No products in the cart.
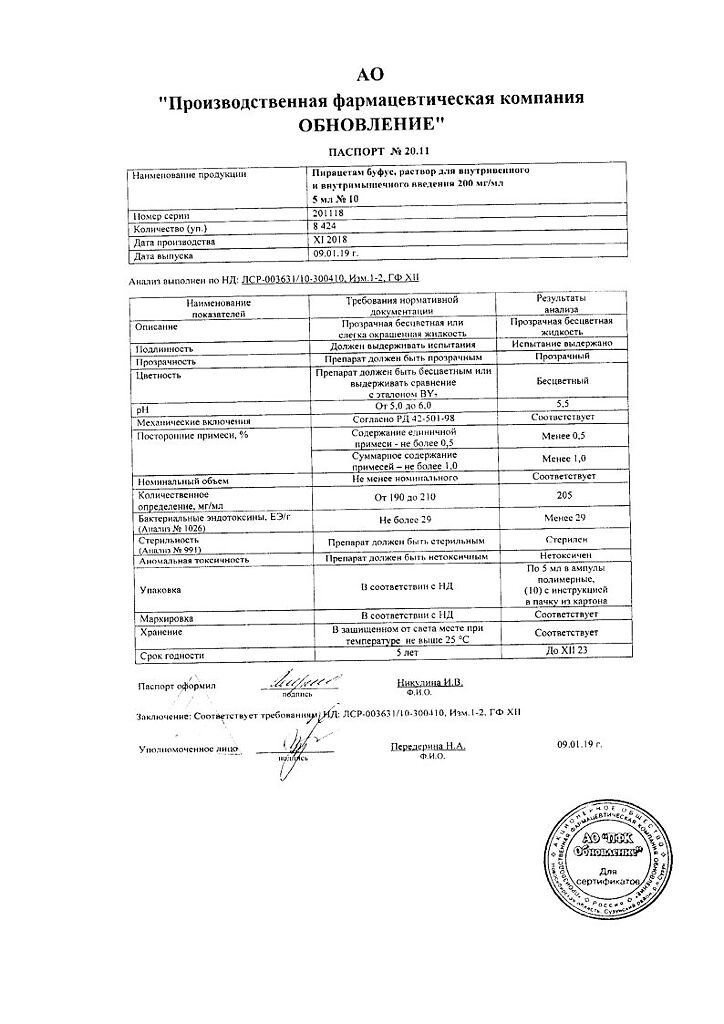
Piracetam bufus Reneval, 200 mg/ml 5 ml 10 pcs
€3.88 €3.53
Description
Pharmacotherapeutic group: Nootropic agent
Pharmacological action
Notropic agent. It has a positive effect on metabolic processes and blood circulation of the brain. Increases glucose utilization, improves metabolic processes, improves microcirculation in ischemic areas, and inhibits activated platelet aggregation. It has a protective effect in brain damage caused by hypoxia, intoxication, electroshock. It improves integrative activity of the brain. It has no sedative and psychostimulant effect.
Pharmacokinetics
Indications
Indications
Alzheimer’s disease, Epilepsy, Tinnitus, Seizures, Stroke sequelae, Impaired cerebral circulation, Mental disorders, Dizziness, Learning disabilities, Schizophrenia, Concussion and other brain injuries, Acquired dementia, Alcoholism, Mental retardation in children, Nausea, Headache, Impaired attention and memory, Disorientation
reduction of manifestations of cortical myoclonias in piracetam-sensitive patients, both as monotherapy and as part of complex therapy (a trial course of treatment may be conducted to determine sensitivity to piracetam in a particular case).
Psychiatric treatment of intellectual and mental disorders in the absence of a diagnosed dementia.li>
Symptomatic treatment of intellectual and mental disorders in the absence of an established diagnosis of dementia;
.
Active ingredient
Active ingredient
Piracetam
Composition
Composition
1 ml of the solution contains:
active ingredient:
piracetam – 200 mg
excipients:
sodium acetate – 1 mg
acetic acid – to pH 5.8
water for injection – to 1 ml.
How to take, the dosage
How to take, the dosage
Intravenously. Intramuscular.
Parenteral administration of piracetam is prescribed when oral forms of the drug cannot be used (unconsciousness, difficulty in swallowing).
Intravenous administration is preferable.
The daily dose is infused intravenously through a catheter at a constant rate over a 24-hour period. The total volume of solution to be infused is based on the clinical indications and the patient’s condition.
The bolus intravenous infusion is given for at least 2 minutes; the daily dose is divided into several infusions (2-4) at regular intervals so that the dose per infusion does not exceed 3 g.
Interaction
Interaction
The possibility of changes in piracetam pharmacokinetics under the influence of other drugs is low, because 90% of the drug is excreted unchanged by the kidneys.
In concurrent use with thyroid hormones there have been reports of confusion, irritability and sleep disturbance.
Special Instructions
Special Instructions
With caution, use in patients with significant impairment of hemostasis, major surgery and severe bleeding; with renal insufficiency.
Continuous monitoring of renal function parameters is recommended.
If sleep disturbances occur, it is recommended that the evening dose of piracetam be stopped and this dose be added to the daytime intake.
Contraindications
Contraindications
- Individual intolerance to piracetam or pyrrolidone derivatives, as well as other drug components.
- Gentington’s chorea.
- Acute cerebral circulation disorder (hemorrhagic stroke).
- Final stage of renal failure (with creatinine clearance less than 20 ml/min).
- Motor agitation at the time of prescription.
- Children under 3 years of age.
With caution:hemostasis disorders; extensive surgical interventions; severe bleeding.
Side effects
Side effects
Central nervous system and peripheral nervous system: motor disinhibition (1.72%), irritability (1.13%), somnolence (0.96%), depression (0.83%), asthenia (0.23%).
In single cases – headache, insomnia, agitation, impaired balance, ataxia, aggravation of the course of epilepsy, anxiety, hallucinations, confusion.
Cardiovascular system disorders: increase or decrease of blood pressure.
Digestive system disorders: in single cases – nausea, vomiting, diarrhea, abdominal pain (including gastralgia).
Metabolism: weight gain (1.29%).
Skin disorders – dermatitis, itching, rash.
Overdose
Overdose
There has been a single case of dyspeptic complaints in the form of diarrhea with blood and abdominal pain when an oral dose of 75 g was taken.
This appears to be related to the use of a large total dose of sorbitol, which was previously used in the oral dosage form. No other cases of overdose have been identified.
Treatment: in case of overdose it is recommended to carry out symptomatic therapy, which may include hemodialysis. There is no specific antidote.
The effectiveness of hemodialysis for piracetam is 50-60%.
Pregnancy use
Pregnancy use
Animal studies have shown no direct or indirect effects on pregnancy, embryo/fetal development, delivery or postnatal development. No controlled studies on the use of the drug during pregnancy have been conducted. Piracetam penetrates the placental barrier.
The drug concentration in newborns reaches 70-90% of its concentration in blood in the mother. Piracetam should be administered during pregnancy only in exceptional cases if the benefit to the mother exceeds the potential risk to the fetus, and the clinical condition of the pregnant woman requires treatment with piracetam.
Piracetam penetrates into the breast milk. Piracetam should not be used while breastfeeding, or breastfeeding should be discontinued during treatment with piracetam. When deciding whether to stop breastfeeding or to discontinue treatment with piracetam, the benefit of breastfeeding for the child must be weighed against the benefit of therapy for the woman.
Similarities
Similarities
Nootropil, Piracetam, Notrocetam
Additional information
| Weight | 0.090 kg |
|---|---|
| Shelf life | 5 years. |
| Conditions of storage | In the light-protected place at the temperature not more than 25 °C. Keep out of reach of children. |
| Manufacturer | Update PFC AO, Russia |
| Medication form | solution |
| Brand | Update PFC AO |
Related products
Buy Piracetam bufus Reneval, 200 mg/ml 5 ml 10 pcs with delivery to USA, UK, Europe and over 120 other countries.