Subtotal: €3.64
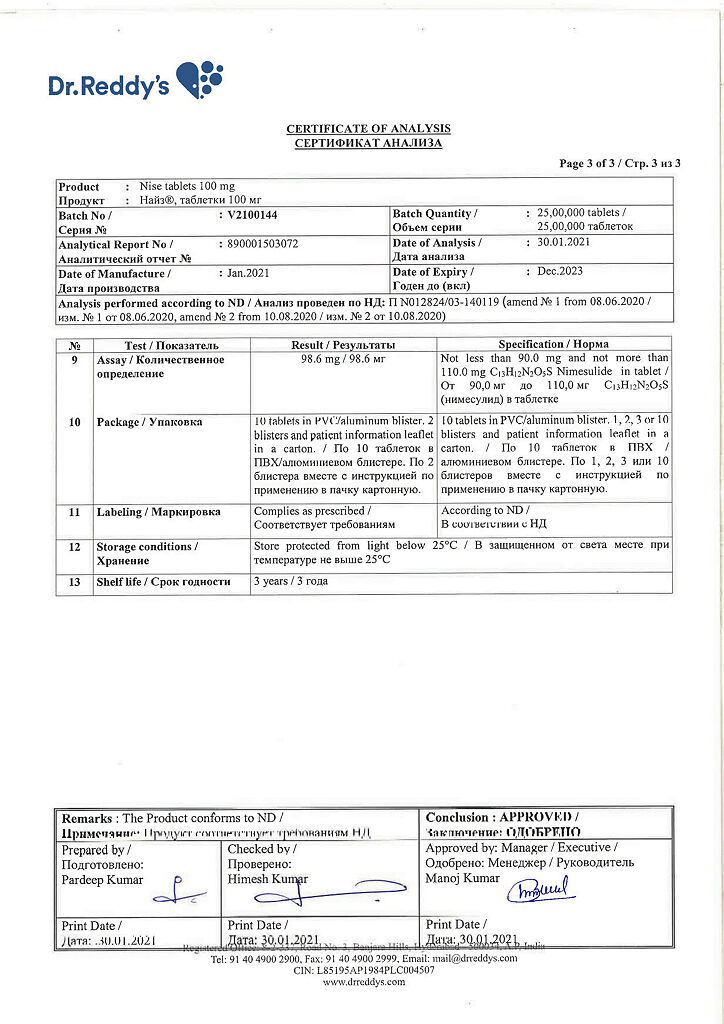
Nais, tablets 100 mg 20 pcs
€7.48 €6.54
Pharmacotherapeutic group: nonsteroidal anti-inflammatory drug (NSAID).
CodeATX:M01AX17.
Indications
Lumbago, Gout, Migraine, Toothache, Inflamed tendons, Fever, Back pain, Sprains and strains, Myalgia (muscle pain), Arthritis, Radiculitis, Headache, Sciatica, Pain after injuries and operations, Rheumatoid arthritis, Osteochondrosis, Bursitis, Myositis, Neck pain, Arthritis, Joint pain (arthralgia), Painful menstruation (algodysmenorrhea), Pain
– rheumatoid arthritis;
– joint syndrome with gout exacerbation;
– psoriatic arthritis;
– ankylosing spondylitis;
– osteochondrosis with radicular syndrome;
– osteoarthritis;
– myalgia of rheumatic and non-rheumatic genesis;
–
Pain syndrome of different genesis (including postoperative period, trauma, algodysmenorrhea, toothache, headache, arthralgia, lumboishalgia).
The drug is intended for symptomatic therapy, reduction of pain and inflammation at the time of use, does not affect the progression of the disease.
Active ingredient
Nimesulide
Composition
Each tablet contains:
Active ingredient:
nimesulide 100 mg.
Excipients:
calcium hydrophosphate 75 mg,
cellulose microcrystalline (type 114) 40 mg,
corn starch 54 mg,
sodium carboxymethyl starch 35 mg,
magnesium stearate 3 mg,
colloidal silica 2 mg,
talc 1 mg.
How to take, the dosage
Tablets are taken with plenty of water and preferably after meals.
Adults and children over 12 years of age orally take 1 tablet 2 times a day.
In case of gastrointestinal tract diseases it is recommended to take the preparation at the end of a meal or after eating.
Maximal daily dose for adults is 200 mg.
In patients with chronic renal insufficiency the daily dose should be reduced to 100 mg.
Interaction
The effect of drugs that reduce blood clotting is increased when they are used simultaneously with nimesulide.
Nimesulide may decrease the effect of furosemide. Nimesulide may increase the possibility of side effects when concomitant use of methotrexate.
Plasma lithium levels are increased when concomitant use of lithium and nimesulide.
Nimesulide may increase the effect of cyclosporine on the kidneys.
The use with glucocorticosteroids, serotonin reuptake inhibitors increases the risk of gastrointestinal bleeding.
Special Instructions
Because Naize® is partially excreted by the kidneys, its dose should be reduced for patients with impaired renal function, depending on creatinine clearance values. Given the reports of visual disturbances in patients taking other NSAIDs, treatment should be stopped immediately if any visual disturbance occurs and the patient should be examined by an ophthalmologist.
The drug may cause fluid retention in tissues; therefore, patients with high blood pressure and with cardiac abnormalities should use Naiz® with extreme caution.
Patients should be monitored regularly if they also take medications with nimesulide that affect the gastrointestinal tract.
In case of signs of liver damage (skin itching, yellowing of the skin, nausea, vomiting, abdominal pain, darkened urine, increased “liver” transaminases) the drug should be stopped and a physician should be consulted. The drug should not be used simultaneously with other NSAIDs.
The drug may modify platelet function, but does not replace the prophylactic effect of acetylsalicylic acid in cardiovascular diseases. The use of the drug may adversely affect female fertility and is not recommended for women planning to become pregnant.
After 2 weeks of using the drug it is necessary to monitor the biochemical parameters of liver function.
This dosage form is contraindicated in children under 12 years of age, but if it is necessary to use nimesulide in children over 7 years of age, dispersible tablets 50 mg and suspension may be used, in strict compliance with the instructions for medical use that are included with them.
As the drug may cause drowsiness, dizziness, and blurred vision, caution should be exercised when driving a vehicle and engaging in other potentially dangerous activities that require increased concentration and quick psychomotor reactions.
Contraindications
- Hypersensitivity to the active ingredient or excipients;
- .Complete or incomplete combination of bronchial asthma, recurrent nasal or paranasal sinus polyposis and intolerance to acetylsalicylic acid and other NSAIDs (including history);
- gastric and duodenal ulcerative mucosal changes, active gastrointestinal bleeding, cerebrovascular or other bleeding;
- inflammatory bowel disease (Crohn’s disease, non-specific ulcerative colitis) in the acute phase;
- hemophilia and other blood clotting disorders;
- decompensated heart failure;
- hepatic insufficiency or any active liver disease;
- anamnestic evidence of hepatotoxic reactions when using nimesulide preparations;
- co-administration of potentially hepatotoxic agents;
- alcoholism, drug addiction;
- expressed renal insufficiency (creatinine clearance less than 30 ml/min), advanced renal disease, confirmed hyperkalemia;
- period after coronary artery bypass surgery;
- pregnancy, lactation;
- children under 12 years of age.
With caution
Ischemic heart disease, cerebrovascular disease, congestive heart failure, dyslipidemia/hyperlipidemia, diabetes, peripheral artery disease, smoking, creatinine clearance less than 60 ml/min. Anamnestic data about the development of gastrointestinal ulcers, the presence of Helicobacter pylori infection, advanced age, long-term use of NSAIDs, frequent use of alcohol, severe somatic diseases, concomitant therapy with anticoagulants (e.g, Warfarin), antiplatelet agents (e.g., acetylsalicylic acid, clopidogrel), oral glucocorticosteroids (e.g., prednisolone), selective serotonin reuptake inhibitors (e.g., citalopram, fluoxetine, paroxetine, sertraline).
Side effects
The frequency of adverse effects is classified according to the frequency of occurrence of the case: frequently (1-10%), sometimes (0.1-1%), rarely (0.01-0.1%), very rarely (less than 0.01%), including individual reports.
Allergic reactions: rare – hypersensitivity reactions; very rare – anaphylactoid reactions.
The central nervous system: infrequent – dizziness; rare – feeling of fear, nervousness, nightmares; very rare – headache, somnolence, encephalopathy (Reis syndrome).
Skin disorders: infrequent itching, rash, increased sweating; rare erythema, dermatitis; very rare: urticaria, angioedema, facial edema, erythema multiforme, including Stevens-Johnson syndrome, toxic epidermal necrolysis (Lyell’s syndrome).
Arenal system: infrequent – edema; rare – dysuria, hematuria, urinary retention, hyperkalemia; very rare – renal failure, oliguria, interstitial nephritis.
Gastro-intestinal tract: frequently – diarrhea, nausea, vomiting; infrequently – constipation, flatulence, gastritis; very rarely – abdominal pain, stomatitis, tar stool, gastrointestinal bleeding, peptic ulcer and/or perforation of the stomach or duodenum.
Hepatic and biliary system disorders: often – increase of “hepatic” transaminases; very rarely – hepatitis, lightning hepatitis, jaundice, cholestasis.
Hematopoietic organs: rarely – anemia, eosinophilia; very rarely – thrombocytopenia, pancytopenia, purpura, prolongation of bleeding time.
Respiratory system: infrequent – shortness of breath; very rare – exacerbation of bronchial asthma, bronchospasm.
Sensory system: infrequent – blurred vision.
Cardiovascular system: infrequent – arterial hypertension; rare – tachycardia, hemorrhages, “hot flashes”. Other: rarely – general weakness; very rarely – hypothermia.
Overdose
Symptoms: apathy, drowsiness, nausea, vomiting. Gastrointestinal bleeding, increased blood pressure, acute renal failure, respiratory depression may occur.
Treatment: symptomatic treatment and supportive care of the patient is required. There is no specific antidote. If overdose occurred within the last 4 hours it is necessary to induce vomiting, provide intake of activated charcoal (60-100 g per adult), osmotic laxatives. Forced diuresis, hemodialysis are ineffective due to high protein binding of the drug.
Similarities
Naiz, Nimesil, Nimesulide, Nemulex, Nimesulide-TEVA, Nimulide, Nimesan tablets 100 mg 20 pcs, Naisulide, Mialais
| Weight | 0.013 kg |
|---|---|
| Shelf life | 3 years |
| Conditions of storage | List B. In dry, dark place at a temperature not exceeding 25 ° C. Keep out of reach of children! |
| Manufacturer | Dr. Reddy's, India |
| Medication form | pills |
| Brand | Dr. Reddy's |
Other forms…
Related products
Buy Nais, tablets 100 mg 20 pcs with delivery to USA, UK, Europe and over 120 other countries.