No products in the cart.
Nicorette, spray 1 mg/dose 150 doses of fruit and mint
€32.70 €28.34
Description
Nicorette® Spray alleviates and/or prevents smoking cravings and withdrawal symptoms that occur with tobacco addiction.
It is indicated as support for smokers who intend to quit smoking or reduce the number of cigarettes smoked before giving up completely; to help smokers who do not want to smoke or in the absence of such an opportunity; and as a safer alternative to smoking.
Active ingredient
Active ingredient
Nicotine
Composition
Composition
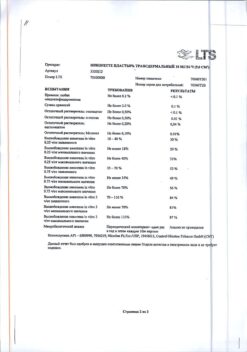
1 ml of product contains:
Active ingredient:
nicotine – 13.6 mg;
Excipients: Propylene glycol – 150.0 mg, ethanol – 97.0 mg, tromethamol – 40.5 mg, poloxamer – 40.0 mg, glycerol – 25.0 mg, sodium hydrocarbonate – 14.3 mg, red fruit flavoring 911441 – 8.0 mg, levomenthol – 5,0 mg, Cooler 2 flavoring SN046680 – 3.0 mg, sucralose – 1.5 mg, acesulfame potassium – 1.5 mg, 10% hydrochloric acid – sufficient to pH 9, purified water – sufficient to 1 ml..
How to take, the dosage
How to take, the dosage
For application on the mucous membrane of the oral cavity.
The patient should do everything possible to quit smoking permanently during the treatment with Nicorette® Spray.
Adults over 18 years
Nicorette® Spray should be used at the moment when there is an irresistible desire to smoke. After preparing the spray for use (see below Instructions for use when using the spray for the first time) put the tip of the spray as close as possible to the open mouth. Press the dispenser upward, thus releasing one dose of the product into your mouth; avoid getting the spray on your lips. To prevent the substance from entering the respiratory tract, do not inhale while pressing the pipette. For best results, do not swallow saliva for a few seconds after injection.
It is not recommended to eat or drink liquid during the use of the spray.
In case of overdose symptoms (see section “Overdose”) the drug should be stopped immediately.
Complete smoking cessation
Nicorette® Spray should be used in all cases of craving for smoking or to avoid cravings in situations that may provoke it.
Smokers who are willing or able to stop smoking immediately should immediately replace cigarette smoking with Nicorette® Spray and, as soon as possible, reduce the number of injections until they cease completely.
For complete smoking cessation, make 1 or 2 injections at a time when you would normally have smoked a cigarette and when cravings arise. If cravings do not decrease within a few minutes after a single injection, a second injection should be given. If two doses are required, the subsequent application of the spray may consist of 2 consecutive injections.
No more than 4 dosed injections of the spray may be taken every hour. No more than 2 doses of spray should be injected at a time or more than 64 doses over a 24-hour period (or 4 doses per hour for 16 hours).Each vial contains at least 150 doses.
The average course of application of the spray at the indicated dose is 6 weeks. The number of injections should then begin to be reduced so that by the end of week 9, the number of doses is half of the average daily dose received during the first 6 weeks, and during week 12, no more than 4 doses per day. When the daily dose is reduced to 2-4 injections, the spray should be discontinued.
After therapy to prevent a return to smoking, patients may use Nicorette® Spray if they have an irresistible desire to smoke. In such situations, 1 injection can be made, and if after a single injection the craving for smoking is not reduced within a few minutes, a second injection should be made. This should not exceed 4 metered injections per day.
Regular use of the spray for more than 6 months is generally not recommended, but some patients may require longer therapy to avoid relapse to smoking.
Reducing the number of cigarettes smoked
Smokers who wish to reduce the number of cigarettes smoked should use the spray as needed between smoking episodes in order to increase the intervals between smoking and to reduce smoking as much as possible.
As soon as readiness is felt, smokers should aim for complete smoking cessation.
After smoking cessation, the recommendations of therapy and gradual dose reduction outlined above for complete smoking cessation should be followed.
Behavioral therapy and psychological support usually increase treatment success. Those who have been successful in quitting smoking but have difficulty quitting the spray are advised to seek medical advice from a physician.
Temporary smoking cessation
The spray can be used at times when you need to abstain from smoking, such as when you are in non-smoking areas or other situations where you need to abstain from smoking. The maximum daily dose for temporary smoking cessation is 64 doses.
In combination with transdermal patch
For smokers with strong nicotine addiction (more than 20 cigarettes per day) or those who have irresistible craving for cigarettes or smokers who could not give up smoking with only one nicotine replacement therapy, oral mucosa spray Nicorette ® can be used in combination with nicorette ® transdermal patch for quick relief of smoking craving.
The patch is applied to an intact area of skin immediately after waking up in the morning and removed before bedtime. The patch should be applied to dry, clean, undamaged, hair-free skin such as the thighs, upper extremities or chest. It is necessary to change the place of application every day: you should not use the same area for the next two days. After applying the patch, wash your hands thoroughly to avoid eye irritation from possible nicotine ingestion.
Initial Therapy:
Treatment should be started with the patch 25 mg/16 hours (Stage 1) in combination with a 1 mg/dose spray. Thirteen doses of spray per day are usually sufficient. The maximum daily dose of the spray is 32 doses.
Patients should completely stop smoking during therapy. Usually the total course of treatment lasts for 8 weeks. After that the nicotine dose should be gradually reduced.
Cancellation of combination therapy:
Cancellation of combination therapy can be done in two ways.
Method 1: For the next 2 weeks, switch from the 25 mg/16 hour patch (Stage 1) to the 15 mg/16 hour patch (Stage 2) and then, for the next 2 weeks, to the 10 mg/16 hour patch (Stage 3), while maintaining the number of Nicorette® spray doses used, if necessary, as with Initial Therapy. Then gradually reduce the number of doses of the spray until complete abolition within the time that the patient needs depending on his/her needs, but no later than 12 months after the start of combined therapy.
Method 2: consists in complete abolition of the patch immediately after the end of the Initial Therapy stage. Then gradually reduce the number of doses of Nicorette® Spray until complete withdrawal within the time required by the patient depending on their needs, but no later than 12 months after the start of combined therapy.
Children and adolescents under 18 years
The drug is not recommended for persons under 18 years of age. There is no experience of treating adolescents under 18 years of age with the spray.
Interaction
Interaction
No clear clinically significant interaction between nicotine replacement therapy and other drugs has been established. Nevertheless, theoretically, nicotine can increase the hemodynamic effects of adenosine, i.e., it can lead to an increase in blood pressure and heart rate, as well as increase the response to pain (angina-like chest pain) provoked by adenosine administration.
Special Instructions
Special Instructions
Nicorette® use is accompanied by lower risks than smoking.
The risk-benefit ratio should be evaluated by a physician of the appropriate specialty in patients with the following conditions:
Comorbid cardiovascular disease
In the stable course of cardiovascular disease, Nicorette® Spray causes less harm than continued smoking. However, smokers with recent myocardial infarction, unstable angina or worsening angina, including Prinzmetal angina, severe arrhythmias, recent cerebrovascular disease and/or patients with uncontrolled hypertension should be advised to stop smoking without pharmacological intervention. If such attempts are unsuccessful, Nicorette® Spray may be considered, but since safety data in this patient population are limited, such treatment should only be initiated under close medical supervision.
Diabetes mellitus
Patients with diabetes mellitus after stopping smoking and starting nicotine replacement therapy are advised to monitor blood glucose concentrations more closely, since a decrease in catecholamines whose release is induced by nicotine may affect carbohydrate metabolism.
Allergic reactions
The drug should be used with caution in patients prone to angioedema and urticaria.
Gastrointestinal diseases
Ingested nicotine may exacerbate symptoms of esophagitis, gastritis or peptic ulcer, so the use of oral nicotine replacement therapy in the above pathology should be used with caution.
Hepatic and renal disorders
In patients with moderate to severe hepatic failure and (or) severe renal failure, the drug should be used with caution because the clearance of nicotine and its metabolites may be reduced, which may increase the risk of adverse events.
Dangers to young children
Doses of nicotine easily tolerated by adult smokers and teenage smokers can cause severe intoxication in children, which can lead to death. It is important not to leave preparations containing nicotine unattended, as this can lead to misuse and ingestion by children (see “Overdose” section).
Pheochromocytoma and uncontrolled hyperthyroidism
In patients with uncontrolled hyperthyroidism and pheochromocytoma, the drug should be used with caution because nicotine causes catecholamine release.
Formation of dependence
Dependence on the drug may develop, but it is less dangerous to health and more easily overcome than smoking addiction.
Smoking cessation
The polycyclic aromatic hydrocarbons in tobacco smoke induce the metabolism of drugs metabolized by the CYP1A2 isoenzyme (and possibly CYP1A1). Smoking cessation may cause slower metabolism and, consequently, increased blood concentrations of these drugs. This has potential clinical implications for drugs with a narrow therapeutic index – for example, theophylline, tacrine, clozapine, and ropinirole.
Excipients
Nicorette® Spray contains a small amount of ethanol (alcohol) – less than 10 mg per dose.
When using Nicorette® Spray, care should be taken to avoid getting the spray in the eyes.
Warnings and precautions for combined therapy with Nicorette® Transdermal Patch are similar to those for each of the drugs separately.
If the medication has expired or is out of date, do not throw it in the sewage or on the street! Place the medication in a bag and put it in a trash container. These measures will help protect the environment!
Synopsis
Synopsis
Oral mucosal spray is a dosed colorless to light yellow transparent or slightly opalescent solution with a peppermint odor.
Contraindications
Contraindications
– Hypersensitivity to nicotine or other ingredients of the drug.
– Children under 18 years of age.
Side effects
Side effects
Regardless of the form of nicotine medication used, some symptoms may result from nicotine withdrawal due to smoking cessation. These include: dysphoria or depressed mood; insomnia; irritability, frustration or anger; anxiety; difficulty concentrating, restlessness or impatience; decreased heart rate; increased appetite or weight gain; cough; aphthous ulcers; nasopharyngitis. A causal relationship has not been established.
In addition, other symptoms associated with smoking cessation have also been reported in individuals using oral mucosal spray: dizziness, presyncopal conditions, coughing, constipation, and bleeding gums.
Nicotine craving, considered a clinically significant symptom, is an important manifestation of nicotine withdrawal after smoking cessation.
Nicorette® Spray may cause nicotine-related adverse reactions similar to those observed with other nicotine-containing medications, these reactions are primarily dose-dependent.
Allergic reactions (such as angioedema, urticaria and anaphylactic shock) may occur in predisposed persons.
Most adverse reactions to Nicorette® Spray were observed in the early phase of treatment and are similar to those for oral drugs. In the first few days of treatment there may be irritation of the mucous membranes of the oral cavity and pharynx, hiccups are common. Continuation of treatment leads to adaptation.
Daily data collection from the subjects of the study showed that very often the emerging adverse events are manifested in the first 2-3 weeks of application of the spray and subsequently disappear.
Overdose
Overdose
When used as directed, nicotine overdose symptoms may occur in patients with low nicotine intake prior to treatment or when different nicotine sources are used simultaneously.
Symptoms
The minimum lethal dose in acute overdose for an unaccustomed adult is 40-60 mg of nicotine. In overdose, the same symptoms as in acute nicotine poisoning are observed, namely nausea, vomiting, increased salivation, abdominal pain, diarrhea, sweating, headache, dizziness, hearing impairment and pronounced general weakness. At higher doses, arterial hypotension, a weak and irregular pulse, respiratory distress, impaired consciousness, collapse and generalized seizures may join them.
Doses of nicotine that are well tolerated during treatment by adult smokers may cause symptoms of severe poisoning in young children and even lead to death. Suspicion of nicotine poisoning in children should be treated as an emergency requiring immediate hospitalization.
Treatment
Nicotine use should be stopped immediately and symptomatic treatment started. If necessary, artificial pulmonary ventilation should be started. Taking activated charcoal prevents absorption of nicotine in the gastrointestinal tract.
Pregnancy use
Pregnancy use
Pregnancy
Smoking during pregnancy is associated with risks such as delayed intrauterine development, premature birth or stillbirth. Smoking cessation is the single most effective intervention to improve the health of both the pregnant woman and her baby. Early smoking cessation is the best option.
Nicotine penetrates the placental barrier and affects fetal respiratory activity and blood flow. The effect on circulation is dose-dependent.
Therefore, ideally smoking cessation during pregnancy should be done without nicotine replacement therapy. However, if a woman is unable (or it is assumed) to quit smoking without pharmacological support, nicotine replacement therapy is used because continued smoking carries a greater risk to the fetus than nicotine replacement therapy. Complete smoking cessation is the best option, but if that is not achievable, Nicorette® spray can be used as a safer alternative to smoking during pregnancy. Because of potential nicotine-free periods, intermittent dosage forms are preferred, but patches may be necessary if there is significant nausea and/or vomiting. The use of Nicorette® Spray by pregnant women should be initiated only after consultation with a physician.
Breastfeeding
Nicotine penetrates into breast milk in amounts that may affect the baby even when the drug is used in therapeutic doses. Therefore, you should refrain from using Nicorette® spray during breastfeeding. If you have not managed to stop smoking, the drug should be started only after consultation with your doctor.
Medicinal forms for occasional use minimize the nicotine content in breast milk and allow breast-feeding at the lowest concentration of nicotine in it. In order to reduce the negative effects of nicotine on the baby, the drug should be used immediately after breastfeeding. The periods between the use of the drug and the next feeding should be as long as possible (recommended at least 2 hours).
Smoking increases the risk of infertility in men and women. In vitro studies have shown that nicotine negatively affects sperm quality in men.
Additional information
| Weight | 0.057 kg |
|---|---|
| Conditions of storage | Store at a temperature not exceeding 25 ° C. Keep out of reach of children. |
| Manufacturer | McNeil AB, Sweden |
| Medication form | oral mucous membrane dispensable spray |
| Brand | McNeil AB |
Other forms…
Related products
Buy Nicorette, spray 1 mg/dose 150 doses of fruit and mint with delivery to USA, UK, Europe and over 120 other countries.