No products in the cart.
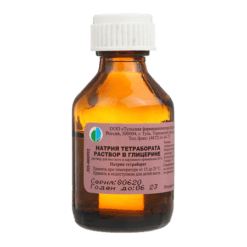
Sodium tetraborate, 20% solution 30 g
€1.00
Out of stock
(E-mail when Stock is available)
Description
Pharmacotherapeutic group: antiseptic.
TATX code: D08AD.
Pharmacological properties
Pharmacodynamics
Antiseptic agent. It has bacteriostatic activity. It is effective for candidiasis. Removes fungus mycelia from mucous membranes, disrupts the process of attachment of the fungus to the mucous membranes and inhibits its reproduction (it is not an antifungal drug, as it does not have fungicidal or fungistatic action). As an antimicrobial drug, it is part of combined preparations for the treatment of inflammatory diseases of the upper respiratory tract. It has insecticidal properties (class IV toxicity).
Pharmacokinetics
It is absorbed in the gastrointestinal tract and through the damaged skin. It is deposited in the bone tissue and the liver. It is excreted unchanged by the kidneys and through the intestine within 1 week.
Indications
Indications
Mold, Fungus, Diaper rash, Bedsores
Candida lesions of the mucous membranes of the mouth, pharynx, upper respiratory tract, urinary tract and genitals; diaper rash, bedsores.
Active ingredient
Active ingredient
Sodium tetraborate
Composition
Composition
Active substance: sodium tetraborate decahydrate – 20 g; excipient: glycerol (glycerin) – 80 g.
How to take, the dosage
How to take, the dosage
Location and externally.
Age range of patients: adults, children from 3 years old.
Use for rinses, dermal lubrication, irrigation 2-3 times a day. It is used as needed until the symptoms are eliminated.
There are no special effects of the drug when it is first taken or when it is discontinued.
Particulars of medical use of the drug in adults with chronic diseases are absent.
Special Instructions
Special Instructions
The condition for efficacy in monotherapy of vaginal candidiasis is the need for treatment procedures by medical personnel, multiple treatments; otherwise, the fungus cells may persist in the vaginal crypts and lead to recurrence.
Impact on ability to drive vehicles, machinery:
No information.
Contraindications
Contraindications
Hypersensitivity to the components of the drug, pregnancy, lactation, children under 3 years of age. Violation of the integrity of the skin (when treating the skin).
.
Side effects
Side effects
Hyperemia and burning sensation at the site of application.
Overdose
Overdose
Overdose when applied topically. Symptoms of overdose by local administration of the drug are unlikely. There are no data on cases of overdose by local administration.
Overdose by accidental ingestion of the drug. Fatal dose for adults is 10 – 20 g, toxic concentration in blood is 40 mg/l, lethal concentration – 50 mg/l. The following symptoms are possible: abdominal pain, vomiting, decreased appetite, diarrhea, dehydration, weakness, confusion, dermatitis, menstrual disorders, anemia, twitching of muscles of face and limbs, alopecia, heart, liver and kidney function disorders. Treatment of overdose when taking the drug orally includes: gastric lavage, forced diuresis, in severe poisoning – hemodialysis, in/m – riboflavin mononucleotide 10 mg/day, correction of water-electrolyte balance and acidosis: intravenous infusion of sodium hydrocarbonate solution, plasma exchange solutions, sodium chloride solution and dextrose. In case of abdominal pain: p/k 1 ml 0.1% atropine solution, 1 ml 0.2% platifylline solution, 1 ml 1% promedol solution, intravenous dextrose procaine mixture (50 ml 2% procaine solution and 500 ml 5% dextrose solution). Maintenance of cardiovascular system functions.
Pregnancy use
Pregnancy use
The use of the drug is contraindicated for pregnant women and during lactation.
Similarities
Similarities
Sodium tetraborate (borax), Sodium tetraborate, Sodium tetraborate solution in glycerol
Additional information
| Weight | 0.072 kg |
|---|---|
| Shelf life | 3 years. Do not use after the expiration date on the label. |
| Conditions of storage | In the dark place at the temperature not more than 30 ° C. Keep out of reach of children. |
| Manufacturer | Samaramedprom, Russia |
| Medication form | solution for topical and external use |
| Brand | Samaramedprom |
Other forms…
Related products
Buy Sodium tetraborate, 20% solution 30 g with delivery to USA, UK, Europe and over 120 other countries.