No products in the cart.
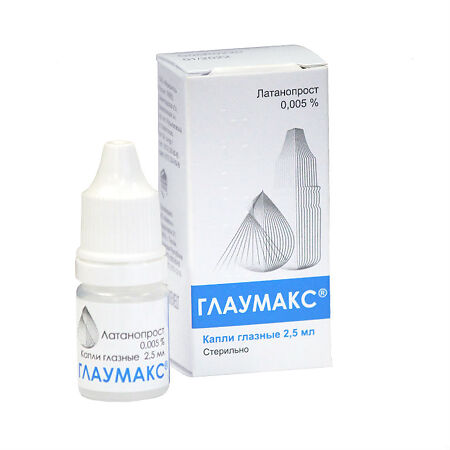
Glaumax, eye drops 0.005% 2.5ml
€21.24 €17.70
Description
Pharmacotherapeutic group: prostaglandin F2αanalog synthetic
ATC code: S01EE01
PHARMACOLOGICAL PROPERTIES
Pharmacodynamics:
Latanoprost, a prostaglandin F2a analog, is a selective FP (prostaglandin F) receptor agonist and reduces intraocular pressure (IOP) by increasing aqueous humor outflow. Decrease of IOP starts approximately 3-4 hours after the drug administration, the maximum effect is observed after 8-12 hours, the action lasts for at least 24 hours.
Studies in animals and humans have shown that the main mechanism of action is the increase of uveoscleral outflow, besides, in humans the outflow is also described as improved (decrease of outflow resistance).
Latanoprost was found to have no significant effect on aqueous humor production or on the blood-ophthalmic barrier. Animal studies have shown that in clinical doses latanoprost has no effect (or insignificant effect) on intraocular blood flow.
Mild to moderate conjunctival or episcleral injection is possible when used topically.
According to fluorescence angiography data, long-term treatment with latanoprost after extracapsular cataract extraction in monkeys had no effect on retinal blood flow.
With short-term use, latanoprost did not promote fluorescein infiltration in the posterior segment of the eyes of patients with an artificial lens.
Latanoprost has no significant pharmacological effects on the cardiovascular and respiratory systems when used at therapeutic doses.
Pharmacokinetics:
Latanoprost (molecular weight 432.58) is a prodrug esterified with isopropyl group, inactive; after hydrolysis to acidic form it becomes biologically active.
Absorption
The prodrug is well absorbed through the cornea and is fully hydrolyzed when exposed to aqueous moisture.
Distribution
Studies in humans have shown that the maximum concentration in aqueous humor is reached 2 hours after instillation.
After instillation in monkeys, latanoprost is distributed predominantly in the anterior chamber of the eye, conjunctiva and eyelids. Only a small amount of latanoprost reaches the posterior chamber of the eye.
Biotransformation
The active form of latanoprost is virtually unmetabolized in the eye, but is biotransformed in the liver.
Elimination
The plasma elimination half-life is 17 minutes.
The animal studies have shown that the major metabolites (1,2-dinor- and 1,2,3,4-tetranormetabolites) have no (or low) biological activity and are excreted mainly in the urine.
Children
Pharmacokinetic studies of latanoprost were performed in 22 adults and 25 children (ages 0-18 years) with ophthalmohypertension and glaucoma. All age groups received latanoprost at a concentration of 0.005%, one drop in each eye for at least 2 weeks. Exposure to latanoprost was approximately 2-fold higher in children aged 3 to 12 years compared to adult patients and 6-fold higher in children younger than 3 years. However, the safety profile of the drug does not differ between children and adults (see section “Overdose”). In all age groups, the duration of maintenance of the maximum plasma concentration of latanoprost acid is 5 min. The half-life of latanoprost acid in children is the same as in adults (<20 min). In equilibrium concentration there is no cumulation of lathanoprost acid in blood plasma.
Indications
Indications
Glaucoma, Elevated Intraocular PressureReducing elevated intraocular pressure (IOP) in adults and children (over 1 year of age) with open-angle glaucoma or ophthalmic hypertension.
Active ingredient
Active ingredient
Latanoprost
Composition
Composition
1 ml of the solution contains:
The active ingredient:
Latanoprost – 50 mcg
Excipients:
benzalkonium chloride – 0.2 mg,
sodium chloride – 5.69 mg,
sodium dihydrophosphate dihydrate – 3.98 mg, <
Sodium hydrophosphate anhydrous – 3.48 mg,
water for injection – up to 1.0 ml.
.
How to take, the dosage
How to take, the dosage
Dosing regimen in adults (including elderly)
Please put one drop in the affected eye(s) once a day. The optimal effect is achieved when the drug is used in the evening.
The drug should not be instilled more frequently than once a day because it has been shown that more frequent instillation reduces the hypotensive effect.
If a dose is missed, treatment continues as usual.
As with any eye drops, in order to reduce the possible systemic effect of the drug, immediately after instillation of each drop it is recommended that pressure be applied for 1 minute to the lower lacrimal point located at the inner corner of the eye on the lower eyelid. This procedure should be done immediately after instillation.
Please remove contact lenses before the instillation and do not insert them until 15 minutes after the instillation (see also the section on “Special considerations”).
If several eye dosage forms are used at the same time, they should be separated by 5 minute intervals.
Dosing regimen in children
Latanoprost is used in children in the same dose as in adults. There are no data on the use of the drug in preterm infants (gestational age <36 weeks). Data in children <1 year old are very limited.
Interaction
Interaction
Unambiguous data on drug interactions of latanoprost are not available.
The simultaneous instillation of two prostaglandin analogues into the eyes has described a paradoxical increase in IOP, so the simultaneous use of two or more prostaglandins, their analogues or derivatives is not recommended.
In in vitro studies, it has been shown that precipitate is formed when eye drops containing thiomersal are mixed with eye drops containing latanoprost. A 5-minute interval between instillations should be observed if simultaneous use of these medications is necessary.
Special Instructions
Special Instructions
Latanoprost may gradually change eye color by increasing the brown pigment content of the iris. Before treatment patients should be informed about a possible irreversible change in eye color. Use of the drug on one eye may cause irreversible heterochromia.
This change in eye color has mainly been observed in patients with unevenly colored irises, namely: brown-blue, gray-brown, yellow-brown and green-brown. In the latanoprost studies, darkening generally began during the first 8 months of treatment, rarely during the second or third year, and was not noted after four years of treatment. Progression of iris pigmentation decreased over time and stabilized after 5 years. There was no evidence of increased pigmentation at 5 years. In an open 5-year safety study of latanoprost, iris pigmentation developed in 33% of patients (see side effects section).
In most cases, iris color changes were minor and often were not clinically detectable. The incidence ranged from 7% to 85% in patients with unequal iris color, predominating in patients with yellow-brown irises. No changes were observed in patients with uniformly colored blue irises, in rare cases changes were observed in uniformly colored gray, green, and brown irises.
Eye color changes are due to increased melanin content in stromal iris melanocytes, not to increased number of melanocytes themselves. In typical cases, brown pigmentation appears around the pupil and spreads concentrically to the periphery of the iris. The entire iris or its parts turn brown. No further pigmentation has been observed after withdrawal of therapy. According to the available clinical data the color change was not associated with any symptoms or pathological disorders.
The drug has no effect on iris nevi and lentigines. According to the results of 5-year clinical studies, accumulation of pigment in the sclero-corneal trabecular network or other parts of the anterior chamber of the eye was not observed. Iris darkening has not been shown to cause undesirable clinical consequences, so the use of latanoprost can be continued if such darkening occurs. Nevertheless, such patients should be monitored regularly and, depending on the clinical situation, treatment may be discontinued.
Experience with latanoprost in the therapy of closed angle and congenital glaucoma, pigmentary glaucoma, open angle glaucoma in patients with pseudoaphakia is limited.
There is no data on the use of latanoprost in the treatment of secondary glaucoma due to inflammatory eye disease and neovascular glaucoma.
Latanoprost has no effect on pupil size. Due to the lack of experience with latanoprost in the treatment of acute attacks of closed angle glaucoma, caution should be exercised when using the drug in these patients.
Due to the fact that data on the use of latanoprost in the postoperative period of cataract extraction are limited, caution should be exercised when using the drug in this category of patients.
Caution should be exercised when using latanoprost in patients with a history of herpetic keratitis. In acute herpetic keratitis, and if there is a history of chronic recurrent herpetic keratitis, administration of latanoprost should be avoided.
Macular edema including cystic edema has been observed during latanoprost therapy mainly in patients with aphakia, pseudoaphakia, rupture of the posterior lens capsule or in patients with risk factors of cystic macular edema development (in particular in diabetic retinopathy and retinal vein occlusion). Caution should be exercised when using latanoprost in patients with aphakia, pseudoaphakia with posterior capsule rupture or anterior chamber intraocular lenses, as well as in patients with known risk factors for cystic macular edema.
Caution should be exercised when using latanoprost in patients with risk factors for iritis/veitis.
Experience with latanoprost in patients with bronchial asthma is limited, but in some cases exacerbation of asthma and/or dyspnea have been reported in the post-registration period. Caution should be exercised when using latanoprost in this category of patients (see also section “Side effects”). There have been cases of periorbital skin darkening, which in some patients has been reversible with continuation of latanoprost therapy.
Latanoprost may cause gradual changes in eyelashes and downy hair such as lengthening, thickening, increasing pigmentation, increasing density and changing direction of lash growth. Eyelash changes were reversible and went away after therapy was discontinued.
Latanoprost contains benzalkonium chloride, often used as a preservative in ophthalmic medications. Benzalkonium chloride can cause ocular irritation, pitting and/or toxic ulcerative keratopathy, and can be absorbed by soft contact lenses and discolor them. Careful monitoring of patients with dry eye syndrome or other corneal diseases during long-term use of latanoprost is required. Contact lenses shall be removed before the drug application and inserted again not earlier than 15 min after instillation (see also section “Dosage and administration”).
Children
The data on efficiency and safety of latanoprost usage in children under one year old are limited. There is no experience of the drug administration in premature children (gestational age less than 36 weeks).
There are no data on safety of long-term use of latanoprost in children.
In primary congenital glaucoma in children from 0 to 3 years old surgical intervention (goniotomy/trabeculotomy) remains a standard method of treatment.
Effects on the ability to drive vehicles and operate machinery
As with other ophthalmic drugs, temporary visual impairment may occur; it is not recommended to drive vehicles or operate machinery until its recovery.
Synopsis
Synopsis
Clear, colorless, odorless liquid
Contraindications
Contraindications
– Hypersensitivity to latanoprost or other components of the drug. Age under 1 year (efficacy and safety have not been established).
– Pregnancy and breastfeeding (see section “Use in pregnancy and breastfeeding”).
WARNING:
Glaumax® should be used with caution in patients with aphakia, pseudoaphakia, damage to the posterior lens capsule, and known risk factors for macular edema (cases of macular edema, including cystic edema, have been described with treatment with latanoprost).
There are currently no data on the use of Glaumax® in inflammatory eye diseases, inflammatory, neovascular, closed-angle and congenital glaucoma. There is little experience of using the drug in open-angle glaucoma in patients with artifactia as well as in pigmentary glaucoma.
Glaumax® has no or little effect on the pupil, but there is no data on the drug action in an acute attack of closed-angle glaucoma. In these conditions, the use of Glaumax® is recommended with caution.
Side effects
Side effects
Most adverse reactions have been reported in the visual organ. In an open 5-year safety study, 33% developed iris pigmentation (see section “Special Precautions”). Other adverse reactions on the visual organs are usually transient and occur immediately after instillation.
Gradation of adverse reactions by frequency of occurrence was performed as follows: Very common (>1/10); common (≥1/100, <1/10), infrequent (≥1/1000, <1/100), rare (≥1/10 000, <1/1000), and very rare (<1/10 000), frequency unknown (frequency cannot be estimated based on available data).
Infections and invasions: Frequency unknown: herpetic keratitis.
Visually:
- very common: iris hyperpigmentation, conjunctival hyperemia, mild to moderate eye irritation (burning sensation, sensation of sand in eyes, itching, tingling and sensation of foreign body), changes in eyelashes (increased length, thickness, number and pigmentation).
- Frequently: transient pitting erosions of epithelium (mostly asymptomatic), blepharitis, pain in the eye.
- Infrequent: swelling of eyelids, dry mucous membrane of the eye, keratitis, blurred vision, conjunctivitis.
- Rarely: iritis/veitis (mainly in predisposed patients), macular edema, eyelid edema, corneal edema, corneal erosion, periorbital edema, darkening of eyelid skin, skin reactions, changes in lash growth direction, thickening, darkening and lengthening of lashes, distichiasis, photophobia.
- Very rare: changes in the periorbital and eyelash area leading to deepening of the upper eyelid sulcus.
- Frequency unknown: iris cyst.
- In some patients with significant corneal damage, very rare cases of corneal calcification have been reported due to the use of phosphate-containing eye drops.
Nervous system disorders: Frequency unknown: dizziness, headache.
Cardiac disorders:
- Very rare: worsening the course of angina in patients with concomitant angina pectoris.
- Frequency unknown: palpitations.
Respiratory: Rarely: bronchospasm (including. exacerbation in patients with bronchial asthma in anamnesis), shortness of breath.
Skin and subcutaneous tissue:
- Infrequent: rash.
- Rarely: darkening of eyelid skin and local cutaneous reactions on eyelids.
Musculoskeletal and connective tissue: Frequency unknown: myalgia, arthralgia.
General disorders and local reactions: Very rare: Chest pain.
Children
According to two short-term (≤12 weeks) clinical studies in 93 children, the safety profile of latanoprost in children did not differ from that of adults. The safety profile between different age groups in children is comparable. Compared to the adult population, nasopharyngitis and fever were most common in children.
Overdose
Overdose
In addition to ocular mucosal irritation and hyperemia, no other adverse reactions from the visual organ have been described in case of latanoprost overdose.
The following information should be considered when latanoprost is accidentally ingested: one bottle of 2.5 ml solution contains 125 mcg of latanoprost. More than 90% of latanoprost is metabolized during “first passage” through the liver. Intravenous infusion at a dose of 3 µg/kg in healthy volunteers caused no symptoms, but nausea, abdominal pain, dizziness, fatigue, “hot flashes” and sweating were observed when a dose of 5.5-10 µg/kg was administered. Intravenous administration of latanoprost to monkeys at a dose of 500 mcg/kg did not cause significant effects on the cardiovascular system.
Intravenous administration of latanoprost to monkeys caused transient bronchospasm. In patients with bronchial asthma of moderate severity the instillation of latanoprost into the eyes at a dose of 7 times therapeutic did not cause bronchospasm. In case of overdose symptomatic treatment is carried out.
Pregnancy use
Pregnancy use
Pregnancy
The safety of latanoprost use during pregnancy in humans has not been established. Latanoprost may have toxic effects on pregnancy, fetus and newborn. Use during pregnancy is contraindicated.
Breastfeeding period
Latanoprost and its metabolites may penetrate into breast milk. It is contraindicated during the breastfeeding period. If it is necessary to use the drug, breastfeeding should be stopped.
Fertility
No effect of latanoprost on male and female fertility was found in animal studies.
Similarities
Similarities
Xalatan, Xalatamax, Glauprost, Prolatan, Prolatan eye drops, Trilactan, Latanoprost-Optic
Additional information
| Weight | 0.015 kg |
|---|---|
| Shelf life | 2 years. After opening the bottle the drug should be used within 4 weeks. Do not use after the expiration date shown on the package. |
| Conditions of storage | Store in a dark place at temperatures from 2 to 8 ° C. Store the opened bottle at a temperature not exceeding 25 ° C. Keep out of reach of children. |
| Manufacturer | Kevelt AS, Estonia |
| Medication form | eye drops |
| Brand | Kevelt AS |
Related products
Buy Glaumax, eye drops 0.005% 2.5ml with delivery to USA, UK, Europe and over 120 other countries.