleeding; history of gastrointestinal tract diseases (ulcerative colitis, Crohn’s disease), gastritis, enteritis, colitis, Helicobacter pylori infection; bronchial asthma or allergic diseases in the acute stage or in history – bronchospasm may develop; systemic lupus erythematosus or mixed connective tissue disease (Sharpe’s syndrome) – increased risk of aseptic meningitis (with short-term use of flurbiprofen the risk is negligible); renal failure, including dehydration (creatinine clearance less than 30-60 ml/min), nephrotic syndrome; liver failure, liver cirrhosis with portal hypertension, hyperbilirubinemia; arterial hypertension and/or heart failure, edema; simultaneous use of medications that may increase the risk of ulcers or bleeding, in particular oral glucocorticosteroids (including prednisolone), anticoagulants (including warfarin), antiplatelet agents (including acetylsalicylic acid, clopidogrel), selective serotonin reuptake inhibitors (including citalopram, fluoxetine, paroxetine, sertraline); pregnancy I-II trimester, breastfeeding period; old age; alcohol consumption.
Side Effects
Side Effects
The risk of side effects can be minimized if the drug is taken in a short course at the minimum effective dose required to eliminate symptoms.
The following adverse reactions were observed with short-term use of the drug. When treating chronic conditions and with long-term use, other adverse reactions may occur.
The incidence of adverse reactions was assessed based on the following criteria: very often (≥ 1/10), often (from ≥ 1/100 to < 1/10), infrequently (from ≥ 1/1000 to < 1/100), rarely (from ≥ 1/10,000 to < 1/1000), very rarely (< 1/10,000), frequency unknown (data on no frequency estimates available).
Blood and lymphatic system disorders
Frequency unknown: hematopoietic disorders (anemia, thrombocytopenia).
Nervous system disorders
Common: dizziness, headache, paresthesia.
Uncommon: drowsiness.
Immune system disorders
Rarely: anaphylactic reactions.
Heart disorders
Frequency unknown: heart failure, edema.
Vascular disorders
Frequency unknown: increased blood pressure.
Respiratory, thoracic and mediastinal disorders
Common: feeling of irritation in the throat.
Uncommon: exacerbation of asthma and bronchospasm, shortness of breath, wheezing, blisters in the mouth and throat, pharyngeal hypoesthesia (decreased sensitivity in the mouth and throat).
Gastrointestinal disorders
Common: diarrhea, oral ulceration, nausea, oral pain, oral paresthesia, oral and pharyngeal pain, oral discomfort (warmth, burning or tingling sensation in the mouth).
Uncommon: bloating, abdominal pain, constipation, dry mouth, dyspepsia, flatulence, glossalgia (burning mouth syndrome), dysgeusia (change in taste perception), oral dysesthesia, vomiting.
Skin and subcutaneous tissue disorders
Uncommon: skin rash, itching.
Not known: severe skin reactions such as bullous reactions including Stevens-Johnson syndrome and toxic epidermal necrolysis (Lyell’s syndrome).
Disorders of the liver and biliary tract
Frequency unknown: hepatitis.
Mental disorders
Uncommon: insomnia.
Others
Uncommon: fever, pain.
Interaction
Interaction
The simultaneous use of flurbiprofen with the following drugs should be avoided:
Acetylsalicylic acid: with the exception of low doses of acetylsalicylic acid (no more than 75 mg per day) prescribed by a doctor, since combined use may increase the risk of side effects.
Other NSAIDs, including ibuprofen and selective cyclooxygenase-2 inhibitors: The simultaneous use of two or more NSAIDs should be avoided due to a possible increased risk of side effects.
Use with caution simultaneously with the following medications:
Anticoagulants: NSAIDs may enhance the effect of anticoagulants, in particular warfarin.
Antiplatelet agents and selective serotonin reuptake inhibitors: increased risk of gastrointestinal bleeding.
Antihypertensives (ACE inhibitors and angiotensin II antagonists) and diuretics: NSAIDs may reduce the effectiveness of these drugs and may increase nephrotoxicity due to cyclooxygenase inhibition, especially in patients with impaired renal function (it is necessary to ensure adequate fluid replacement in such patients).
Alcohol: may increase the risk of adverse reactions, especially bleeding in the gastrointestinal tract.
Cardiac glycosides: simultaneous use of NSAIDs and cardiac glycosides can lead to worsening heart failure, a decrease in glomerular filtration rate and an increase in the concentration of cardiac glycosides in the blood plasma.
Cyclosporine: Increased risk of nephrotoxicity with concomitant use of NSAIDs and cyclosporine.
Glucocorticosteroids: increased risk of gastrointestinal ulceration and gastrointestinal bleeding.
Lithium preparations: there is evidence of the likelihood of an increase in the concentration of lithium in the blood plasma during the use of NSAIDs.
Methotrexate: there is evidence of the likelihood of an increase in the concentration of methotrexate in the blood plasma during the use of NSAIDs. It is necessary to take NSAIDs 24 hours before or after taking methotrexate.
Mifepristone: NSAIDs should be started no earlier than 8-12 days after stopping mifepristone, as NSAIDs may reduce the effectiveness of mifepristone.
Quinolone antibiotics: In patients receiving concomitant treatment with NSAIDs and quinolone antibiotics, the risk of seizures may be increased.
Tacrolimus: Concomitant use of NSAIDs and tacrolimus may increase the risk of nephrotoxicity.
Zidovudine: Concomitant use of NSAIDs and zidovudine may result in increased hematotoxicity.
Oral hypoglycemic drugs: possible changes in blood glucose levels (it is recommended to increase the frequency of monitoring blood glucose levels).
Phenytoin: an increase in the serum level of phenytoin is possible (monitoring the serum level of phenytoin and, if necessary, dose adjustment is recommended).
Potassium-sparing diuretics: Concomitant use of potassium-sparing diuretics and flurbiprofen may lead to hyperkalemia.
Probenecid and sulfinpyrazone: Medicines containing probenecid or sulfinpyrazone may delay the elimination of flurbiprofen.
Tolbutamide and antacids: To date, studies have shown no interactions between flurbiprofen and tolbutamide or antacids.
Overdose
Overdose
Symptoms: nausea, vomiting, epigastric pain or, less commonly, diarrhea, tinnitus, headache and gastrointestinal bleeding. In more severe cases, manifestations from the central nervous system are observed: drowsiness, rarely – agitation, convulsions, disorientation, coma. In cases of severe poisoning, metabolic acidosis and increased prothrombin time, acute renal failure, liver tissue damage, decreased blood pressure, respiratory depression and cyanosis may develop. In patients with bronchial asthma, exacerbation of this disease is possible.
Treatment: symptomatic, with mandatory maintenance of airway patency, monitoring of ECG and vital signs until the patient’s condition is normalized. Oral use of activated charcoal or gastric lavage is recommended within one hour after taking a potentially toxic dose of flurbiprofen. Frequent or prolonged seizures should be treated with intravenous diazepam or lorazepam. When bronchial asthma worsens, the use of bronchodilators is recommended. There is no specific antidote to flurbiprofen.
Storage conditions
Storage conditions
Store in a dry place at a temperature not exceeding 25° C.
Shelf life
Shelf life
3 years.
Manufacturer
Manufacturer
Reckitt Benckiser Healthcare International Ltd, UK
Additional information
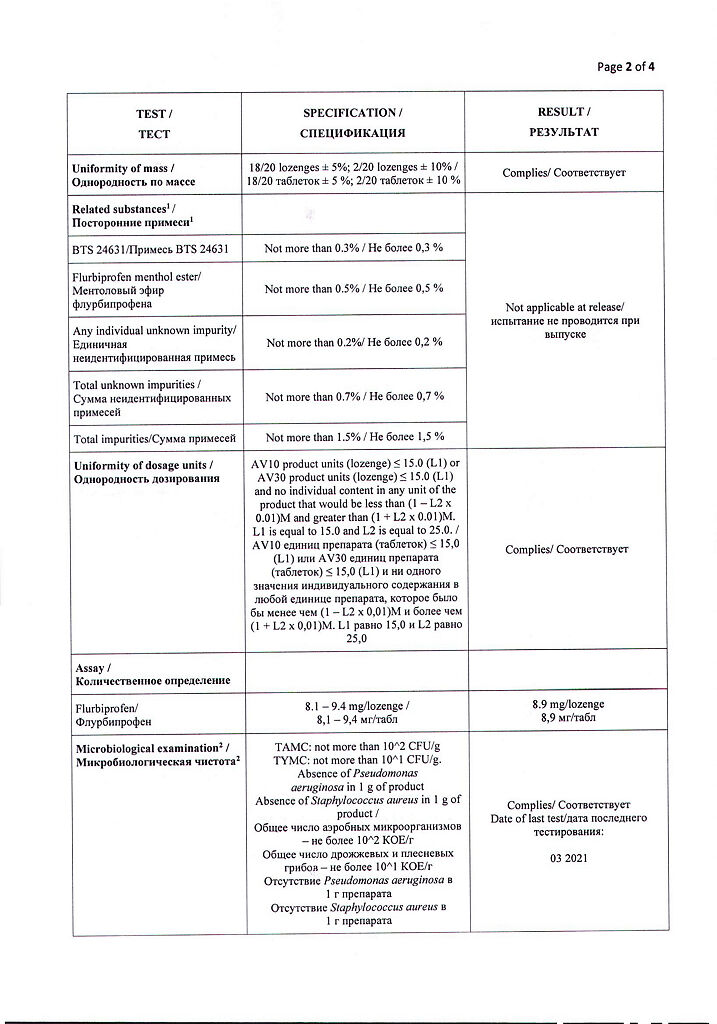
| Shelf life | 3 years. |
|---|---|
| Conditions of storage | Store in a dry place at a temperature not exceeding 25 ° C. |
| Manufacturer | Reckitt Benckiser Healthcare International Ltd, United Kingdom |
| Medication form | lozenges |
| Brand | Reckitt Benckiser Healthcare International Ltd |
Other forms…
Related products
Buy Strepsils Intensive, honey-lemon tablets 24 pcs. with delivery to USA, UK, Europe and over 120 other countries.