No products in the cart.
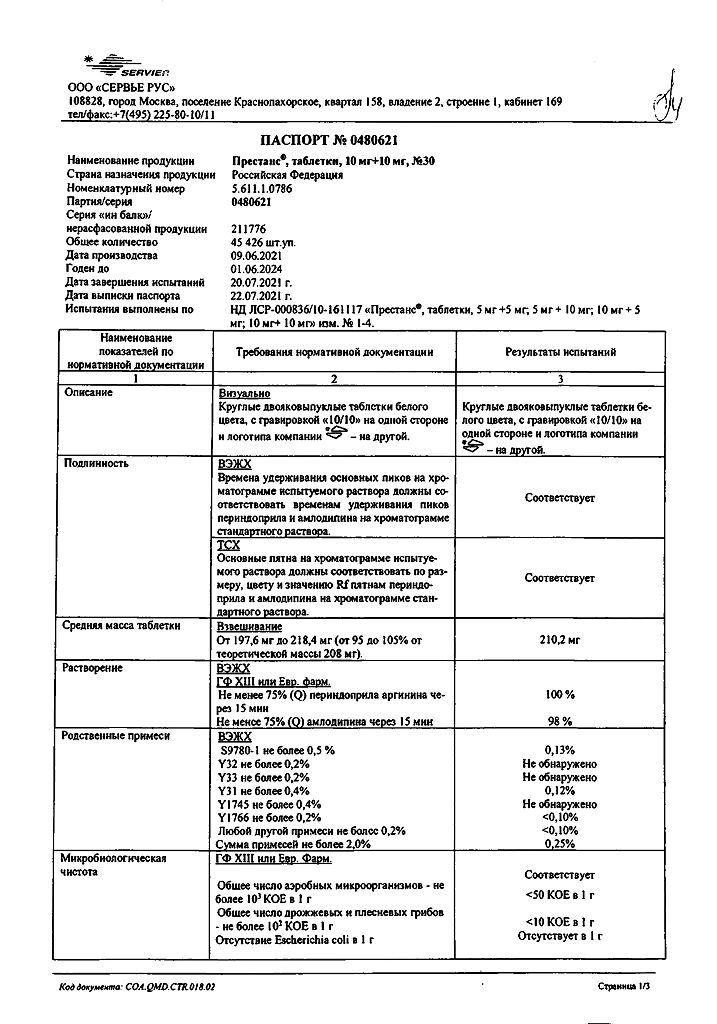
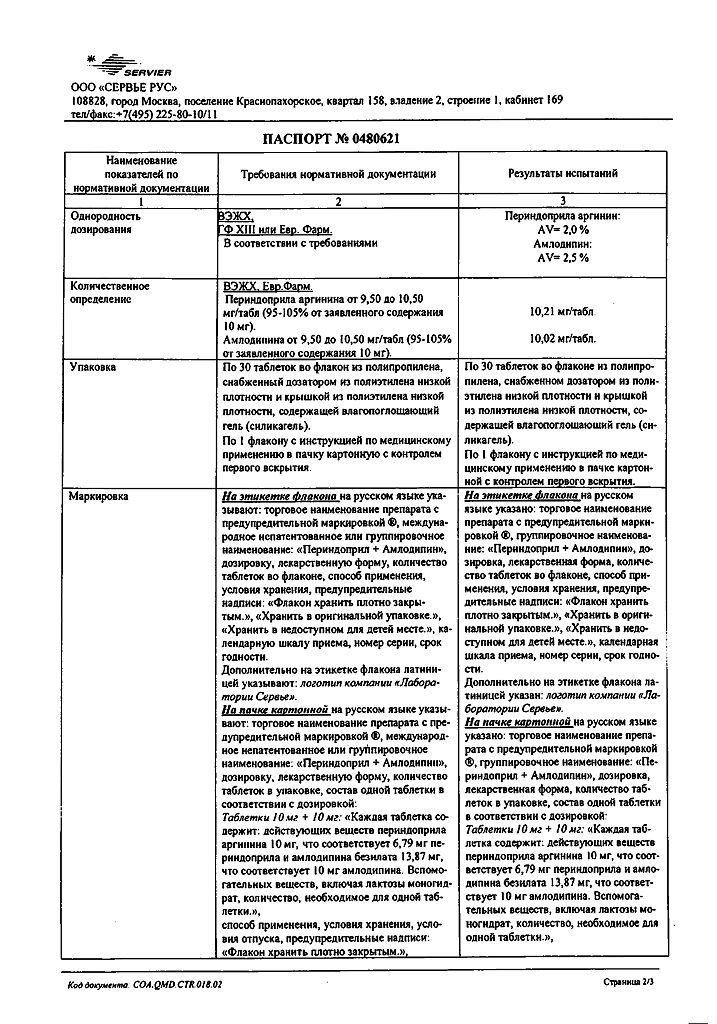
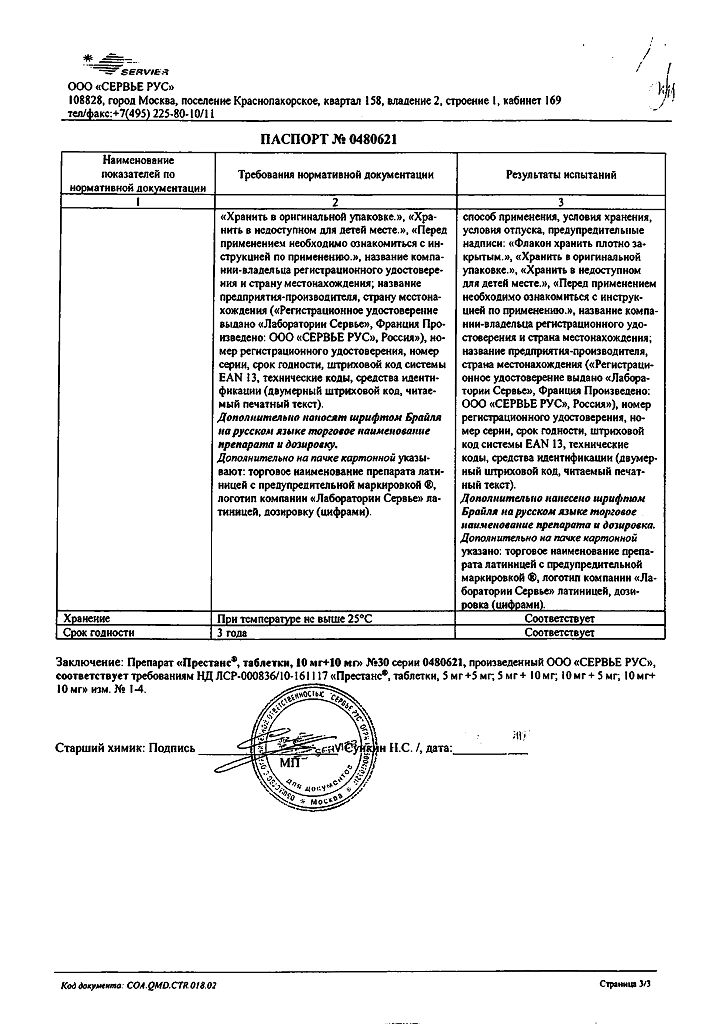
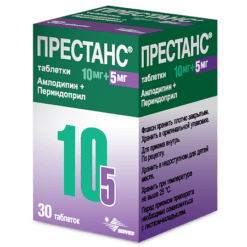
Prestance, 10 mg+10 mg tablets 30 pcs
€24.96 €20.80
Description
Perindopril is an inhibitor of the enzyme that converts angiotensin I to angiotensin II (ACE inhibitor). Amlodipine is a “slow” calcium channel blocker, a dihydropyridine derivative, inhibits transmembrane transition of calcium ions into cardiomyocytes and smooth muscle cells of the vascular wall.
Indications
Indications
Hypertension (high blood pressure), Angina pectoris, Cardialgia (heart pain)In adults: Arterial hypertension and/or coronary heart disease: stable angina pectoris in patients who require therapy with amlodipine and perindopril.
Composition
Composition
Prestance in 5 mg/5 mg, 5 mg/10 mg, 10 mg/5 mg, 10 mg/10 mg tablets containing / amlodipine (AMLO) 5 mg / perindopril arginine (PER) 5 mg, AMLO 5 mg / PER 10 mg, AMLO 10 mg / PER 5 mg, AMLO 10 mg / PER 10 mg. Contains lactose as an excipient.
How to take, the dosage
How to take, the dosage
1 tablet a day in the morning before meals. Prestance is not intended for initial therapy. If necessary, the dose of the drug may be changed or the doses of the individual components may be adjusted.
Elderly patients and patients with impaired renal function. Regularly monitor creatinine and plasma potassium (K+). Contraindicated in creatinine clearance (CK) < 60 ml/min. Impaired liver function. Dose selection should be done individually, amlodipine and perindopril in monotherapy. Children. Do not prescribe.
Interaction
Interaction
Contraindicated: aliskiren in patients with diabetes mellitus and/or impaired renal function. Extracorporeal therapy. Valsartan + sacubitril.
Do not recommend: aliskiren: in patients without diabetes or renal dysfunction; ARA II, estramustine, K+ saving diuretics (triamterene, amiloride), K+ salts, lithium preparations, dantrolene (intravenous), grapefruit or grapefruit juice.
Special attention: .Hypoglycemic agents (insulin, sulfonylurea derivatives), K+unsaving diuretics, K+saving diuretics (eplerenone, spironolactone), racecadotril, mTOR inhibitors (sirolimus, everolimus, temsirolimus), nonsteroidal anti-inflammatory drugs (NSAIDs) including acetylsalicylic acid ³3 g/day, CYP3A4 inducers and inhibitors, baclofen. Cautions: sympathomimetics, gold drugs, tacrolimus, cyclosporine, simvastatin, hypotensive agents, vasodilators, corticosteroids, tetracosactide, α-adrenoblockers (prazosin, alfuzosin, doxazosin, tamsulosin, terazosin), amifostine, tricyclic antidepressants, neuroleptics, general anesthetic agents.
Drugs that cause hyperkalemia: Aliskiren, potassium salts, potassium-saving diuretics, ACE inhibitors, ARA II, NSAIDs, heparins, immunosuppressants (such as cyclosporine or tacrolimus), trimethoprim and co-trimoxazole (sulfamethoxazole + trimethoprim).
Special Instructions
Special Instructions
Elevated sensitivity/angioneurotic edema. Stop taking the drug and monitor the patient until the signs of edema have completely disappeared. Angioneurotic edema with laryngeal edema may be fatal. Co-administration with valsartan + sacubitril, ratsecadotril, mTOR inhibitors (e.g., sirolimus, everolimus, temsirolimus), glyptins (linagliptin, saxagliptin, sitagliptin, wildagliptin) increases risk of angioedema (e.g., tongue, vocal fold or pharyngeal edema with or without airway obstruction).
Combined use with drugs containing valsartan + sacubitril. Contraindicated. Use of one drug no earlier than 36 hours after discontinuation of the other drug.
Anaphylactoid reactions during LDL apheresis. Rarely, life-threatening reactions may develop. Temporarily discontinue therapy before each procedure.
Anaphylactoid reactions during desensitization. Anaphylactoid reactions occurred again if the drug was accidentally administered.
Neutropenia/agranulocytosis/thrombocytopenia/anemia. With extreme caution in the background of systemic connective tissue diseases, immunosuppressants, allopurinol or procainamide. Control of white blood cells in the blood.
Renovascular hypertension. In patients with bilateral renal artery stenosis or artery stenosis of the single kidney, the risk of severe hypotension and renal failure increases. Taking diuretics is an additional risk factor. Deterioration of renal function is possible with little change in creatinine concentration in patients with unilateral renal artery stenosis.
Double RAAS blockade Co-administration of ACE inhibitors with APA II or aliskiren increases the risk of hypotension, hyperkalemia, and renal dysfunction (including acute renal failure). Therefore, dual RAAS blockade is not recommended. The use of ACE inhibitors in combination with APA II in patients with diabetic nephropathy is contraindicated. Pregnancy. Discontinue treatment. If necessary, prescribe other hypotensive therapy.
Primary hyperaldosteronism. Administration of drugs whose action is based on inhibition of the RAAS is not recommended: patients are not susceptible. Arterial hypotension. In patients with increased risk of symptomatic arterial hypotension (with decreased circulating blood volume (DCV), with severe hypertension with high renin activity) and in patients with angina and cerebrovascular disease it is necessary to monitor BP, renal function and K+ content. Transient arterial hypotension is not an obstacle for further drug administration. After restoration of the RBC and BP, the treatment may be continued.
Mitral stenosis/aortic stenosis/hypertrophic cardiomyopathy. Caution.
Heart failure.With caution.
Renal function impairment.When CK is less than 60 ml/min, individual selection of doses of perindopril and amlodipine and monitoring of serum K+ and creatinine. Patients with renal artery stenosis may have increased blood urea and creatinine. In renovascular hypertension – increased risk of severe hypotension and renal failure.
Renal failure.Amlodipine is not excreted by dialysis.
Hepatic insufficiency. Rarely, cholestatic jaundice occurs against ACE inhibitors. With progression, fulminant necrosis of the liver develops, sometimes with a fatal outcome. If jaundice or significant increase in liver enzymes occurs, discontinue the drug. In severe hepatic failure, increase the dose gradually, ensuring monitoring of the condition.
Ethnic differences. In the Negro race, perindopril is less effective and the risk of angioedema is higher.
Dry cough. Surgery/anesthesia. Discontinue treatment the day before surgery.
Hyperkalemia. Regular control of blood K+ in patients over 70 years of age, with renal failure, impaired renal function, diabetes mellitus, dehydration, acute decompensated heart failure, metabolic acidosis, combined use of K+-saving diuretics, K+ salts.
Diabetes mellitus. Control blood glucose during the first month.
Heart failure.With caution.
Hypertensive crisis.Efficacy and safety have not been established.
Elderly patients. Increase dose with caution.
Galactose intolerance, complete lactase deficiency. glucose-galactose malabsorption syndrome. Should not be taken.
Driving vehicles, machinery. Due to dizziness, drowsiness and other adverse reactions, use caution.
Contraindications
Contraindications
- hypersensitivity to the active ingredients, to other ACE inhibitors or other dihydropyridine derivatives, or to any of the excipients listed in the List of Excipients;
- Hereditary/idiopathic angioedema;
- Pregnancy and breastfeeding (see Pregnancy and lactation. Pregnancy and lactation);
- Combined use with aliskiren and medications containing aliskiren in patients with diabetes and/or moderate to severe renal function impairment (glomerular filtration rate (GFR) < 60 ml/min/1.73m2 body surface area (see Sections Interaction of Pharmacology and Pharmacotherapy).
- combined use with angiotensin II receptor antagonists (ARA II) in patients with diabetic nephropathy (see Interactions and Pharmacodynamics).
- combined use with Valsartan + Sacubitril Combination Therapy (see Cautions and Interactions).
- extracorporeal therapy leading to blood contact with negatively charged surfaces (see Interactions); significant bilateral renal artery stenosis or arterial stenosis of the only functioning kidney (see Special Precautions). Severe arterial hypotension (BP <90 mmHg);
- shock (including cardiogenic shock)
- obstruction of the left ventricular exit tract (eg, left ventricular outflow tract obstruction (e.g., marked aortic stenosis);
- hemodynamically unstable heart failure after acute myocardial infarction;
- renal insufficiency (CK less than 60 ml/min).
.
Side effects
Side effects
Very often:pulmonary edema.
Frequently: drowsiness, dizziness, headache, dysgeusia, paresthesia, visual disturbances (including diplopia), tinnitus, vertigo, palpitations, blood “rushes” to the facial skin, arterial hypotension, shortness of breath, cough, abdominal pain, Nausea, vomiting, dyspepsia, change in stool frequency and nature, diarrhea, constipation, skin itching, exanthema, skin rash, swollen joints (swollen ankles), muscle spasms, increased fatigue, asthenia.
Infrequent: Rhinitis, eosinophilia, hypersensitivity, hypoglycemia, hyperkalemia, hyponatremia, insomnia, mood lability, depression, sleep disturbance, tremor, hypoesthesia, syncope, tachycardia, arrhythmia (including bradycardia, ventricular tachycardia and atrial fibrillation), vasculitis, bronchospasm, dry mouth, angioedema of the face, extremities, lips, mucous membranes, tongue, vocal folds and/or larynx, alopecia, purpura, skin discoloration, increased sweating, urticaria, photosensitivity reactions, pemphigoid, arthralgia, myalgia, back pain, urinary disorders, nycturia, pollakiuria, renal failure, Erectile dysfunction, gynecomastia, peripheral edema, chest pain, pain, malaise, fever, weight gain, weight loss, increased blood urea and creatinine concentrations, falls. Rarely:confusion, exacerbation of psoriasis, increased concentration of bilirubin in the blood, increased activity of “liver” enzymes.
Very rare:. Leukopenia/neutropenia, agranulocytosis, pancytopenia, thrombocytopenia, hemolytic anemia in patients with congenital glucose-6-phosphate dehydrogenase deficiency, hyperglycemia, hypertonicity, peripheral neuropathy, stroke, angina, myocardial infarction, eosinophilic pneumonia, gum hyperplasia, pancreatitis, gastritis, hepatitis, jaundice, cytolytic or cholestatic hepatitis, increased activity of “liver” enzymes, angioedema (Quincke’s edema), erythema multiforme, Stevens-Johnson syndrome, exfoliative dermatitis, acute renal failure, decrease of hemoglobin and hematocrit.
Unspecified frequency:extrapyramidal disorders, Raynaud’s syndrome, toxic epidermal necrolysis. Inadequate antidiuretic hormone secretion syndrome can be considered a very rare complication associated with ACE inhibitors.
Pregnancy use
Pregnancy use
PREGNANCY AND LACTATION. Contraindicated.
FERTILITY. In some patients receiving calcium channel blockers reversible biochemical changes in the sperm head have been found.
Additional information
| Weight | 0.029 kg |
|---|---|
| Shelf life | 2 years |
| Manufacturer | Servier Rus LLC, Russia |
| Medication form | pills |
| Brand | Servier Rus LLC |
Other forms…
Related products
Buy Prestance, 10 mg+10 mg tablets 30 pcs with delivery to USA, UK, Europe and over 120 other countries.