No products in the cart.
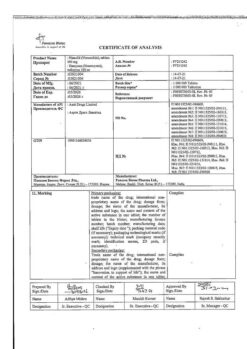
Piroxicam, 10 mg capsules 20 pcs
€1.00
Out of stock
(E-mail when Stock is available)
Description
NSAID, has anti-inflammatory, analgesic, antiaggregant and antipyretic effects. It inhibits COX-1 and COX-2 nonselectively.
Analgesic effect is seen 30 minutes after oral administration, anti-inflammatory effect occurs by the end of 1 week of treatment.
Pharmacokinetics: Well absorbed when taken orally and rectally. Food slows down absorption rate, but does not affect absorption rate. The binding to plasma proteins is 99%.
Plasma concentration is proportional to the dose, TCmax is 3-5 hours. After a single dose of 20 mg Cmax is 1.5-2 mcg/ml, after regular use of 20 mg/day – 3-8 mcg/ml.
It penetrates into synovial fluid (40-50%), into breast milk (1-3%).
Metabolized in the liver by hydroxylation of pyridine ring of side chain with subsequent conjugation with glucuronic acid and formation of inactive metabolites. CYP2C9 isoenzyme is also involved in metabolism. T1/2 is 36-45 hours. Saccharin is one of the metabolites. It is excreted as conjugates by kidneys and, to a lesser extent, by intestines. In unchanged form it is 5%.
Indications
Indications
Inflammatory and degenerative diseases of the musculoskeletal system: Rheumatoid and acute gouty arthritis, ankylosing spondylitis, osteoarthritis.
It is intended for symptomatic therapy, to reduce pain and inflammation at the time of use; does not affect the progression of the disease.
Active ingredient
Active ingredient
How to take, the dosage
How to take, the dosage
Internal, intramural, rectal, external. Inside.
In rheumatoid arthritis, osteoarthritis and Bechterew’s disease – 10-30 mg/day in 1 dose, for a long time. In acute skeletal muscle diseases – 40 mg/day in 1 or more intakes within 2 days, in the next 7-10 days – 20 mg/day. In treatment of acute gout the initial dose is 40 mg, once for the first 2 days, then for 4-6 days. – 40 mg once or by 20 mg twice a day.
In disalgomenorrhea, treatment should be started when the first symptoms appear in an initial dose of 40 mg/day in 1 or more doses over 2 days. During the next 1-3 days it is 20 mg at a single dose.
As an antipyretic drug – 10-20 mg/day; if it is necessary to achieve rapid effect – the first 2 days the dose is prescribed in 40 mg/day with subsequent dose lowering.
In juvenile rheumatoid arthritis it is prescribed once a day: in children with body weight less than 15 kg – 5 mg/day, 16-25 kg – 10 mg/day, 26-45 kg – 15 mg/day, over 46 kg – 20 mg/day.
V/m. For treatment of acute conditions or relieving acute exacerbation of a chronic process – 20-40 mg once a day. After relief of an acute phase of a process it is necessary to switch over to supporting therapy with tablets or capsules.
Rectally, 10-40 mg 1-2 times a day.
Outwardly, the gel or cream is applied to the affected areas (5-10 mm columns) 3-4 times a day; an occlusive dressing is not applied.
Interaction
Interaction
Antiaggregants, anticoagulants increase the risk of bleeding.
Displaces from binding to blood proteins other drugs.
GCS, selective serotonin reuptake inhibitors increase the risk of gastrointestinal mucosal ulceration.
Simultaneous use with other NSAIDs and ASA increases toxicity of piroxicam.
ASA (more than 3.9 g / day) reduces the serum concentration of piroxicam by 20%. In combination with anticoagulants reduces blood clotting (risk of bleeding).
Methotrexate increases the hematotoxicity of piroxicam (deaths are possible).
Cimetidine increases AUC and Cmax of pyroxicam by 13-15%.
Reduces diuretic effect of furosemide.
Reduces renal clearance and increases Css of Li+ drugs (requires monitoring of its concentration).
Special Instructions
Special Instructions
Pyroxicam should be prescribed by physicians experienced in the diagnosis and treatment of inflammatory and degenerative joint diseases.
Concurrent use with misoprostol, proton pump inhibitors, H2-histamine receptor blockers reduces gastrointestinal toxicity.
NSAIDs, including piroxicam, can cause severe gastrointestinal side effects, including bleeding, ulceration and perforation of the stomach, small and large intestine (including fatal). These phenomena can occur at any time of treatment and are not even accompanied by threatening symptoms. The risk of these phenomena is particularly high for piroxicam; it increases in the elderly.
Taking piroxicam significantly increases the risk of severe skin reactions (exfoliative dermatitis, Stevens-Johnson syndrome, toxic epidermal necrolysis) especially in the early treatment period. If the first signs of rash, mucosal ulceration or other signs of hypersensitivity occur, pyroxicam should be immediately discontinued.
Piroxicam, as well as other NSAIDs, increases the risk of thrombosis. NSAIDs increase the risk of thrombotic complications (including myocardial infarction and stroke), patients with risk factors are prescribed these drugs with caution.
Piroxicam, as well as other NSAIDs can contribute to the occurrence of thrombotic complications (including myocardial infarction and stroke). In women the effect of piroxicam on myometrial tone may decrease the contraceptive effect of VIC.
During the treatment it is necessary to avoid ethanol intake.
During the treatment it is necessary to be careful when driving vehicles and engaging in other potentially dangerous activities that require high concentration and quick psychomotor reactions.
Contraindications
Contraindications
Hypersensitivity; gastric and/or 12 duodenal ulcer (including anamnesis), gastrointestinal perforation or bleeding (including anamnesis), gastritis, duodenitis, inflammatory bowel disease (including ulcerative colitis, Crohn’s disease, diverticulitis).ulcerative colitis, Crohn’s disease, diverticulitis), including a history, malignant tumors of the gastrointestinal tract (including a history); concomitant use of other NSAIDs (including a history of other drugs). NSAIDs (including selective COX-2 inhibitors or ASA in analgesic doses), anticoagulants;
severe allergic reactions in past history, especially skin forms (including erythema multiforme syndrome). erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis); full or partial combination of bronchial asthma, recurrent polyposis of the nose and sinuses and intolerance to ASA or others. NSAIDs (including in anamnesis);
severe CPP (CKD less than 30 ml/min), advanced renal disease, severe hepatic insufficiency or active liver disease, conditions after ACH, severe CHF; hyperkalemia, coagulation disorders (including Hemophilia, prolongation of bleeding time, susceptibility to bleeding, hemorrhagic diathesis;
childhood (under 14 years), age over 65 years, pregnancy, lactation;
for rectal administration (optionally) – proctitis, anorectal bleeding. CHD of mild or moderate degree, cerebrovascular diseases, dyslipidemia/hyperlipidemia, diabetes mellitus, peripheral arterial disease, smoking, CKD (IQ 30-60 ml/min), Helicobacter Ñylori infection, alcoholism, simultaneous use of oral GCS (including prednisolone).
The simultaneous use of oral GCS (including prednisolone), antiplatelet agents (including ASA, clopidogrel), selective serotonin reuptake inhibitors (including citalopram, fluoxetine, paroxetine, sertraline).
Side effects
Side effects
Digestive system disorders: abdominal pain and discomfort (including epigastrium), flatulence, nausea, constipation, diarrhea, peptic ulcer, GI bleeding, anorexia, vomiting, dyspepsia, stomatitis, glossitis, pancreatitis, hepatitis.
CNS: dizziness, headache, increased appetite, sedation, somnolence, amnesia, anxiety, depression, anxiety, hallucinations, insomnia, unusual dreams, nervousness, paresthesia, personality changes, tremor, akathisia.
Senses: tinnitus, vertigo, deafness, blurred vision, eye irritation or edema.
Laboratory data: increased activity of liver enzymes (LDH, alkaline phosphatase, transaminases), increased concentration of urea nitrogen, hypercreatininemia, decreased Hb and hematocrit, hypoproteinemia, thrombocytopenia, leukopenia, anemia (including aplastic hematocrit).Anemia (including aplastic and hemolytic), hyperglycemia, hypoglycemia, detection of antinuclear antibodies.
Cardiovascular disorders: blood pressure increased, tachycardia, palpitations, blood rushes to the skin.Percutaneous disorders: rash (including petechial), purpura, including thrombocytopenic, ecchymoses, itching, onycholysis, alopecia, photosensitization, exfoliative dermatitis, erythema multiforme, Lyell syndrome, Stevens-Johnson syndrome, vesicle-bullez reactions.
Urinary system: edema, dysuria, frequent urination, hematuria, oliguria, menorrhagia.
Respiratory system: nasal bleeding, dyspnea.
Other: pain in the chest, thirst, chills, increased sweating, weight gain or loss.
Overdose
Overdose
Symptoms: nausea, vomiting, abdominal pain, drowsiness, lethargy; GI bleeding, increased BP, acute renal failure, respiratory depression, coma.
Treatment: symptomatic. There is no specific antidote. In case of recent overdose we should induce vomiting, take osmotic laxatives, activated carbon (60-100 g for adults, 1-2 g/kg for children). Multiple use of activated charcoal (6 hours after intake) reduces T1/2 of piroxicam by 50% and its bioavailability by 37%.
Forced diuresis, urine alkalinization, hemodialysis and hemoperfusion are ineffective (high plasma protein binding).
Similarities
Similarities
Additional information
| Weight | 0.016 kg |
|---|---|
| Manufacturer | Manufacturing of medicines, Russia |
| Medication form | capsules |
| Brand | Manufacturing of medicines |
Other forms…
Related products
Buy Piroxicam, 10 mg capsules 20 pcs with delivery to USA, UK, Europe and over 120 other countries.