No products in the cart.
Raso, 20 mg 30 pcs.
€13.59 €11.32
EAN: 8901148233911
SKU: 265116
Categories: Medicine, Stomach, intestines, liver, Ulcer and gastritis
Description
Pharmacodynamics
Mechanism of action
Rabeprazole belongs to the class of antisecretory drugs which are chemically substituted benzimidazoles. Rabeprazole inhibits the activity of the enzyme H+/K+ATPase (“proton pump”), thus blocking the final stage of hydrochloric acid synthesis. This effect is dose-dependent and leads to inhibition of both basal and stimulated hydrochloric acid secretion regardless of the irritant.
Rabeprazole has no anticholinergic properties.
Antisecretory activity
After oral administration of 20 mg rabeprazole the antisecretory effect occurs within one hour. Inhibition of basal and stimulated hydrochloric acid secretion 23 h after the first dose of rabeprazole is 62% and 82%, respectively, and continues until 48 h. This duration of pharmacokinetic action is much longer than that predicted by the half-life (T1/2), which is approximately 1 h.
Indications
Indications
Active ingredient
Active ingredient
Composition
Composition
How to take, the dosage
How to take, the dosage
Interaction
Interaction
Concomitant use with digoxin may increase (mild to moderate) plasma concentrations of digoxin.
Concomitant use with ketoconazole decreases its bioavailability.
Special Instructions
Special Instructions
Contraindications
Contraindications
Side effects
Side effects
Pregnancy use
Pregnancy use
Similarities
Similarities
Additional information
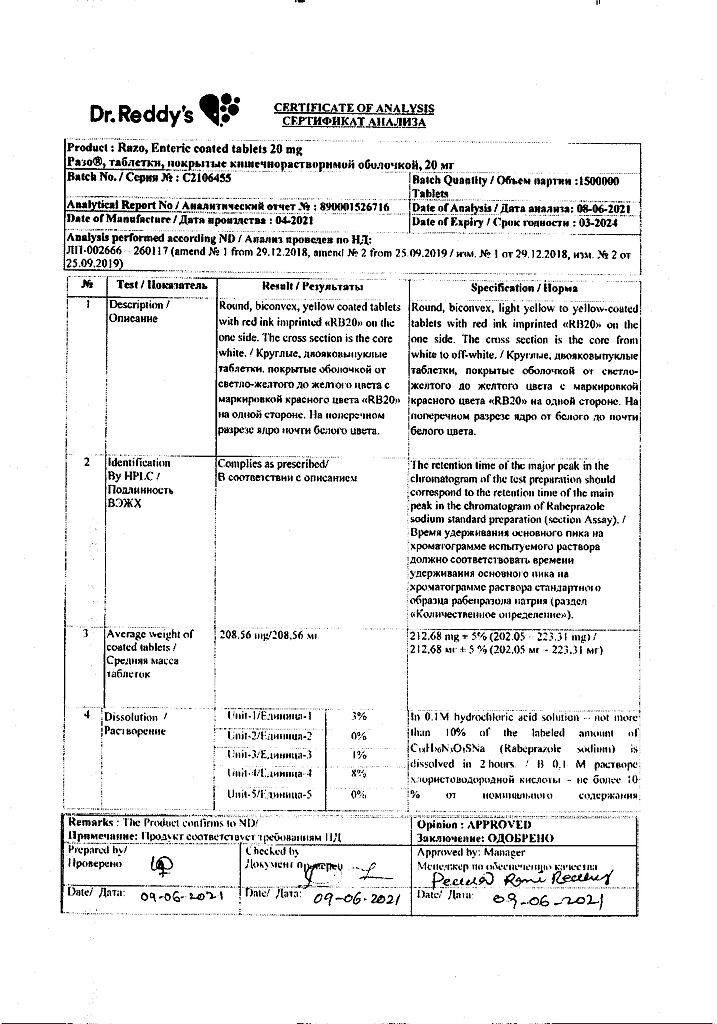
| Shelf life | 2 years (in cans), 3 years (in blisters). Do not use after the expiration date stated on the package. |
|---|---|
| Conditions of storage | At the temperature not more than 25 ° C. Keep out of reach of children! |
| Manufacturer | Dr. Reddy's, India |
| Medication form | enteric-soluble film-coated tablets |
| Brand | Dr. Reddy's |
Related products
Buy Raso, 20 mg 30 pcs. with delivery to USA, UK, Europe and over 120 other countries.