No products in the cart.
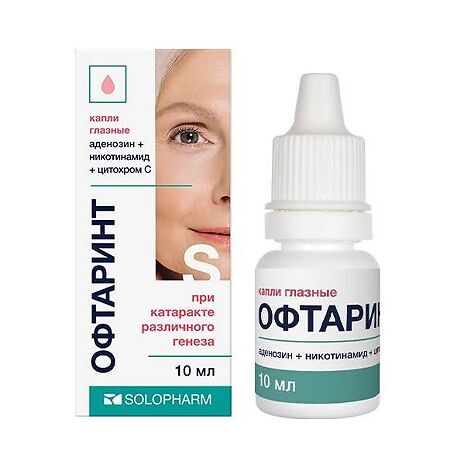
Oftarint, eye drops 10 ml
€10.47 €9.16
Description
Pharmacotherapeutic group: Tissue repair stimulator.
ATX code: S01XA
Pharmacological properties
Pharmacodynamics
Complex drug that improves energy metabolism of the lens. Cytochrome C plays an important role in biochemical redox processes in the eyeball tissues and is an antioxidant. Adenosine is a precursor of adenosine triphosphate (ATP) and takes part in the metabolic processes of the lens. Nicotinamide stimulates the synthesis of nicotinamide dinucleotide (NAD), a cofactor of dehydrogenases.
Pharmacokinetics
When used topically, cytochrome C is not absorbed into the systemic bloodstream and is fully metabolized in the body. The breakdown of cytochrome C follows the same metabolic pathways as the breakdown of amino acids, and heme, which is broken down to bilirubin, is excreted in the bile.
Adenosine passes freely through the cornea. The half-life of adenosine from plasma is less than one minute, it is metabolized in almost all tissues to form inosine, xanthine and urate, which are freely excreted with the urine. Ribose, which is a constituent of adenosine, is metabolized to glyceraldehyde-3-phosphate, then to pyruvate and is burned in the Krebs cycle.
Nicotinamide is partially metabolized to nicotinic acid. Both of these compounds are methylated to N-methylnicotinamide, which is further broken down in the liver. Metabolites and unchanged nicotinamide are excreted in the urine.
Indications
Indications
Cataract of various genesis.
Active ingredient
Active ingredient
Adenosine, Nicotinamide, Cytochrome C
Composition
Composition
Composition of the drug per 1 ml:
The active ingredients:
Adenosine 2.0 mg
Nicotinamide 20.0 mg
Cytochrome C 0.675 mg
Excipients:
Sorbitol 10.0 mg
Sodium dihydrophosphate dihydrate 4.415 mg
Sodium hydrophosphate dihydrate 2.847 mg
/p>
Sodium succinate hexahydrate 1.0 mg
Benzalkonium chloride 0.04 mg
Injection water up to 1 ml.
How to take, the dosage
How to take, the dosage
Topically, 1-2 drops into the conjunctival sac 3 times a day.
The order of the vial:


- Turn the vial counterclockwise to open and remove the cap.
- Carefully turn the vial upside down without touching the tip with your fingers and place it between your thumb and forefinger with one hand.
- Tilt your head backwards, place the tip of the vial over your eye and with the forefinger of your other hand pull the lower eyelid down. Slightly press the bottle and drop the desired amount into the conjunctival sac of the eye.
Contact of the tip of the open bottle with the surface of the eye and your hands should be avoided. - After use put the cap on the bottle and close by rotating it clockwise.
After opening the bottle the drug may be used within 1 month. At the end of this period the product should be discarded.
The product should not be used if the vial is visibly damaged.
Special Instructions
Special Instructions
Benzalkonium chloride, which is often used as a preservative in ophthalmic products, can cause keratopathy and/or toxic ulcerative keratopathy. Because it contains benzalkonium chloride, close monitoring is required for frequent or prolonged use in patients with dry eye syndrome or in cases of corneal damage. Benzalkonium chloride may discolor soft contact lenses. Soft contact lenses should be removed before application and put on again 15 minutes after dosing.
Influence on driving and operating ability
Patients who have transient eye irritation after injection should not drive a vehicle or operate machinery, machines, or any other equipment requiring good visual acuity immediately after using the eye drops.
Contraindications
Contraindications
Individual hypersensitivity to the drug components, children under 18 years of age (due to the lack of data on the effectiveness and safety of the drug in children and adolescents under 18 years).
Side effects
Side effects
Ocular side effects: transient burning and tingling of the eyes, allergic conjunctivitis, contact dermatitis.
Systemic side effects are extremely rare. Short-term nausea, arterial hypotension, dizziness and shortness of breath may be observed. Nicotinic acid has a vasodilator effect and may cause a feeling of fever, fainting, and a throbbing sensation in the temples.
Pregnancy use
Pregnancy use
There is no sufficient experience of using the drug during pregnancy and breastfeeding. It is possible to use the drug for treatment during pregnancy and during breastfeeding if the expected therapeutic effect for the mother justifies the potential risk for the fetus and child.
Similarities
Similarities
Oftan Katahrom
Additional information
| Weight | 0.021 kg |
|---|---|
| Shelf life | 3 years. After opening the bottle – 1 month. Do not use after the expiration date! |
| Conditions of storage | At a temperature of 2 to 8 ° C. Store out of the reach of children. |
| Manufacturer | Grotex Ltd, Russia |
| Medication form | eye drops |
| Brand | Grotex Ltd |
Related products
Buy Oftarint, eye drops 10 ml with delivery to USA, UK, Europe and over 120 other countries.