No products in the cart.
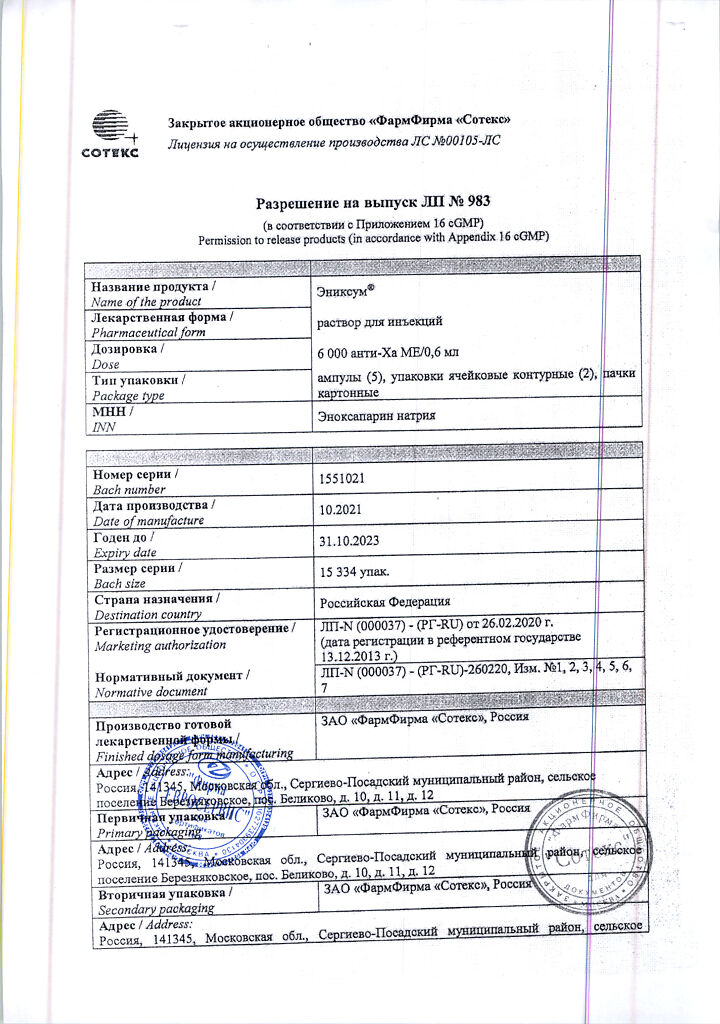
Enixum, 6000 anti-ha me/0.6 mL 0.6 mL 10 pcs
€1.00
Out of stock
(E-mail when Stock is available)
Description
Pharmacotherapeutic group: direct acting anticoagulant.
ATC code: B01AB05
Pharmacological properties
Characteristics
Enoxaparin sodium is a low molecular weight heparin. The average molecular weight is about
4500 daltons: less than 2000 daltons – < 20%, 2000 to 8000 daltons – > 68%, more than 8000 daltons – < 18%. Enoxaparin sodium is obtained by alkaline hydrolysis of benzyl ether
heparin, isolated from the mucous membrane of the small intestine of pigs. Its structure is characterized by a nonreducible fragment of 2-O-sulfo-4-enpyrazinosuronic
acid and a reducing fragment of 2-N,6-O-disulfo-D-glucopyranoside.
The structure of sodium enoxaparin contains about 20% (ranging from 15% to 25%) of 1,6-anhydro derivative in the reducing fragment of polysaccharide chain.
Pharmacodynamics
In vitro enoxaparin sodium has high activity against factor Xa clotting (anti-Xa activity of approximately 100 IU/ml) and low activity against
factor IIa clotting (anti-IIa or antithrombin activity of approximately 28 IU/ml).
This anticoagulant activity is mediated by antithrombin III (AT-III). In addition to anti-Xa/IIa
activity, additional anticoagulant and anti-inflammatory
properties of enoxaparin sodium in both human and animal models have also been identified, which include AT-III dependent inhibition of other clotting factors such as factor
VIIa, activation of tissue factor pathway inhibitor release, and reduction of Willebrand factor release from the vascular endothelium into the bloodstream. These factors
provide anticoagulant effect of enoxaparin sodium in general.
When used in prophylactic doses enoxaparin sodium slightly changes activated partial thromboplastin time (PPT), has almost no
effect on platelet aggregation and on the degree of binding of fibrinogen to platelet receptors.
Anti-IIa activity in plasma is about 10 times lower than anti-Xa activity. The average maximum anti-IIa activity is observed approximately 3-4 hours after subcutaneous
administration and reaches 0.13 IU/ml and 0.19 IU/ml after repeated administration of 1 mg/kg body weight – when administered twice and 1.5 mg/kg body weight – when administered once,
respectively.
Mean maximum plasma anti-Xa activity is observed 3-5 hours after subcutaneous administration of the drug and is approximately 0.2; 0.4; 1.0 and 1.3 anti-Xa IU/ml after
subcutaneous administration of 20 mg, 40 mg and 1 mg/kg and 1.5 mg/kg, respectively.
Pharmacokinetics
The pharmacokinetics of enoxaparin sodium at therapeutic doses are linear. Variability within and between patient groups is low. After a single subcutaneous administration of Enixum® at the dose of 1 mg/kg Cmax is 0.49±0.07 IU/ml, Òmax is 3.19+1.08 h, AUC0-24 = 4.44+0.91 IU×ml/hour. According to the literature data,
after repeated subcutaneous administration of Enoxaparin sodium at a dose of 40 mg once a day and after subcutaneous administration of Enoxaparin sodium at a dose of 1.5 mg/kg once a day the equilibrium concentration is reached by the 2nd day, and the area under the pharmacokinetic curve is on average 15% higher than after a single injection. After repeated subcutaneous
injections of enoxaparin sodium at a daily dose of 1 mg/kg twice a day the equilibrium concentration is reached after 3-4 days, and the area under the pharmacokinetic curve is on
average 65 % higher than after a single injection. Bioavailability of enoxaparin sodium at subcutaneous administration, estimated on the basis of anti-Xa activity, is close to 100%. The volume of distribution of anti-Xa activity of sodium enoxaparin is approximately 5 liters and approximates the blood volume.
Sodium enoxaparin is a drug with low clearance. After IV administration for 6 h at a dose of 1.5 mg/kg body weight, the average anti-Xa clearance in plasma was
0.74 L/h.
Enoxaparin sodium is mainly metabolized in the liver by desulfation and/or depolymerization to form low molecular weight substances with very low biological activity.
Patients in the elderly
Excretion is delayed due to physiological decreases in renal function. This change does not affect the dosing and administration regimen for prophylactic therapy as long as the
renal function of such patients remains within acceptable limits, i.e., is mildly impaired.
Patients with impaired renal function
The clearance of enoxaparin sodium is reduced in patients with decreased renal function.
Decreased clearance of enoxaparin sodium has been noted in renal failure. After repeated subcutaneous administration of 40 mg of sodium enoxaparin once a day there is an increase in anti-Xa activity represented by the area under in mild (creatinine clearance (CK) 50-80 ml/min) and moderate (CK 30-50 ml/min) renal insufficiency. In patients with severe renal insufficiency (CKR less than 30 ml/min) the area under pharmacokinetic curve at equilibrium is, on average, 65% higher with repeated subcutaneous administration of 40 mg of the drug once daily.
Patients with excess body weight
People with excess body weight have slightly lower clearance when the drug is administered subcutaneously. After a single subcutaneous injection of 40 mg of Enoxaparin sodium, if no dose adjustments are made for patient weight, the anti-Xa activity will be 50% higher in women with a body mass below 45 kg and 27% higher in men with a body mass below 57 kg compared to patients of normal average body mass.
Indications
Indications
Prevention of venous thrombosis and embolism in surgical interventions, especially in orthopedic and general surgery;
– The prophylaxis of venous thromboses and embolisms in bed-ridden patients due to acute therapeutic diseases (including acute heart failure and decompensation of chronic heart failure (III or IV class NYHA), acute respiratory failure; acute infectious diseases; acute stages of rheumatic diseases in combination with one of the risk factors of venous thrombosis (see “Special indications”). “Special indications”);
– treatment of deep vein thrombosis, which is accompanied or not accompanied by pulmonary embolism;
– treatment of unstable angina and myocardial infarction without Q-wave in combination with acetylsalicylic acid;
– Prevention of thrombosis in extracorporeal circulation system during hemodialysis (usually with the duration of the session not more than 4 hours);
– Treatment of acute myocardial infarction with ST-segment elevation in patients undergoing medical treatment or subsequent percutaneous coronary intervention.
Active ingredient
Active ingredient
Enoxaparin sodium
Composition
Composition
Active ingredient: Enoxaparin sodium 6000 anti-Ha IU (60 mg); excipients: water for injection – up to 0.6 ml
How to take, the dosage
How to take, the dosage
Except in special cases (see “Treatment of ST-segment elevation myocardial infarction, medication-assisted or percutaneous coronary intervention” and “Prevention of thrombosis in the extracorporeal circulatory system during hemodialysis” below), sodium enoxaparin is injected deep subcutaneously. The injections should preferably be given in the supine position. Injections should be given alternately on the left or right anterolateral or posterolateral surface of the abdomen.
The needle must be inserted vertically (not laterally) into the full length skin fold, gathered and held between the thumb and forefinger until the injection is complete.
The pre-filled disposable syringe is ready for use.
The product should not be injected intramuscularly!
Prevention of venous thrombosis and embolism in surgical procedures, especially orthopedic and general surgical procedures
Prevention of venous thrombosis and embolism in surgical procedures.
In patients at moderate risk of thrombosis and embolism (e.g., abdominal surgeries), the recommended dose is 20 mg once daily subcutaneously. The first injection should be given 2 hours before surgery.
. Patients at high risk of thrombosis and embolism (e.g., orthopedic surgery, oncologic surgery, patients with additional non-surgical risk factors such as congenital or acquired thrombophilia, malignant neoplasia, bed rest of >3 days (obesity, history of venous thrombosis, varicose veins of the lower limbs, pregnancy), the drug is recommended in a dose of 40 mg once daily subcutaneously, with the first dose given 12 hours before surgery, or in a dose of 30 mg twice daily, starting 12-24 hours after surgery.
The duration of treatment with the drug is on average 7-10 days. If necessary, therapy may be continued as long as the risk of thrombosis and embolism persists and as long as the patient remains ambulatory.
In orthopedic surgeries, it may be appropriate to continue treatment after initial therapy with a dose of 40 mg once daily for 3 weeks.
The peculiarities of using the drug in spinal/epidural anesthesia as well as in coronary revascularization procedures are described in section “Special Precautions”.
Prevention of venous thrombosis and embolism in bedridden patients due to acute therapeutic conditions.
The recommended dose of enoxaparin sodium is 40 mg once daily subcutaneously for at least 6 days. The therapy should be continued until the patient is fully ambulatory (maximum of 14 days).
Treatment of deep vein thrombosis with or without throm- pulmonary embolism
Enixum® is administered subcutaneously at a rate of 1.5 mg/kg body weight once daily or at a dose of 1 mg/kg body weight twice daily. In patients with complicated thromboembolic disorders the drug is recommended in a dose of 1 mg/kg twice daily.
The duration of treatment is on average 10 days. Indirect anticoagulant therapy should be initiated immediately, and therapy with enoxaparin sodium should be continued until therapeutic anticoagulant effect is achieved (INR [International Normalized Ratio] values should be 2.0-3.0). If necessary, control of anticoagulant effect should be assessed by anti-Xa activity. Treatment of unstable angina and myocardial infarction without Q-wave in combination with acetylsalicylic acid
The treatment of unstable angina and myocardial infarction without Q-waveacetylsalicylic acid. Enixum® is given at a rate of 1 mg/kg body weight every 12 hours, subcutaneously, with a concomitant oral dose of 100-325 mg of acetylsalicylic acid once daily.
The average duration of treatment is at least 2 days and continues until the patient’s clinical condition stabilizes. It usually lasts from 2 to 8 days.
Treatment of ST-segment elevation myocardial infarction, medication-assisted or with coronary intervention
. Treatment begins with an intravenous bolus injection of enoxaparin sodium at a dose of 30 mg and immediately thereafter (within 15 minutes) administered subcutaneously at a dose of 1 mg/kg body weight (with a maximum of 100 mg of enoxaparin sodium in the first two subcutaneous injections). Thereafter, all subsequent subcutaneous doses are administered every 12 hours at a rate of 1 mg/kg of body weight (i.e., if body weight exceeds 100 kg, the dose may exceed 100 mg).
In patients 75 years of age and older, an initial intravenous bolus injection is not used. Enoxaparin sodium is administered subcutaneously at a dose of 0.75 mg/kg every 12 hours (with a maximum of 75 mg of enoxaparin sodium being administered for the first two subcutaneous injections). Thereafter, all subsequent subcutaneous doses are administered every 12 hours at a rate of 0.75 mg/kg body weight (i.e., if the body weight exceeds 100 kg, the dose may exceed 75 mg). When combined with thrombolytics (fibrin-specific and fibrin-unspecific), enoxaparin sodium should be administered between 15 minutes before thrombolytic therapy and 30 minutes after it. After detection of acute ST-segment elevation myocardial infarction, concomitant administration of acetylsalicylic acid should be started as soon as possible and should be continued for at least 30 days at doses of 75 to 325 mg daily, if there are no contraindications.
The recommended duration of treatment with the drug is 8 days or until the patient is discharged from the hospital if the period of hospitalization is less than 8 days.
The intravenous bolus infusion of sodium enoxaparin should be given through a venous catheter and sodium enoxaparin should not be mixed or coadministered with other medications. In order to avoid traces of other drugs in the system and their interaction with sodium enoxaparin, the venous catheter should be flushed with sufficient amounts of 0.9% sodium chloride solution or 5% dextrose solution before and after intravenous bolus injection of sodium enoxaparin. Enoxaparin sodium is compatible with 0.9% sodium chloride solution and 5% dextrose solution.
For 30 mg bolus infusion of enoxaparin sodium in the treatment of acute ST-segment elevation myocardial infarction, the 60 mg, 80 mg and 100 mg glass syringes are discarded so that only 30 mg (0.3 ml) remains. The 30 mg dose can be given directly intravenously.
Pre-filled 60 mg, 80 mg and 100 mg hypodermic syringes may be used for intravenous bolus injection of enoxaparin sodium via a venous catheter. The use of 60 mg syringes is recommended because it reduces the amount of product removed from the syringe. The 20 mg syringe is not used because it does not contain enough product for a 30 mg bolus of Enoxaparin sodium. The 40 mg syringe is not used because it has no graduations and therefore cannot accurately measure 30 mg.
In patients undergoing percutaneous coronary intervention, if the last subcutaneous injection of sodium enoxaparin was given less than 8 hours before the balloon catheter inserted into the coronary artery constriction was inflated, no additional injection of sodium enoxaparin is required. If the last subcutaneous injection of sodium enoxaparin was administered more than 8 hours before balloon catheter inflation, an intravenous additional bolus injection of sodium enoxaparin at a dose of 0.3 mg/kg should be given.
In order to improve the accuracy of additional intravenous bolus injection of small volumes into the venous catheter during percutaneous coronary interventions, it is recommended to dilute the drug to a concentration of 3 mg/ml. Dilution of the solution is recommended immediately prior to administration.
To obtain a solution of 3 mg/ml enoxaparin sodium using a pre-filled syringe, it is recommended to use a container with infusion solution, from which a part of the solution is withdrawn to the required volume using a normal syringe. Enoxaparin sodium (the contents of the hypodermic syringe) is injected into the remaining infusion solution in the container.
Volume of pre-filled syringe | Amount of infusion solution left in the reservoir | |
0.3 ml | 10 ml | |
0.6 ml | 20 ml | |
The contents of the container of diluted enoxaparin sodium solution are gently stirred. A syringe is used to extract the desired volume of the diluted enoxaparin sodium solution which is calculated according to the formula:
The volume of the diluted solution = Patient’s body weight (kg) x 0.1 or using the table below.
Volumes to be administered intravenously after dilution
Patient body weight [kg] | Required dose (0.3 mg/kg) [mg] | Volume of solution diluted to a concentration of 3 mg/ml needed to administer | |
45 /p> | 13.5 | 4.5 | |
50 | 15 | 5 | |
55 | 16.5 | 5.5 | 60 | 18 | 6 |
65 | 19.5 |
||
70 | 21 | 7 | |
75 | 22.5 | 7.5 | |
80 | 24 | 8 | |
85 | 25.5 | 8.5 | |
90 | 27 | 9 | |
95 | 28.5 | 9.5 | |
100 | 30 | 10 |
Prevention of thrombosis in the extracorporeal system blood circulation during hemodialysis(usually when the session is no longer than 4 hours)
The dose of enoxaparin sodium averages 1 mg/kg body weight. For patients at high risk of bleeding, the dose should be reduced to 0.5 mg/kg body weight for double vascular access or 0.75 mg for single vascular access.
In hemodialysis, Enixum® should be administered to the arterial shunt site at the beginning of the hemodialysis session. A single dose is usually sufficient for a four-hour session; however, if fibrin rings are detected during longer hemodialysis, an additional dose of 0.5-1 mg/kg body weight may be administered.
The dosing regimen in special patient groups
Elderly patients
Except for treatment of ST-segment elevation myocardial infarction (see above) for all other indications, no dose reduction of enoxaparin sodium is required in elderly patients if they do not have renal impairment.
Patients with renal impairment
Severe renal impairment (endogenous creatinine clearance less than 30 mL/min). The dose of enoxaparin sodium is reduced according to the tables below,
because these patients have increased systemic exposure (duration of action) of the drug.
The following dosing regimen adjustments are recommended when using the drug for therapeutic purposes:
The usual dosing regimen | The dosing regimen for severe renal failure | |
1 mg/kg body weight subcutaneously twice daily | 1 mg/kg body weight subcutaneously once daily | |
1.5 mg/kg body weight subcutaneously once daily /td> | 1 mg/kg body weight subcutaneously once daily | |
Treatment of acute ST-segment elevation myocardial infarction in patients younger than 75 years | ||
Once: A bolus intravenous injection of 30 mg plus 1 mg/kg body weight subcutaneously; followed by a subcutaneous injection of 1 mg/kg body weight once daily (maximum 100 mg for the first subcutaneous injection) /p> | ||
Treatment of acute ST-segment elevation myocardial infarction in patients 75 years and older | ||
0.75 mg/kg body weight subcutaneously twice daily without initial bolus injection (maximum 75 mg for each of the first two subcutaneous injections) /td> | 1 mg/kg body weight subcutaneously once daily without initial intravenous bolus injection (maximum 100 mg for the first subcutaneous injection) | |
The following dosing regimen adjustment is recommended when the drug is used prophylactically
Common dosing regimen | Dosing regimen in severe renal failure |
40 mg subcutaneously once daily | 20 mg subcutaneously once daily /p> |
20 mg subcutaneously once daily | 20 mg subcutaneously once daily |
The recommended dosing adjustment is not applicable for hemodialysis.
In mild (creatinine clearance 50-80 ml/min) and moderate (creatinine clearance 30-50 ml/min) renal failure, dosage adjustment is not required, but patients should be under close medical supervision.
Patients with hepatic impairment
In the absence of clinical studies, caution should be exercised when using enoxaparin sodium in patients with hepatic impairment.
Interaction
Interaction
Do not mix Enixum® with other medicinal products in the same syringe. When concomitant use with other drugs affecting hemostasis (salicylates, incl. Acetylsalicylic acid, nonsteroidal anti-inflammatory drugs (NSAIDs), including ketorolac, 40 kDa dextran, ticlopidine, clopidogrel, systemic glucocorticosteroids (GCS), thrombolytics or anticoagulants, other antiplatelet drugs, including glycoprotein IIb/IIIa receptor antagonists), there is an increased risk of bleeding (see “Precautions”). “Special Precautions”).
Special Instructions
Special Instructions
General
Low molecular weight heparins are not interchangeable, since they differ in manufacturing process, molecular weight, specific anti-Xa activity, dosing units and dosing regimen, with associated differences in their pharmacokinetics and biological activity (antithrombin activity and interaction with thrombocytes). Therefore, it is necessary to strictly follow the recommendations for the use of each drug belonging to the class of low-molecular-weight heparins.
Bleeding
As with other anticoagulants, the use of Enixum® may cause bleeding of any localization (see “Side effects”). In case of bleeding it is necessary to find its source and prescribe an appropriate treatment.
Bleeding in elderly patients
When enoxaparin sodium is used in prophylactic doses in elderly patients, no increased risk of bleeding has been noted.
When using enoxaparin sodium at therapeutic doses, there is an increased risk of bleeding in elderly patients (especially those aged 80 years and older). Close monitoring of these patients is recommended (see section “Pharmacokinetics” and section “Administration and doses”, subsection “Elderly patients”).
Simultaneous use other drugs that affect on hemostasis
It is recommended that the use of drugs that affect hemostasis (salicylates, incl. ч. acetylsalicylic acid, NSAIDs including ketorolac, 40 kDa dextran, ticlopidine, clopidogrel, GCS, thrombolytics, anticoagulants, antiaggregants including glycoprotein IIb/IIIa receptor antagonists) should be stopped before starting enoxaparin sodium treatment unless their use is necessary. If combinations of enoxaparin sodium with these drugs are indicated, close clinical observation and monitoring of relevant laboratory parameters should be performed.
In patients with impaired renal function, there is a risk of bleeding as a result of increased systemic exposure to enoxaparin sodium.
In patients with severe renal impairment (creatinine clearance less than 30 ml/min), there is a significant increase in sodium enoxaparin exposure, therefore it is recommended that dosage adjustment is performed, both during prophylactic and therapeutic use of the drug. Although there is no need to perform dose adjustment in patients with mild to moderate renal dysfunction (creatinine clearance 30-50 ml/min or 50-80 ml/min), close monitoring of these patients is recommended (see sections “Pharmacokinetics” and “Dosage and administration”, subsection “Patients with renal impairment”).
Low body weight
Enoxaparin sodium exposure has been observed to increase with prophylactic administration in women with a body weight less than 45 kg and in men with a body weight less than 57 kg, which may lead to an increased risk of bleeding. Close monitoring of such patients is recommended.
Patients with obesity
Patients with obesity have an increased risk of thrombosis and embolism. The safety and efficacy of enoxaparin sodium at prophylactic doses in patients with obesity (BMI greater than 30 kg/m2) is not fully defined, and there is no general consensus on dose adjustment. These patients should be closely monitored for symptoms and signs of thrombosis and embolism.
Peripheral blood platelet count monitoring
The risk of antibody-mediated heparin-induced thrombocytopenia
is also present with low molecular weight heparins. If thrombocytopenia develops, it is usually detected between days 5 and 21 after initiation of therapy with enoxaparin sodium. Therefore, it is recommended to monitor the peripheral blood platelet count before and during treatment with Enixum® at regular intervals. In case of confirmed significant decrease of platelet count (by 30-50 % compared to the initial value), Enoxaparin sodium should be immediately stopped and the patient should be transferred to another therapy.
Spinal/epidural anesthesia
There have been reports of neuroaxial hematomas when using enoxaparin sodium concomitantly with spinal/epidural anesthesia with the development of long-standing or irreversible paralysis. The risk of these phenomena is reduced when using enoxaparin sodium at a dose of 40 mg or lower.
The risk increases with higher doses of enoxaparin sodium and with the use of permanent catheters after surgery or with the simultaneous use of additional drugs that affect hemostasis, such as NSAIDs (see “Interaction with other drugs”). The risk also increases with traumatic or repeated spinal tap or in patients with a history of spinal surgery or spinal deformity. To reduce the possible risk of bleeding associated with the use of enoxaparin sodium and epidural or spinal anesthesia/analgesia, the pharmacokinetic profile of the drug should be considered (see “Pharmacokinetics”). Catheter placement or removal is best performed when the anticoagulant effect of enoxaparin sodium is low, but the exact time to achieve sufficient reduction in anticoagulant effect in different patients is unknown.
. Catheter insertion or removal should be performed at least 12 h after administration of lower doses of Enixum® (20 mg once daily, 30 mg once or twice daily, 40 mg once daily) and at least 24 h after administration of higher doses of Enixum® (0.75 mg/kg body weight twice daily, 1 mg/kg body weight twice daily, 1.5 mg/kg body weight once daily). Enoxaparin sodium anti-Xa activity is still detectable at these time points, and time delays are no guarantee that neuroaxial hematoma development will be avoided.
Patients receiving enoxaparin sodium at doses of 0.75 mg/kg body weight twice daily or 1 mg/kg body weight twice daily on this (twice daily) dosing regimen should not administer a second dose in order to extend the interval before inserting or replacing a catheter. Similarly, consideration should be given to delaying the next dose of enoxaparin sodium for at least 4 hours based on a benefit/risk assessment (risk of thrombosis and bleeding during the procedure, taking into account patients’ risk factors). However, it is not possible to give clear recommendations on the timing of the next dose of sodium enoxaparin after catheter removal. Note that in patients with a creatinine clearance of less than 30 ml/min, sodium enoxaparin excretion is slower. Therefore, in this category of patients, consideration should be given to doubling the time from catheter removal: at least 24 h for lower doses of sodium enoxaparin (30 mg once daily) and at least 48 h for higher doses (1 mg/kg body weight daily).
If anticoagulant therapy is prescribed by a physician during an epidural/spinal anesthesia or lumbar puncture, the patient should be monitored continuously for any neurologic symptoms such as back pain, sensory and motor impairment (numbness or weakness in lower extremities), bowel and/or bladder function. The patient should be instructed to inform the physician immediately if the symptoms described above occur. Urgent diagnosis and treatment, including decompression of the spinal cord if symptoms associated with a spinal cord hematoma are suspected.
Heparin-induced thrombocytopenia
Enixum® should be used with particular caution in patients with a history of heparin-induced thrombocytopenia with or without thrombosis.
The risk of heparin-induced thrombocytopenia may persist for several years. If heparin-induced thrombocytopenia is suspected anamnesthetically, in vitro platelet aggregation tests are of limited value in predicting the risk of its development. The decision to use Enixum® in this case should be made only after consultation with an appropriate specialist.
Prescapular coronary angioplasty
. In order to minimize the risk of bleeding associated with invasive vascular instrumentation in the treatment of unstable angina and myocardial infarction without Q wave and acute myocardial infarction with ST-segment elevation, these procedures should be performed in intervals between the administration of Enixum®. This is necessary in order to achieve hemostasis after percutaneous coronary intervention. If a femoral artery intraductal device is used, the femoral artery intraductor can be removed immediately. When manual compression is used, the femoral artery intromedullary tube should be removed 6 hours after the last intravenous or subcutaneous injection of enoxaparin sodium. If treatment with sodium enoxaparin is continued, the next dose should not be administered before 6-8 hours after removal of the femoral artery intromedullary tube. The site of insertion of the intraductor should be monitored for signs of bleeding and hematoma in a timely fashion.
Patients with mechanical heart valves
The use of sodium enoxaparin for thrombotic prophylaxis in patients with mechanical heart valves has not been well studied. There have been isolated reports of heart valve thrombosis in patients with mechanical heart valves on therapy with enoxaparin sodium for thrombosis prophylaxis. The evaluation of these reports is limited because of competing factors contributing to the development of artificial heart valve thrombosis, including underlying disease, and because of insufficient clinical data.
Pregnant women with mechanical artificial valves heart
Pregnant women women who are pregnant with mechanical and have a heart.
The use of enoxaparin sodium to prevent thrombosis in pregnant women with mechanical artificial heart valves has not been well studied. In a clinical study of pregnant women with mechanical heart valves, when enoxaparin sodium was used at a dose of 1 mg/kg body weight twice daily to reduce the risk of thrombosis and embolism, 2 of 8 women developed thrombi, leading to heart valve blockage and maternal and fetal death.
There have been anecdotal post-marketing reports of heart valve thrombosis in pregnant women with mechanical artificial heart valves treated with enoxaparin sodium to prevent thrombosis. Pregnant women with mechanical heart valves have a high risk of thrombosis and embolism.
Laboratory tests
In doses used for the prevention of thromboembolic complications, sodium enoxaparin has no significant effect on bleeding time and clotting parameters, or on platelet aggregation or binding to fibrinogen.
Additional doses may lengthen the ACTV and activated clotting time. Increases in ACTV and activated clotting time are not in direct linear relationship to increases in anticoagulant activity of the drug, so there is no need to monitor them.
Prevention of venous thrombosis and embolism in patients with acute therapeutic conditions who are on bed rest
Prevention of venous thrombosis and embolism in patients with acute therapeutic conditions. In the case of acute infection, acute rheumatic conditions, prophylactic use of enoxaparin sodium is warranted only if the above conditions are combined with one of the following risk factors for venous thrombosis:
– age greater than 75 years;
– malignant neoplasms;
– history of thrombosis and embolism;
– obesity;
– hormone therapy;
– heart failure;
– chronic respiratory failure.
Pediatric use
The safety and effectiveness of enoxaparin sodium in children under the age of 18 years has not been established.
Impact on performance of potentially hazardous activities requiring particular attention and responsiveness
There are no data indicating adverse effects of enoxaparin sodium on the ability to drive vehicles and engage in other potentially hazardous activities requiring increased concentration and psychomotor responsiveness.
Contraindications
Contraindications
Hypersensitivity to enoxaparin sodium, heparin or its derivatives, including other low molecular weight heparins;
– active major bleeding, as well as conditions and diseases with a high risk of bleeding: threatened abortion, cerebral aneurysm or dissecting aortic aneurysm (except in cases of surgical intervention for this purpose), recent
hemorrhagic stroke, uncontrolled bleeding, thrombocytopenia in combination with a positive test in vitro for antiplatelet antibodies in
the presence of enoxaparin sodium;
– children under 18 years of age, since the effectiveness and safety in this category of
patients has not been established (see section “Indications.
– children under 18 years of age.)
With caution
Conditions with a potential risk of bleeding:
– hemostasis disorders (including hemophilia, thrombocytopenia, hypocoagulation, Willebrand’s disease and others).), severe vasculitis;
– gastric or duodenal ulcer or other erosive-ulcerative lesions of the gastrointestinal tract in the anamnesis;
– uncontrolled severe arterial hypertension;
– diabetic or hemorrhagic retinopathy;
– severe diabetes;
– recent or suspected neurological or ophthalmologic
surgery;
– spinal or epidural anesthesia (potential risk of hematoma), spinal tap (recent);
– recent childbirth;
– bacterial endocarditis (acute or subacute);
– pericarditis or pericardial effusion;
– renal and/or liver failure;
– intrauterine contraception (IUD);
– severe trauma (especially central nervous system (CNS), open wounds on
large surfaces;
– simultaneous use of drugs that affect the hemostatic system;
– heparin-induced thrombocytopenia (in anamnesis) in combination with thrombosis or
without it.
There are no data on the clinical use of enoxaparin sodium in the following
diseases: active tuberculosis, radiation therapy (recent)
.
Side effects
Side effects
Side effects were classified by frequency as follows: very frequent ( ≥ 1/10), frequent ( ≥ 1/100 to < 1/10), infrequent ( ≥ 1/1000 to < 1/100), rare ( ≥ 1/10000 to < 1/1000), very rare (< 1/10000).
bleeding
Hemorrhage may occur, especially in the presence of associated risk factors: organic changes with propensity for bleeding, age, renal insufficiency, low body weight, and certain drug combinations (see “Interactions with other drugs”). If bleeding develops, the drug administration should be stopped, the cause of the bleeding identified and appropriate therapy initiated.
Very common are bleeding during prophylaxis of venous thrombosis, in surgical patients and treatment of deep vein thrombosis with or without thromboembolism.
Frequent – Bleeding in prophylaxis of venous thrombosis in bed-ridden patients and in treatment of angina pectoris, myocardial infarction without Q wave and myocardial infarction with ST-segment elevation.
Infrequent – retroperitoneal bleeding and intracranial bleeding in patients treated for deep vein thrombosis with or without thromboembolism and ST-segment elevation myocardial infarction.
Rarely, retroperitoneal bleeding in the prevention of venous thrombosis in surgical patients and in the treatment of angina, myocardial infarction without the Q-wave.
When using enoxaparin sodium against the background of spinal/epidural anesthesia and postoperative use of penetrating catheters, there have been rare cases of neuroaxial hematomas resulting in neurologic impairment of varying severity, including long-term or permanent paralysis (see “Special Considerations”).
Thrombocytopenia and thrombocytosis
Very common – thrombocytosis in the prevention of venous thrombosis in surgical patients and treatment of deep vein thrombosis with or without thromboembolism.
Frequent – thrombocytopenia. In prophylaxis of venous thrombosis in surgical patients and treatment of deep vein thrombosis with or without thromboembolism, and in ST-segment elevation myocardial infarction.
Infrequent – thrombocytopenia in the prevention of venous thrombosis in patients on bed rest and in the treatment of angina pectoris, myocardial infarction without Q-wave. Very rare – autoimmune thrombocytopenia in ST-segment elevation myocardial infarction.
In rare cases, the development of autoimmune thrombocytopenia in combination with thrombosis has been reported. In some of these cases, thrombosis has been complicated by organ infarction or limb ischemia (see section “Special Precautions”).
Others
Very often, increased hepatic transaminase activity.
Often – allergic reactions, urticaria, itching, redness of the skin, bruising and pain at the injection site.
Infrequent – skin (bullous rashes), inflammatory reaction at the injection site, skin necrosis at the injection site.
Rarely – anaphylactic and anaphylactoid reactions, hyperkalemia. Necrosis of the skin may develop at the injection site preceded by purpura or erythematous painful papules. In such cases, therapy with the drug should be discontinued. Solid inflammatory nodules/infiltrates may form at the injection site, which disappear after a few days and are not a reason for discontinuation of the drug.
Overdose
Overdose
Symptoms: hemorrhagic complications of accidental overdose with subcutaneous injection of enoxaparin sodium. Even high doses are unlikely to be absorbed by accidental ingestion.
Treatment: Neutralize the effects of enoxaparin sodium by slow intravenous (IV) administration of protamine sulfate. 1 mg of protamine sulfate neutralizes the anticoagulant effect of 1 mg of enoxaparin sodium if the drug was administered no more than 8 h before administration of protamine sulfate.
The 0.5 mg of protamine sulfate will neutralize the anticoagulant effect of 1 mg of enoxaparin sodium if administered more than 8 h before or if a second dose of protamine sulfate must be administered.
If, however, 12 hours or more have elapsed since the administration of enoxaparin sodium, the administration of protamine sulfate is unnecessary. However, even with high doses of protamine sulfate, the anti-Xa activity of sodium enoxaparin is not completely neutralized (maximum 60%).
Pregnancy use
Pregnancy use
Pregnancy
Currently, there are insufficient clinical data available to determine the possible teratogenic or fetotoxic effects of enoxaparin sodium when administered for prophylactic purposes during pregnancy. Enixum® should not be used during pregnancy unless the potential benefit to the mother outweighs the possible risk to the fetus.
A spinal or epidural anesthesia should not be performed during treatment with the drug.
If epidural anesthesia is planned, preventive treatment with Enoxaparin sodium should be discontinued if possible at least 12 h prior to anesthesia.
Enoxaparin sodium is not recommended in pregnant women with prosthetic heart valves.
Breastfeeding
It is not known whether unchanged enoxaparin sodium is excreted into human breast milk. Absorption of enoxaparin sodium into the gastrointestinal tract in the infant is unlikely. However, as a precautionary measure, breastfeeding women treated with enoxaparin sodium should be advised to interrupt breastfeeding.
Similarities
Similarities
Clexane
Additional information
| Weight | 0.043 kg |
|---|---|
| Shelf life | 2 years. Do not use after the expiration date stated on the package. |
| Conditions of storage | At a temperature not exceeding 25 ° C. Do not freeze. Keep out of reach of children. |
| Manufacturer | PharmFirm Sotex, Russia |
| Medication form | solution for injection |
| Brand | PharmFirm Sotex |
Other forms…
Related products
Buy Enixum, 6000 anti-ha me/0.6 mL 0.6 mL 10 pcs with delivery to USA, UK, Europe and over 120 other countries.