No products in the cart.
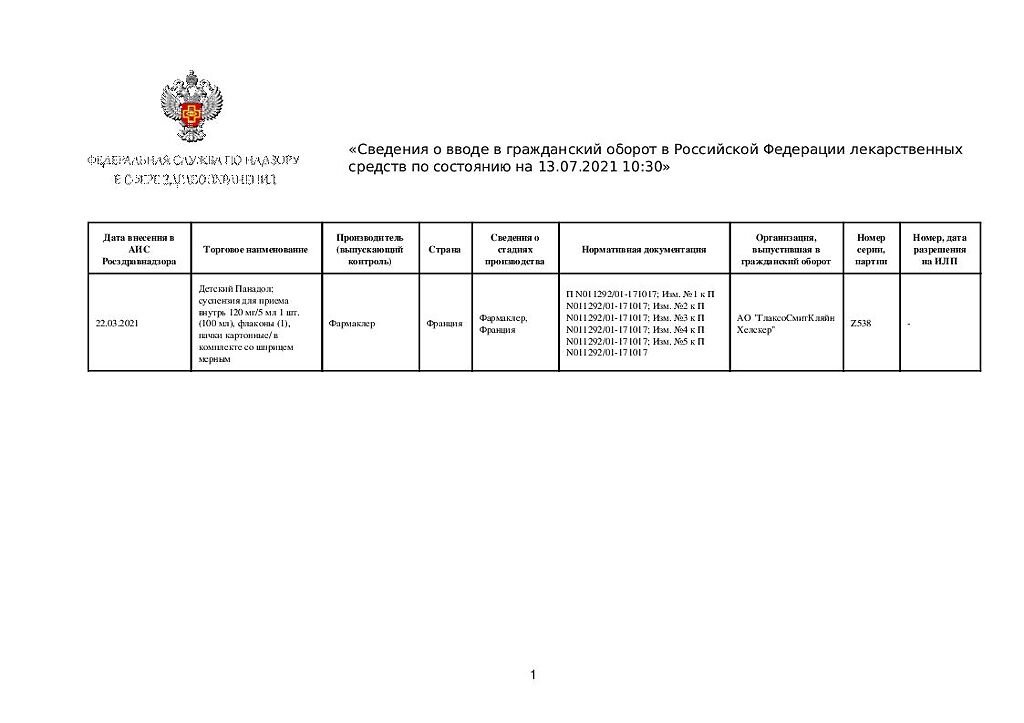
Panadol Baby, 120 mg/5 ml suspension 100 ml
€1.00
Out of stock
(E-mail when Stock is available)
Description
The drug has analgesic and antipyretic properties.
Blocks cyclooxygenase in the central nervous system, affecting the centers of pain and thermoregulation.
Anti-inflammatory effect is virtually absent. It has no effect on the state of the gastrointestinal mucosa and water-salt metabolism because it does not affect the synthesis of prostaglandins in the peripheral tissues.
Indications
Indications
Infectious diseases, pain, migraine, toothache, headache, flu, pain in the joints (arthralgia).
In children from 3 months to 12 years as an antipyretic – to reduce the body temperature in colds, influenza and childhood infectious diseases (chickenpox, mumps, measles, rubella, scarlet fever and others.
For children aged 2-3 months of life a single use is possible to reduce the fever after vaccination. If the temperature does not decrease, you need to consult a doctor.
Active ingredient
Active ingredient
Paracetamol
Composition
Composition
5 ml of suspension contains paracetamol 120 mg;
excipients:
malic acid,
xanthan gum,
maltitol (glucose syrup hydrogenate),
sorbitol, <
citric acid,
Nipasept sodium,
strawberry flavoring,
surfactant,
water.
How to take, the dosage
How to take, the dosage
For oral administration.
The contents of the bottle should be well shaken for at least 10 seconds before use. The measuring syringe placed inside the package allows dosing the preparation correctly. The drug dose depends on the age and body weight of the child.
Children (from 3 months to 12 years)
Maximum single dose is 15 mg/kg of body weight. The maximum daily dose is 60 mg/kg of body weight. The frequency of administration should be not more than 3-4 times a day. If necessary the recommended dose may be given to the child every 4-6 hours but not more than 4 doses within 24 hours.
In all other cases the drug “Children’s Panadol” should be consulted before taking it.
The duration of use without medical advice – 3 days.
Do not exceed the recommended dose!
In case of accidental exceeding of the recommended dose you should immediately consult a physician, even if the child is feeling well, because there is a risk of liver damage (see “Overdose”).
If the condition of the child does not improve during the drug administration, consult a physician.
Interaction
Interaction
If the child is already taking other medicines, before starting to take the drug “Children’s Panadol” you should consult your doctor.
Barbiturates, carbamazepine, phenytoin, diphenin, primidone and other anticonvulsants, ethanol, rifampicin, zidovudine, flumecinol, phenylbutazone, butadion, preparations of St. John’s wort and other inducers of microsomal oxidation increase the production of hydroxylated active metabolites, causing the possibility of severe liver damage in small overdoses of paracetamol (5 g or more).
Inhibitors of microsomal liver enzymes decrease the risk of hepatotoxic action.
Under the influence of paracetamol the excretion time of levomycetin (chloramphenicol) increases by 5 times, thereby increasing the risk of levomycetin (chloramphenicol) poisoning.
When taken regularly for a long time the drug increases the effect of indirect anticoagulants (warfarin and other coumarins), which increases the risk of bleeding. Episodic administration of a single dose of the drug has no significant effect on the effect of indirect anticoagulants. Metoclopramide and domperidone increase, and colestyramine reduces the rate of absorption of paracetamol. The drug may reduce the effectiveness of uricosuric drugs.
Special Instructions
Special Instructions
Children under 3 months of age and children born prematurely, “”Children’s Panadol”” may be given only as prescribed by the doctor.
During tests to determine uric acid and blood sugar levels, inform the doctor about the use of the drug “Children’s Panadol”.
When taking the drug for more than 7 days it is recommended to monitor the peripheral blood and functional status of the liver. Deficit of glutathione due to eating disorders, cystic fibrosis, HIV-infection, starvation, exhaustion determines the possibility of development of severe liver damage in small overdoses of paracetamol (5 g and more).
The drug should not be used simultaneously with other paracetamol-containing drugs.
“Children’s Panadol” contains maltitol and sorbitol. Patients with rare hereditary fructose intolerance should not take this medicine. The drug contains a mixture of esters of parahydroxybenzoic acid (methyl-, ethyl-, propyl-parahydroxybenzoates sodium), which may cause delayed allergic reactions.
“Children’s Panadol” contains no sugar, alcohol and acetylsalicylic acid.
Synopsis
Synopsis
Oral suspension is pink to dark pink, opaque, viscous, strawberry-scented; crystals may be present in the suspension.
Contraindications
Contraindications
Hypersensitivity to paracetamol or any other ingredient of the drug; marked liver or renal dysfunction; period of infancy.
Use with caution in patients with liver dysfunction (including Gilbert syndrome).Including Gilbert’s syndrome), renal, genetic absence of glucose-6-phosphate dehydrogenase enzyme, severe forms of blood diseases (severe anemia, leukopenia, thrombocytopenia). If any of the above diseases are present, consult a physician before taking the drug.
Side effects
Side effects
The drug is usually well tolerated in the recommended doses. The following side effects were detected spontaneously during post-registration use of the drug.
Side effects are classified by organ system and frequency. The frequency of side effects is defined as follows: very frequently (greater than or equal to 1/10), frequently (greater than or equal to 1/100 and less than 1/10), infrequently (greater than or equal to 1/1000 and less than 1/100), rarely (greater than or equal to 1/10 000 and less than 1/1000) and very rarely (greater than or equal to 1/100 000 and less than 1/10 000).
Allergic reactions:
Very rare – in the form of skin rashes, itching, urticaria, angioedema, Stevens-Johnson syndrome, anaphylaxis;
On the blood forming system:
Very rarely – thrombocytopenia, anemia, leukopenia
Respiratory system:
Very rare – bronchospasm (in patients with hypersensitivity to acetylsalicylic acid and other non-steroidal anti-inflammatory drugs);
Hepatobiliary system disorders:
Very rare – liver function disorder.
Sometimes nausea, vomiting, pain in the stomach area are possible.
If any of the above side effects occur, stop taking the drug and immediately consult a physician.
Overdose
Overdose
Symptoms: during the first 24 hours after overdose nausea, vomiting, stomach pain, sweating, pale skin, anorexia. After 1-2 days, signs of liver damage are determined (soreness in the liver area, increased activity of “liver” enzymes). Abnormal carbohydrate metabolism and metabolic acidosis may develop.
In adults the liver damage occurs after ingestion of more than 10 g of paracetamol; in children – when administered more than 125 mg/kg of child body weight. In the presence of the factors influencing the liver toxicity of paracetamol (see section “Interaction with other medicinal products”, “Cautions”) liver damage is possible after ingestion of 5 or more grams of paracetamol.
In severe cases of overdose as a result of liver failure may develop encephalopathy (brain impairment), bleeding, hypoglycemia, brain edema, up to and including death. Acute renal failure with acute tubular necrosis may develop, characterized by pain in the lumbar region, hematuria (blood or red blood cells in the urine), proteinuria (high protein content in the urine), while severe liver damage may be absent. There have been observed the cases of heart rhythm disorders, pancreatitis.
In case of prolonged use in excess of the recommended dose hepatotoxic and nephrotoxic effect may be observed (renal colic, nonspecific bacteriuria, interstitial nephritis, papillary necrosis).
Treatment: If overdose is suspected, even if the first symptoms are not expressed, the drug shall be discontinued and medical attention shall be sought immediately. Within 1 hour after overdose it is recommended to wash the stomach and take enterosorbents (activated charcoal, polyphepan). It is necessary to determine the level of paracetamol in blood plasma, but not earlier than 4 hours after overdose (earlier results are unreliable).
Acetylcysteine administration within 24 hours after overdose. Maximum protective effect is provided during the first 8 hours after overdose, with time the effectiveness of the antidote decreases sharply. If necessary, acetylcysteine is administered intravenously. If there is no vomiting before the patient is admitted to the hospital, methionine may be used.
The need for additional therapeutic measures (further administration of methionine, intravenous administration of acetylcysteine) is determined depending on the concentration of paracetamol in the blood, as well as on the time elapsed after its administration. Treatment of patients with severe liver dysfunction 24 hours after paracetamol administration should be carried out together with specialists from a toxicology center or a specialized department of liver diseases.
Similarities
Similarities
Efferalgan, Cefekon D, Paracetamol, Paracetamol for children, Paracetamol Renewal, Paracetamol, tablets 500 mg 10 pcs
Additional information
| Weight | 0.235 kg |
|---|---|
| Shelf life | 3 years. |
| Conditions of storage | At the temperature not more than 30 ° C. Protect from light. Do not freeze. Keep out of reach of children. |
| Manufacturer | Farmaclaire, France |
| Medication form | oral suspension |
| Brand | Farmaclaire |
Related products
Buy Panadol Baby, 120 mg/5 ml suspension 100 ml with delivery to USA, UK, Europe and over 120 other countries.