No products in the cart.
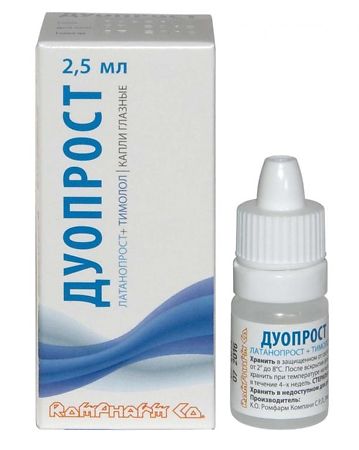
Duoprost, eye drops 2.5 ml
€26.38 €21.98
Description
Duoprost is an anti-glaucoma combination drug.
Latanoprost is a prostaglandin F2α analog and a selective FP receptor agonist. It reduces intraocular pressure by increasing outflow of aqueous humor and has anti-glaucoma effect.
The main mechanism of action of latanoprost is associated with an increase in uveoscleral outflow. It has no significant effect on aqueous humor production and does not affect the blood-ophthalmic barrier.
Timolol is a non-selective β-adrenoreceptor blocker with minor intrinsic sympathomimetic and membrane stabilizing activity. When used topically, it lowers intraocular pressure by reducing aqueous humor formation and slightly increasing its outflow.
Pharmacokinetics
Latanoprost
Absorption and distribution/p>
Cmax in aqueous humor (approximately 15-30 ng/mL) is reached 2 h after instillation into the eye. After application of the eye drops, latanoprost is distributed primarily in the anterior segment of the eye, in the conjunctiva and in the eyelids. Only a small amount of the drug reaches the posterior segment. Latanoprost undergoes hydrolysis in the cornea by esterases to form a biologically active acid.
Metabolism and excretion
The metabolism of latanoprost acid in the eye tissues is almost non-existent. The main metabolism occurs in the liver. T1/2 from plasma is 17 min. The major metabolites, 1,2-dinor- and 1,2,3,4-tetranor-metabolites, have little or no biological activity and are mostly excreted in the urine.
Timolol
Timololol penetrates rapidly through the cornea when administered topically. After instillation of eye drops, the Cmax of timolol in the aqueous humor of the eye is reached after 1 hour.
T1/2 from plasma is 6 hours. A small amount enters the systemic bloodstream by absorption through the vessels of the conjunctiva, nasal mucosa and lacrimal tract. Excretion of thymolol metabolites is mainly by the kidneys.
Indications
Indications
Increased intraocular pressure of any nature
Open angle glaucoma
Pharmacological effect
Pharmacological effect
Duoprost is an antiglaucoma combination drug.
Latanoprost is a prostaglandin F2α analog and a selective FP receptor agonist. Reduces intraocular pressure by increasing the outflow of aqueous humor and has an antiglaucoma effect.
The main mechanism of action of latanoprost is associated with an increase in uveoscleral outflow. It does not have a significant effect on the production of aqueous humor and does not affect the blood-ophthalmic barrier.
Timolol is a non-selective β-adrenergic receptor blocker with minor intrinsic sympathomimetic and membrane-stabilizing activity. When applied topically, it lowers intraocular pressure by reducing the formation of aqueous humor and slightly increasing its outflow.
Pharmacokinetics
Latanoprost
Suction and distribution
Cmax in aqueous humor (approximately 15-30 ng/ml) is achieved 2 hours after instillation into the eye. After application of eye drops, latanoprost is distributed primarily in the anterior segment of the eye, in the conjunctiva and in the eyelids. Only a small amount of the drug reaches the posterior segment. Latanoprost undergoes hydrolysis in the cornea under the action of esterases to form a biologically active acid.
Metabolism and excretion
Metabolism of latanoprost acid in the tissues of the eye practically does not occur. The main metabolism occurs in the liver. T1/2 from plasma is 17 minutes. The main metabolites, 1,2-dinor and 1,2,3,4-tetranor metabolites, have no or weak biological activity and are mainly excreted in the urine.
Timolol
When applied topically, timolol quickly penetrates the cornea. After instillation of eye drops, the Cmax of timolol in the aqueous humor of the eye is reached after 1 hour.
T1/2 from plasma – 6 hours. A small amount enters the systemic circulation by absorption through the vessels of the conjunctiva, nasal mucosa and lacrimal tract. Excretion of timolol metabolites is carried out primarily by the kidneys.
Special instructions
Special instructions
Duoprost can cause changes in eye color due to increased levels of brown pigment in the iris.
The drug contains benzalkonium chloride, which is absorbed by contact lenses, damaging eye tissue. Therefore, before using it, do not forget to remove your contact lenses and reinsert them after 15 minutes.
Instillation of eye drops can cause temporary changes in vision, which must be taken into account when driving a car or working with moving machinery.
Active ingredient
Active ingredient
Latanoprost, Timolol
Composition
Composition
Active ingredients:
latanoprost 50 mcg;
timolol (in the form of maleate) 5 mg;
Excipients:
disodium hydrogen phosphate dodecahydrate – 11.85 mg,
sodium dihydrogen phosphate dihydrate – 5.2 mg,
sodium chloride – 4.1 mg,
benzalkonium chloride – 0.2 mg,
sodium hydroxide solution 1M/hydrochloric acid 1M solution – up to pH 6.0±0.1,
purified water – up to 1 ml
Pregnancy
Pregnancy
Adequate and strictly controlled clinical studies on the safety of the drug during pregnancy and lactation (breastfeeding) have not been conducted.
The use of the drug during pregnancy is possible only if the expected benefit to the mother outweighs the potential risk to the fetus or child.
The drug should not be prescribed to nursing women, or breastfeeding should be avoided (timolol, latanoprost and its metabolites can pass into breast milk).
Contraindicated for children and adolescents under 18 years of age.
Contraindications
Contraindications
Hypersensitivity;
COPD, bronchial asthma (including medical history);
Sinus bradycardia, chronic heart failure in the stage of decompensation, II-III degree of atrioventricular block, cardiogenic shock;
Children and adolescents under 18 years of age;
Breastfeeding period.
Duoprost may be prescribed with caution when:
inflammatory, neovascular, closed-angle or congenital glaucoma, as well as open-angle glaucoma combined with pseudophakia and pigmentary glaucoma (due to lack of clinical experience);
aphakia, pseudophakia and rupture of the posterior capsule of the lens, pregnancy;
patients at high risk of macular edema (including cystoid).
Side Effects
Side Effects
Visual impairment, blepharitis, cataracts, conjunctivitis, allergic reactions of the conjunctiva (follicles, pinpoint hemorrhages, papillary reactions of the conjunctiva, etc.), corneal lesions (pigmentation, erosion, punctate keratitis, etc.), refractive errors, redness of the eye, pain, irritation of the eye, increased pigmentation of the iris, keratitis, photophobia, loss of part of the visual fields, decreased sensitivity of the cornea, ptosis, changes in refraction, diplopia.
Cerebral ischemia, circulatory disorders.
Diarrhea, dyspepsia, dry mouth, nausea, retroperitoneal fibrosis.
Shortness of breath, bronchospasm.
Upper respiratory tract infections, sinusitis, etc.
Diabetes mellitus, hypercholesterolemia.
Angioedema, anaphylaxis, urticaria, generalized rash.
Depression, headache, dizziness, confusion.
Increased blood pressure.
Hypertrichosis, skin irritations, rash, chalazion, etc.
Arthritis, pain in muscles, joints.
Intermittent claudication, Raynaud’s syndrome, decreased blood pressure.
Impotence, Peyronie’s disease.
Interaction
Interaction
The simultaneous use of Duoprost and blockers of “slow” calcium channels, antiarrhythmic drugs, beta-blockers, cardiac glycosides, cholinomimetics, narcotic analgesics, monoamine oxidase inhibitors, as well as drugs that reduce the activity of catecholamines, may enhance its hypotensive effect or cause severe bradycardia.
The use of timolol with epinephrine occasionally causes mydriasis.
Overdose
Overdose
Symptoms of latanoprost overdose: conjunctival or episcleral hyperemia, eye irritation, dizziness; signs of timolol overdose: arrhythmia, headache, bronchospasm, bradycardia, nausea, vomiting.
Therapeutic measures: rinse eyes with water or saline solution, apply symptomatic therapy.
Storage conditions
Storage conditions
The drug should be stored in a place protected from light at a temperature of 2° to 8°C.
Shelf life
Shelf life
3 years. After opening the bottle – 4 weeks.
Manufacturer
Manufacturer
K.O.Rompharm Company S.R.L., Romania
Additional information
| Shelf life | 3 years. After opening the bottle – 4 weeks. |
|---|---|
| Conditions of storage | The drug should be stored in the dark place at temperatures from 2 ° to 8 ° C. |
| Manufacturer | C.O.Rompharm Company S.R.L., Romania |
| Medication form | eye drops |
| Brand | C.O.Rompharm Company S.R.L. |
Related products
Buy Duoprost, eye drops 2.5 ml with delivery to USA, UK, Europe and over 120 other countries.