No products in the cart.
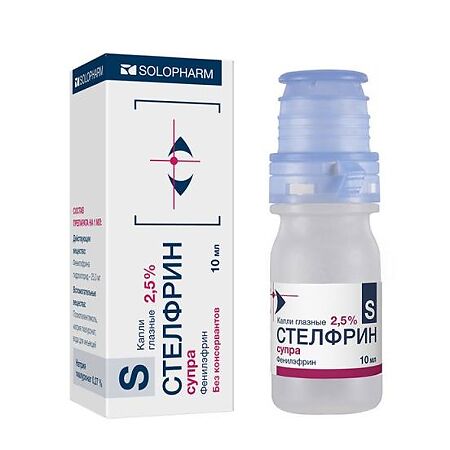
Stelfrin, eye drops 2.5% 10 ml
€20.18 €17.49
Out of stock
(E-mail when Stock is available)
Description
Pharmacotherapeutic group: Alpha-adrenomimetic
ATC code: S01FB01
Pharmacodynamics:
Phenylephrine is a sympathomimetic. It has pronounced alpha-adrenergic activity.
When used topically in ophthalmology, it causes dilation of the pupil improves the outflow of intraocular fluid and narrows the conjunctival vessels.
Phenylephrine has a strong stimulatory effect on the postsynaptic alpha-adrenoreceptors and has very little effect on the beta-adrenoreceptors of the heart. The drug has a vasoconstrictor effect similar to that of norepinephrine (noradrenaline), while it has almost no chronotropic and ionotropic effect on the heart. The vasopressor effect of phenylephrine is weaker than that of norepinephrine, but it is longer lasting. Causes vasoconstriction 30-90 s after instillation duration is 2-6 h.
After instillation, phenylephrine contracts the dilator of the pupil and the smooth muscles of the conjunctival arterioles, thus causing pupil dilation. Midriasis occurs within 10-60 min after a single instillation. Midriasis persists after instillation of 25% solution for 2 hours. Midriasis caused by phenylephrine is not accompanied by cycloplegia.
Phenylephrine easily penetrates into ocular tissues and reaches maximum concentration in plasma 10-20 min after instillation into eye. Phenylephrine is excreted by the kidneys unchanged (< 20%) or as inactive metabolites.
Indications
Indications
Iridocyclitis (to prevent posterior synechiae and reduce exudation from the iris).
For diagnostic pupil dilation during ophthalmoscopy and other diagnostic procedures necessary to monitor the condition of the posterior segment of the eye.
The provocation test in patients with a narrow anterior chamber angle profile and suspected closed angle glaucoma.
The differential diagnosis of superficial and deep eyeball injection.
The “red eye” syndrome (to reduce hyperemia and irritation of the mucous membrane of the eye).
Accommodation spasm.
Active ingredient
Active ingredient
Phenylephrine
Composition
Composition
1 ml of the drug contains:
The active ingredient:
Phenylephrine hydrochloride 25.0 mg
Excipients:
Polyethylene glycol 300 19.0 mg
(macrogol 300)
Sodium hyaluronate 2.7 mg
Injection water to 1.0 ml.
How to take, the dosage
How to take, the dosage
In iridocyclitis the drug is used to prevent development and rupture of already formed posterior synechiae; to reduce exudation into the anterior chamber of the eye. For this purpose, 1 drop of the drug is put into the conjunctival sac of the affected eye (eye) 2-3 times a day. If necessary to maintain mydriasis for a long time another instillation of the drug after 1 hour is possible. A single instillation of the drug is used for diagnostic procedures: – as a provocative test in patients with a narrow anterior chamber angle profile and suspected closed-angle glaucoma. If the difference between intraocular pressure values before and after pupil dilation is between 3 and 5 mmHg. For differential diagnosis of the type of ocular injection: if a narrowing of the globe vessels is noted 5 minutes after instillation, the injection is categorized as superficial. If redness remains, the patient must be closely monitored for iridocyclitis or scleritis, as this indicates dilatation of the deeper vessels. To relieve Spasm of Accommodation in children from 6 years of age and adults, the drug is instilled 1 drop in each eye at night daily for 4 weeks. Performance of the tube dropper: 1. Separate one tube dropper. 2. Open the dropper tube (making sure the solution is at the bottom of the dropper tube, rotate and remove the valve). 3. Drip the desired amount into the eye. The dose contained in the tube is sufficient for one instillation in both eyes. The drip tube should be discarded after one use, even if there is still some left. To use the bottle with the dropper dispenser: 1.Remove the safety ring before using the bottle for the first time. 2. Carefully remove the cap from the bottle. Without touching the pipette, place the bottle upside down with the pipette tip between your thumb and forefinger. Preferably before use, pump the pipette until the first drop of product appears by pressing the top of the bottle several times with the index finger. 3. With your head tilted back, position the bottle dispenser over your eye and with the index finger of one hand pull the lower eyelid down. Lightly press down on the bottle and drip the desired amount of solution into the conjunctival sac of the eye. The open vial tip should not come into contact with the surface of the eye or hands. 4. Place the cap back on the bottle after use. The product has a shelf life once the vial has been opened. At the end of the shelf life, the product should be discarded. If the vial is visibly damaged, the product should not be used.
Interaction
Interaction
The mydriatic effect of phenylephrine is enhanced when it is used in combination with topical administration of atropine. Due to the increased vasopressor effect, tachycardia may develop.
Phenylephrine should be used with monoamine oxidase inhibitors and within 21 days after stopping their use with caution as this has the potential for uncontrolled rise in arterial pressure. The vasopressor effect of α-adrenomimetics may also be potentiated by concomitant use with the tricyclic antidepressants propranolol reserpine guanethidine methyldopa and m-cholinoblockers.
Phenylephrine may potentiate cardiovascular depression during inhalation anesthesia.
Concomitant use with other adrenomimetics and sympathomimetics may increase the cardiovascular effects of phenylephrine.
The use of phenylephrine may cause impairment of concomitant hypotensive therapy and lead to increased blood pressure tachycardia.
Special Instructions
Special Instructions
Exceeding the recommended dose of 25% solution in patients with trauma of the eye or its appendages in the postoperative period or with reduced tear production may lead to increased absorption of phenylephrine and development of systemic side effects. The drug does not contain a preservative, thus avoiding undesirable effects of the preservative on the external tissues of the eye conjunctiva and cornea and eliminating the risk of allergic reactions to the preservative. Influence on the ability to drive motor vehicle and machine:
After the use of the drug due to the changes in accommodation and pupil width visual acuity may decrease, so until its recovery it is not recommended to drive vehicles and engage in potentially dangerous activities that require high concentration and quick psychomotor reactions.
Contraindications
Contraindications
Hypersensitivity to the components of the drug.
Narrow-angle or closed-angle glaucoma.
Elderly age.
Severe cardiovascular or cerebrovascular disease.
Arterial hypertension combined with coronary heart disease aortic aneurysm atrioventricular block of MP degree arrhythmia.
Tachycardia.
Tear production disorder.
Prematurity; childhood under 6 years of age (with accommodation spasm).
Hyperthyroidism.
Hepatic porphyria.
Congenital deficiency of glucose-6-phosphate dehydrogenase.
Rinitis.
Cautions:
Diabetes mellitus (risk of increased blood pressure associated with autonomic dysregulation).
Concomitant use with monoamine oxidase inhibitors (including for 21 days after stopping their use).
Sickle cell anemia wearing contact lenses after surgical interventions (decreased healing due to conjunctival hypoxia).
Side effects
Side effects
Eye disorders
Conjunctivitis periorbital edema.
In some cases, patients report a burning sensation at the beginning of use blurred vision irritation feeling of discomfort in the eye increased lacrimation increased intraocular pressure.
Phenylephrine may cause reactive miosis the day after use. Repeated instillations of the drug at this time may result in less severe mydriasis than previously observed. This effect is more common in older patients.
Phenylephrine-induced significant contraction of the pupil dilation muscle within 30-45 minutes after instillation may cause detection of pigment particles from the pigmented iris in the humor of the anterior chamber. Suspension in the chamber humor must be differentiated from manifestations of anterior uveitis or from blood cells in the anterior chamber humor.
Systemic reactions
Skin and its appendages: contact dermatitis.
Cardiovascular system disorders: palpitations tachycardia arrhythmia increased arterial pressure ventricular arrhythmia reflex bradycardia coronary artery occlusion pulmonary embolism.
Overdose
Overdose
Symptoms: restlessness nervousness dizziness sweating vomiting palpitations weak or shallow breathing.
Treatment: if the systemic effects of phenylephrine occur, unwanted effects can be stopped by using α-adrenoblockers such as 5-10 mg of phentolamine intravenously. The injection may be repeated if necessary.
Pregnancy use
Pregnancy use
The action of phenylephrine in pregnant women has not been adequately studied; therefore, the drug may be used in this category of patients only if the expected benefits to the mother exceed the potential risk to the fetus or child. It is unknown whether the drug is excreted with breast milk.
In animals in late pregnancy, phenylephrine caused fetal growth retardation and stimulated early labor.
If prescribed during lactation, breastfeeding should be discontinued for the duration of treatment.
Similarities
Similarities
Mesaton, Relief, Irifrin, Irifrin BK, Nasol Baby, Phenylephrine Optic, Bebifrin, Hemorrhoid Remedies
Additional information
| Weight | 0.021 kg |
|---|---|
| Shelf life | 2 years. Do not use after the expiration date! |
| Conditions of storage | At a temperature not exceeding 25 ° C. Store out of the reach of children. |
| Manufacturer | Grotex Ltd, Russia |
| Medication form | eye drops |
| Brand | Grotex Ltd |
Related products
Buy Stelfrin, eye drops 2.5% 10 ml with delivery to USA, UK, Europe and over 120 other countries.