No products in the cart.
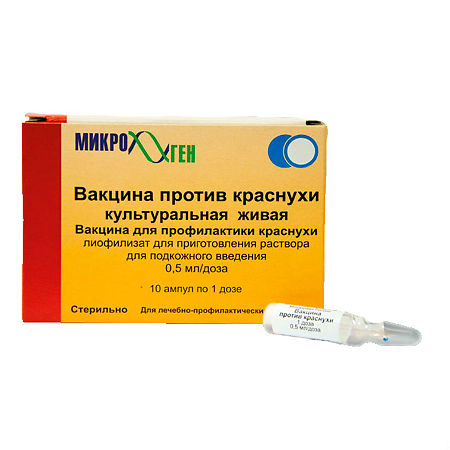
Live rubella culture vaccine, 0.5 ml/dose
€1.00
Out of stock
(E-mail when Stock is available)
Description
A live attenuated vaccine for the prevention of rubella. The attenuated vaccine virus (strain Wistar RA 27/3M) is cultured on human diploid cells.
Specific immunity develops within 15 days after vaccination and is reported to last for at least 20 years.
Indications
Indications
Active ingredient
Active ingredient
Composition
Composition
How to take, the dosage
How to take, the dosage
Interaction
Interaction
Vaccines
The rubella vaccine can be administered simultaneously (on the same day) with vaccines against pertussis, diphtheria, tetanus, polio (live and inactivated), hepatitis B, measles, epidemic parotitis in different syringes or one month after the previous vaccination.
Immunoglobulins and blood products
After the introduction of blood products (immunoglobulin, plasma, etc.) it is recommended to administer the vaccine not earlier than 3 months. After administration of rubella vaccine blood products can be administered not earlier than in 2 weeks. If immunoglobulin has to be given earlier than this, the rubella vaccination should be repeated after 3 months.
In the presence of rubella antibodies in the serum, vaccination should not be repeated.
Tuberculin
Tuberculin tests are recommended before or 4-6 weeks after rubella vaccine administration.
Special Instructions
Special Instructions
Women of childbearing age should use effective contraception for at least 2 months after vaccination.
The use of this vaccine in women in the early stages of a pregnancy not yet established is not an indication for termination of that pregnancy.
The decision to vaccinate a baby born of an HIV-infected mother, and the determination of the child’s immune status, will be delayed until the baby is 9-10 months old, since maternal IgG which has penetrated the placenta can persist in the blood up to 14 months of age. If the child is found to be HIV-infected after this age, a panel of doctors will decide whether or not to vaccinate the child. If the child is found to be uninfected, a regular vaccination is administered.
The rubella (live) vaccine can be given along with other vaccines. The most widely used combination vaccine is for the prevention of measles, mumps and rubella.
Due to possible inactivation of the vaccine strain by serum antibodies, vaccination is not carried out for 6 weeks (if possible, for 3 months) after administration of immunoglobulins or other blood products, and also after the vaccine administration no immunoglobulins are used for 2 weeks.
The tuberculin test may be false-negative for some time after vaccination.
Contraindications
Contraindications
Note:
HIV-infected children may be immunized with rubella vaccine.
Side effects
Side effects
Additional information
| Weight | 0.040 kg |
|---|---|
| Shelf life | 2 years |
| Conditions of storage | The vaccine is stored and transported at +2 ° C to +8 ° C in a place protected from light. Freezing is not allowed. |
| Manufacturer | Microgen NPO, Russia |
| Medication form | solution for injection |
| Brand | Microgen NPO |
Related products
Buy Live rubella culture vaccine, 0.5 ml/dose with delivery to USA, UK, Europe and over 120 other countries.