No products in the cart.
Sedalgin Plus, tablets 10 pcs
€9.75 €8.53
Description
Sedalgin Plus is a combined drug with analgesic, antipyretic and anti-inflammatory effect.
Sodium metamizole is a derivative of pyrazolone and has analgesic, antipyretic and mild anti-inflammatory effects, the mechanism of which is associated with inhibition of prostaglandin synthesis by inhibiting cyclooxygenase.
Caffeine has a stimulating effect on the central nervous system (CNS), mainly on the cerebral cortex, respiratory and vasomotor centers.
It increases mental and physical performance, reduces sleepiness and fatigue. Caffeine increases permeability of histohematic barriers and increases bioavailability of non-narcotic analgesics, thus strengthening the therapeutic effect.
Thiamine (vitamin B,) is involved in metabolism and helps to improve nerve-reflex
conduction.
Indications
Indications
Neuritis, Joint pain (arthralgia), Sciatica, Toothache, Headache, Radiculitis, Arthrosis and arthritis, Flu, Myositis, Lumbago, Colds, MigraineA mild to moderate pain syndrome of various genesis: Headache (including migraine), toothache, neuralgia, muscle pain (myalgia), joint pain (arthralgia), radicular syndrome, menstrual pain (algodysmenorrhea). Fever syndrome in “colds” and other infectious-inflammatory diseases (if diagnosed).
Composition
Composition
1 tablet contains:
acting ingredients:
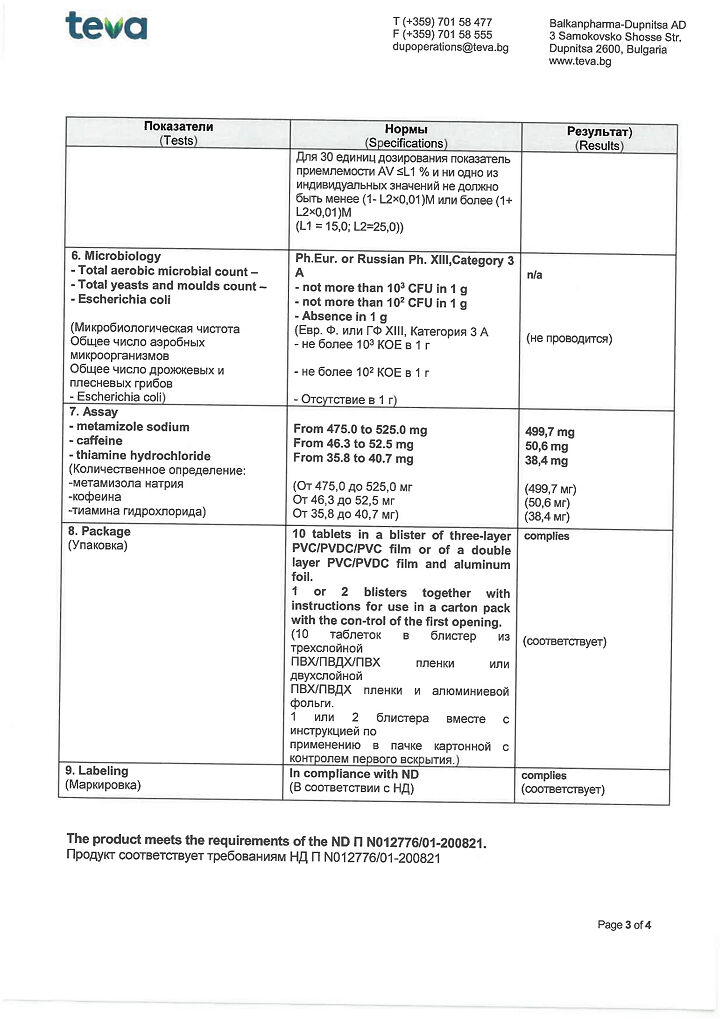
metamizole sodium 500 mg,
caffeine 50 mg,
Thiamine hydrochloride 38.75 mg;
excipients:
microcrystalline cellulose 45.75 mg,
wheat starch 11.00 mg,
gelatin 4.00 mg,
colloidal silica 2.50 mg,
talc 12.00 mg,
magnesium stearate 6.00 mg.
How to take, the dosage
How to take, the dosage
Sedalgin® Plus is taken orally. The tablets should be swallowed without chewing, with plenty of water.
The minimum dose sufficient to control pain and fever should be used.
If pain persists or increases in intensity, a physician should be consulted to determine the cause of the symptoms.
The drug should not be used for a long time or the dose should be increased without a physician’s prescription. If prolonged use, it is necessary to monitor the general blood count (number of blood cells).
The single dose for adults and adolescents older than 15 years is 1 tablet. The maximum single dose is 2 tablets. Unless otherwise prescribed, a single dose may be taken 2-3 times a day. The maximum daily dose is 4 tablets.
When used as an analgesic, the duration of therapy is 1-5 days; when used as an antipyretic, it is 1-3 days.
Elderly patients
The dose should be reduced in elderly patients because excretion of sodium metabolites may be delayed.
Patients with impaired hepatic function
As the excretion rate of the drug is reduced in impaired hepatic function, repeated high doses of the drug should be avoided. No dose reduction is required if the drug is used for a short time.
Patients with impaired renal function
Patients with impaired general condition and decreased creatinine clearance should reduce the dose because excretion of sodium metabolites may be delayed.
Because in patients with impaired renal function the excretion rate of the drug is reduced, repeated administration of high doses of the drug should be avoided. No dose reduction is required if the drug is used for a short time.
Interaction
Interaction
Simultaneous use of Sedalgin® ® Plus with other medicinal products requires caution due to the content of sodium metamizole which may affect the metabolism of the drug when used once and is an enzyme inducer when used for a long time.
Cedalgin® Plus may decrease serum concentrations of cyclosporin and may jeopardize tissue transplantation, so cyclosporine concentrations should be monitored when they are used together.
The concomitant use of Sedalgin® Plus withother non-narcotic analgesics, antipyretics, and anti-inflammatory drugs may result in mutual enhancement of toxic effects and also increases the risk of hypersensitivity reactions.
Tricyclic antidepressants, oral contraceptives and allopurinol impair the metabolism of sodium metamizole in the liver and increase its toxicity.
Barbiturates, phenylbutazone and other inducers of liver microsomal enzymes weaken the effects of sodium metamizole.
Neuroleptics, sedatives and tranquilizers increase the analgesic effect of Sedalgin ® Plus. Severe hypothermia may develop when co-administered withchlorpromazine.
Sodium metamizole displaces oral hypoglycemic agents, indirect anticoagulants, glucocorticosteroids and indomethacin (drugs with high plasma protein binding) by increasing their effects.
In concomitant use with myelotoxic drugs (e.g., gold salt preparations, anticancer drugs, chloramphenicol, etc.) the manifestation of hematotoxicity of sodium metamizole is increased, there is a risk of damage to white blood cells.
The addition of sodium methamisole to treatment with methotrexate may increase the hematotoxic effects of methotrexate, especially in elderly patients. Therefore, coadministration of these drugs should be avoided.
Tiamazole and sarcolysin when co-administered with sodium metamizole increase the risk of leukopenia.
Codeine, blockers H2-histamine receptors and propranolol increase the effects of sodium metamizole.
When treating with sodium metamizole, do not use radiographic contrast agents, colloidal blood substitutes and penicillin (increased risk of anaphylactic/anaphylactoid reactions).
When used together, sodium metamizole may decrease the effect of acetylsalicylic acid on platelet aggregation. Therefore, this combination should be used with caution when treating patients taking low doses of acetylsalicylic acid for cardioprotection (prevention of thrombosis).
Sodium metamizole may decrease the blood concentration of bupropion, which should be taken into account when using metamizole sodium and bupropion concomitantly.
The concomitant use of sympathomimetics may cause central nervous system overstimulation.
Special Instructions
Special Instructions
When treating patients receiving cytostatic drugs, the drug should be taken only under the supervision of a physician.
Anaphylactic/anaphylactoid reactions
The following conditions cause an increased risk of hypersensitivity reactions to sodium metamizole:
- bronchial asthma induced by taking analgesics;
- intolerance of analgesics in the form of urticaria or angioedema;
- chronic urticaria;
- alcohol intolerance (hypersensitivity to alcohol) against which, even when taking small amounts of some alcoholic beverages, patients experience sneezing, lacrimation and marked facial redness. Alcohol intolerance may indicate a previously unidentified bronchial asthma syndrome associated with analgesics (aspirin asthma);
- intolerance or hypersensitivity to dyes (e.g., tartrazine) or preservatives (e.g., benzoates);
- anaphylactic or other immunological reactions to other pyrazolones, pyrazolidines and other non-narcotic analgesics (see
If the patient has a history of anaphylactoid reactions, the drug should only be given after careful consideration of the risks and benefits. If the decision to use the drug in such patients will require close medical monitoring of their condition, and it is necessary to have means for immediate aid in case of developing anaphylactic/anaphylactoid reactions.
In predisposed patients anaphylactic shock may occur, so patients with bronchial asthma or atopy should be given sodium metamizole with caution.
Serious skin reactions
. Life-threatening skin reactions such as Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) have been described with sodium methamisole. If symptoms of SSD or TEN (such as a progressive skin rash, often with blisters or mucous membrane lesions) occur, treatment should be stopped immediately. Treatment with the drug should never be repeated.
Skin reactions should be monitored carefully, especially during the first weeks of treatment.
Agranulocytosis
The agranulocytosis that develops during treatment with metamizole is of immune-allergic origin and lasts at least one week. This reaction is very rare and can be severe, life-threatening and even fatal. It is not dose-dependent and can occur at any time during treatment.
The drug should be discontinued and the following subjective or objective symptoms, possibly associated with neutropenia, should be immediately consulted with the physician: fever, chills, sore throat, mouth ulcers. In case of neutropenia (neutrophil count < 1500 in mm3), the treatment should be stopped immediately, a detailed general blood test should be performed urgently and the blood count should be continued until the number of blood cells returns to normal values.
Pancytopenia
If pancytopenia occurs, treatment should be stopped immediately and a complete blood count should be monitored until normal.
People should seek medical attention immediately if subjective or objective symptoms occur during treatment with the drug that suggest abnormal blood changes (e.g., general malaise, infections, persistent fever, bruising, bleeding, paleness).
Isolated hypotensive reactions
The use of sodium metamizole may cause isolated hypotensive reactions. These reactions probably depend on the dose of the drug and occur more frequently after parenteral administration.
Acute abdominal pain
The use of Sedalgin is inadmissible® Plus to relieve acute abdominal pain (until the cause is known).
Hepatic and renal impairment
In patients with hepatic or renal impairment, Cedalgin® Plus should only be used after consulting a physician because of the reduced excretion rate of the drug in these patients.
Influence on laboratory results
In patients treated with sodium metamizole, changes in the results of laboratory tests performed using Trinder reaction and similar reactions (e.g., analysis of serum creatinine, triglycerides, HDL cholesterol and uric acid concentrations) have been reported.
Caffeine can affect the results of a test using adenosine or dipyridamole, so do not take Sedalgin®® Plus less than 12 hours before the test.
The wheat starch in Cedalgin® Plus contains a small amount of gluten (considered gluten-free) and is not likely to cause problems in patients with celiac disease. One tablet contains no more than 0.01 micrograms of gluten. Patients with wheat allergies (other than celiac disease) should not take this medication.Influence on driving, operating machinery
There is no evidence that Sedalgin® Plus, when used in the recommended doses, has an adverse effect on concentration and reaction time.
But when therapy with the drug at high doses, consideration should be given to the possibility of impaired ability to concentrate and reduced reaction time, which may pose a risk in situations where these abilities are particularly important (e.g., driving or operating a moving machine), especially with concomitant alcohol use.
Contraindications
Contraindications
- High sensitivity to sodium metamizole, caffeine, thiamine and/or excipients as well as other pyrazolones (phenazole, propiphenazole, isopropylaminophenazole) or pyrazolidones (phenylbutazone, oxyphenbutazole), including, for example, a history of agranulocytosis when using one of the drugs.
- Disorders of medullary hematopoiesis (e.g., after treatment with cytostatics) or diseases of the hematopoietic environment.
- An history of bronchospasm or other anaphylactic reactions (e.g., urticaria, rhinitis, angioedema) when using analgesic drugs such as salicylates, paracetamol, diclofenac, ibuprofen, indomethacin, naproxen.)
- Acute intermittent hepatic porphyria (risk of exacerbations of porphyria).
- Congenital deficiency of 6-phosphate dehydrogenase (risk of hemolysis).
- Pregnancy.
- Breastfeeding period.
- Childhood (under 15 years).
With caution
- Artial hypotension (systolic blood pressure below 100 mmHg.), hemodynamic instability (myocardial infarction, multiple trauma, onset of shock), decreased circulating blood volume, onset of heart failure, high fever (increased risk of sudden drop in blood pressure).
- In diseases in which a significant decrease in blood pressure may pose an increased risk (patients with severe coronary heart disease and pronounced stenosis of the cerebral arteries).
- In alcoholism.
- In patients at increased risk of severe anaphylactic/anaphylactoid reactions with:
- bronchial asthma, especially in combination with concomitant polyposis rhinosinusitis;
- chronic urticaria and other types of atopy (allergic diseases in the development of which a significant role belongs to hereditary predisposition to sensitization: polinosis, allergic rhinitis, etc.п.
- intolerance of alcohol (reaction even to small amounts of certain alcoholic beverages, with symptoms such as itching, lacrimation and marked redness of the face);
- intolerance of dyes (eg tartrazine) or preservatives (eg benzoates).
- In severe hepatic and renal dysfunction (low doses are recommended due to the possibility of delayed excretion of sodium metamizole).
.
Side effects
Side effects
Unwanted reactions are categorized according to the World Health Organization (WHO) Classification: very common (≥1/10); common (≥1/100, < 1/10); infrequent (≥1/1000, < 1/100); rare (≥1/10000, < 1/1000); very rare (< 1/10000); frequency unknown (cannot be determined based on available data).
Blood and lymphatic system disorders
Frequency unknown: Transient leukopenia, agranulocytosis, hemolytic anemia, purpura, thrombocytopenia.
Immune system disorders
Frequency unknown: anaphylactic shock or other anaphylactic reactions.
Nervous system disorders
Frequency unknown: insomnia, vertigo, increased excitability.
Cardiac disorders
Hfrequency unknown: Tachycardia, palpitations.
Relatory system, chest and mediastinum disorders Frequency unknown: bronchospasm.
Gastrointestinal disorders
Frequency unknown: loss of appetite, nausea, vomiting, cholestasis, jaundice.
Skin and subcutaneous tissue disorders
Hfrequency unknown: rash, itching.
All side effects should be reported to the treating physician.
Overdose
Overdose
Symptoms
The following symptoms may occur in overdose: Nausea, vomiting, abdominal pain, decreased renal function/acute renal failure with oliguria (e.g., due to the development of interstitial nephritis), more rarely symptoms of the central nervous system (agitation, insomnia, headache, dizziness), tinnitus, melena, hematoma, and cardiac rhythm disturbances (tachycardia). In more severe cases – oliguria to anuria, epileptopharyngeal seizures, coma, seizures, agranulocytosis, aplastic or hemolytic anemia, hemorrhagic diathesis.
The non-toxic metabolite of sodium metamizole (rubazonic acid) may cause red staining of the urine after very high doses of the drug are administered.
Treatment
In recent administration of the drug, primary detoxification measures aimed at limiting further absorption of the drug (use of vomiting agents, gastric lavage, activated charcoal, laxatives) may be taken. There is no specific antidote. The major metabolite of metamizole (4-N-methylaminoantipyrine) is excreted by hemodialysis, hemofiltration, hemoperfusion or plasma filtration. Symptomatic treatment is used if necessary. If a seizure syndrome develops, intravenous diazepam and fast-acting barbiturates are administered.
Pregnancy use
Pregnancy use
Data related to metamizole
Pregnancy
There are no data on the adverse effects of metamizole on the fetus: In rats and rabbits, methamisole had no teratogenic effects, and toxic effects on the fetus were observed only at high doses toxic to the mother. Nevertheless, clinical data on the use of Sedalgin® Plus during pregnancy are insufficient.
Methamisole penetrates the placenta.
The use of Sedalgin® Plus during pregnancy is contraindicated. This is because although methamisole is a weak inhibitor of prostaglandin synthesis, the possibility of premature closure of the arterial (botallic) duct and complications in the perinatal period associated with impaired platelet aggregation of the mother and the newborn cannot be completely excluded.
Breastfeeding period
Methamisole metabolites penetrate into breast milk. Do not breastfeed during treatment with Sedalgin ® Plus and for 48 hours after the last dose of the drug.
Additional information
| Weight | 0.100 kg |
|---|---|
| Shelf life | 2 years. |
| Conditions of storage | In a dry, light-protected place at a temperature not exceeding 25 ° C. Keep out of reach of children! |
| Manufacturer | Balkanpharma – Troyan AD, Bulgaria |
| Medication form | pills |
| Brand | Balkanpharma – Troyan AD |
Related products
Buy Sedalgin Plus, tablets 10 pcs with delivery to USA, UK, Europe and over 120 other countries.