No products in the cart.
Skin-cup, cream 0.2% 50 g
€100.97 €84.15
Description
Pharmgroup:
A drug with antiproliferative, antibacterial and antifungal action for external use.
Pharmic action: < br>
Zinc pyrithione activated has anti-inflammatory, antibacterial and antifungal activity. Antifungal activity is especially pronounced against Pityrosporum ovale and Pityrosporum orbiculare, which cause inflammation and excessive peeling in skin diseases. Antibacterial activity is shown against a number of pathogenic microorganisms Streptococcus spp., Staphylococcus spp., Escherichia coli, Pseudomonas aeruginosa, Proteus spp.
Zinc pyrithione reduces intracellular ATP level, promotes depolarization of cell membranes, causing death of fungi and bacteria. The mechanism of anti-inflammatory action is not studied.
Pharmacokinetics: < br>
When applied externally, zinc pyrithione is deposited in the epidermis and superficial layers of the dermis. Systemic absorption is slow. It is detected in the blood in trace amounts.
Indications
Indications
Psoriasis, atopic dermatitis, eczema, neurodermatitis, dry skin, seborrheic dermatitis in adults and children over 1 year.
Pharmacological effect
Pharmacological effect
Pharmacotherapeutic group
Dermatoprotective agent.
ATX code [D 11 AX 12]
Pharmacological properties
Pharmacodynamics: Activated zinc pyrithione has anti-inflammatory, antibacterial and antifungal activity. Antifungal activity is especially pronounced against Pityrosporum ovale and Pityrosporum orbiculare, which support inflammation and excessive flaking in dandruff, seborrhea, psoriasis and other skin diseases. Zinc pyrithione reduces the intracellular level of ATP, promotes depolarization of cell membranes and the death of fungi and bacteria.
Pharmacokinetics
When applied externally, activated zinc pyrithione is deposited in the epidermis and superficial layers of the dermis. Systemic absorption occurs slowly. Found in the blood in trace amounts.
Active ingredient
Active ingredient
Pyrithione zinc
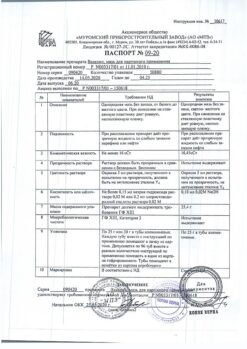
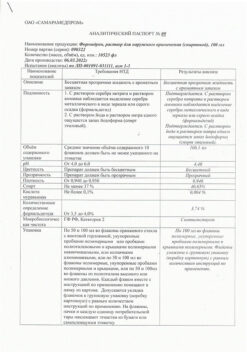
Composition
Composition
100 g of the drug contains:
Active ingredient:
pyrithione zinc activated 200 mg
Excipients:
glyceryl mono- and distearate (Tegin M), capryl caprylate (Tegosoft EE), methyldextrose and macrogol-20 ether (Tegosoft E20), isopropyl palmitate (Tegosoft R), methyldextrose polyglyceryl distearate (Tego Care 450), butylated hydroxytoluene, glycerol, propyl parahydroxybenzoate (Nipagin), sucrose coconut oil fatty acid ester (Tegosoft LSE 65K), stearyl alcohol (TegoAlkanol 18), ethanol, flavoring (citronellol, phenylethanol, geraniol, terpineol, cinnamic alcohol), cyclomethicone (Abil K 4), water.
Contraindications
Contraindications
Hypersensitivity to the components of the drug.
Side Effects
Side Effects
Allergic reactions. In the first days of treatment, a short-term burning sensation is possible at the site of application of the drug, which, as a rule, does not require discontinuation of the drug.
Interaction
Interaction
No clinically significant interactions of Skin-cap with other drugs have been identified.
Recommendations for use
Recommendations for use
Externally. After vigorous shaking, the cream is applied in a thin layer to the affected areas of the skin 2 times a day. The course of treatment for psoriasis is 1-1.5 months, for atopic dermatitis 3-4 weeks.
Method of administration and doses in pediatrics: the method of application and doses in children do not differ from the method of application and doses in adults. If necessary, you can repeat the course of treatment 1-1.5 months after consultation with your doctor.
Storage conditions
Storage conditions
At a temperature of +15 to +25 °C, out of the reach of children.
Shelf life
Shelf life
3 years. Do not use after expiration date.
Manufacturer
Manufacturer
B.Brown Medical S.A., Spain
Additional information
| Shelf life | 3 years. |
|---|---|
| Conditions of storage | At +4 to +20 °C, out of the reach of children. |
| Manufacturer | B. Brown Medical S.A., Spain |
| Medication form | exterior cream |
| Brand | B. Brown Medical S.A. |
Other forms…
Related products
Buy Skin-cup, cream 0.2% 50 g with delivery to USA, UK, Europe and over 120 other countries.