No products in the cart.
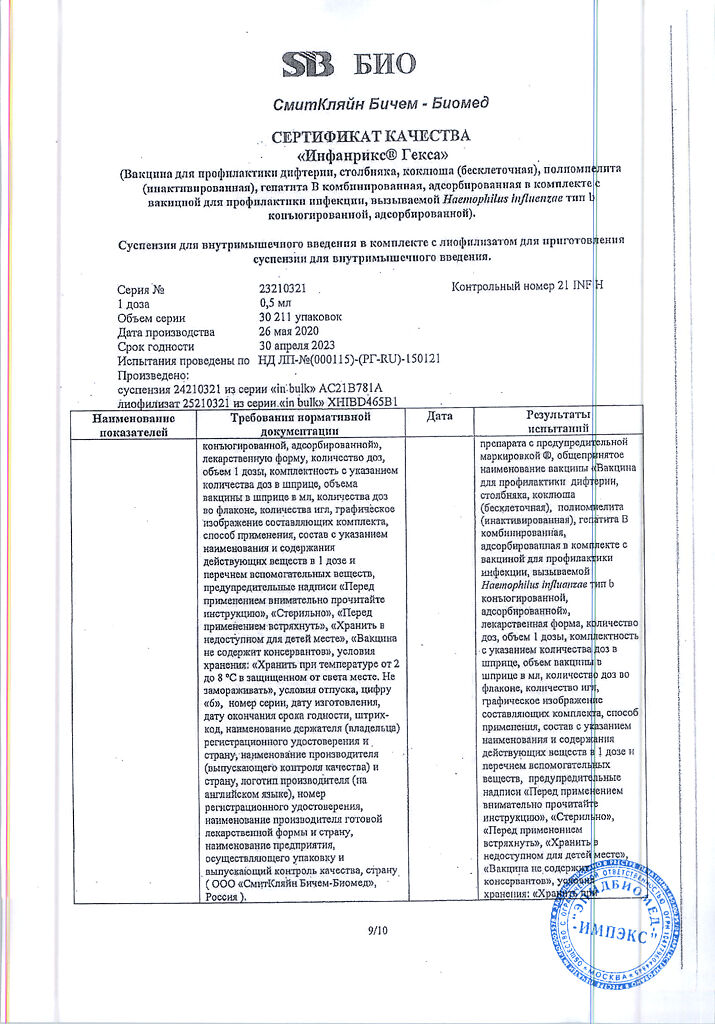
Infanrix Hexa, 0.5 ml syringe
€1.00
Out of stock
(E-mail when Stock is available)
Description
Infantrix complies with WHO requirements for the production of biologics and vaccines against diphtheria, tetanus and pertussis.
One month after the three-dose course of initial Infantrix vaccination in the first months of life, more than 99% of children immunized with Infantrix vaccine have antibody titers to diphtheria and tetanus anatoxins greater than 0.1 IU/ml.
Antibodies to pertussis antigens (CA, FHA and pertactin) are produced in more than 95% of vaccinated children.
After revaccination with Infantrix vaccine in the second year of life (13 to 24 months), all children who were initially immunized with Infantrix vaccine have antibody titers of more than 0.1 IU/ml for diphtheria and tetanus toxoids.
The secondary immune response to pertussis antigens occurs in more than 96% of children.
The protective efficacy of the Infanrix vaccine reaches an average of 88%.
Indications
Indications
- Primary vaccination against diphtheria, tetanus and pertussis in children from 3 months.
- Revaccination of children who were previously immunized with three doses of cell-free pertussis-diphtheria-tetanus vaccine or whole cell pertussis-diphtheria-tetanus vaccine.
- At the start of the pertussis-diphtheria-tetanus cell-free vaccine course, subsequent doses of pertussis-diphtheria-tetanus cell-free vaccine may be administered and vice versa.
How to take, the dosage
How to take, the dosage
The primary vaccination course consists of three vaccine doses administered according to the Russian National Preventive Vaccination Calendar at 3, 4, 5 and 6 months of age; revaccination is carried out at 18 months of age.
Before vaccination the vaccine is shaken well until a homogeneous turbid suspension is formed and examined carefully. If foreign particles, unbreakable flakes or changes in appearance are found, the vaccine should not be used.
Infantrix vaccine should be given by intramuscular injection and alternate sites during the course of the vaccination.
The vaccine should never be given intravenously!
Interaction
Interaction
To provide appropriate recommendations, there is insufficient data regarding vaccine efficacy and safety to administer the Infantrix Hexa vaccine at the same time as the measles/mumps/rubella vaccine.
Special Instructions
Special Instructions
Caution should be exercised when administering the vaccine to persons with thrombocytopenia or disorders of hematopoietic function, since in such patients there is a risk of bleeding after the IV/m injection.
Contraindications
Contraindications
- Knowledgeable hypersensitivity to any component of this vaccine, or if the patient has had hypersensitivity symptoms since the previous Infantrix administration.
- Severe reaction (fever above 40°C, hyperemia or swelling greater than 8 cm in diameter) or complication (collapse or shock-like condition developed within 48 hours of vaccine administration; Continuous crying for 3 hours or more, occurring within 48 hours of vaccination; convulsions, with or without fever, occurring within 3 days of vaccination) on a previous Infantrix vaccine administration.
- Encephalopathy developed within 7 days after the previous administration of a vaccine containing the pertussis component. In this case, the vaccine should be continued with diphtheria-tetanus vaccine.
.
Side effects
Side effects
Clinical Studies
The following composite of safety parameters is based on data from immunizations in more than 16,000 patients.
As with the AACDS vaccine and combination vaccines that contain AACDS, an increased incidence of local reactogenicity and fever after booster vaccination with Infantrix Hexa has been reported compared to the initial course.
Additional information
| Weight | 0.027 kg |
|---|---|
| Shelf life | 3 years |
| Conditions of storage | At 2-8 °C (do not freeze) |
| Manufacturer | GlaxoSmithKline Biologics, France |
| Medication form | suspension |
| Brand | GlaxoSmithKline Biologics |
Related products
Buy Infanrix Hexa, 0.5 ml syringe with delivery to USA, UK, Europe and over 120 other countries.