No products in the cart.
Diuver, tablets 10 mg 20 pcs
€1.00
Out of stock
(E-mail when Stock is available)
Description
Pharmacotherapeutic group: diuretic agent.
AtCode C03CA04
Pharmacological properties
Pharmacodynamics
Indications
Indications
Edema, Hypertension (high blood pressure), Heart failure, Liver damage
– edema syndrome of different genesis, including chronic heart failure, liver, lung and kidney disease;
– arterial hypertension.
Active ingredient
Active ingredient
Torasemide
Composition
Composition
1 tablet 5 mg contains:
active ingredient: torasemide 5.00 mg;
excipients: lactose monohydrate 58.44 mg, corn starch 14.56 mg, sodium carboxymethyl starch 0.80 mg, colloidal anhydrous silica 0.60 mg, magnesium stearate 0.60 mg.
1 tablet 10 mg contains:
acting substance: torasemide 10.00 mg;
complementary substances: lactose monohydrate 116.88 mg, corn starch 29.12 mg, sodium carboxymethyl starch 1.60 mg, colloidal anhydrous silica 1.20 mg, magnesium stearate 1.20 mg.
How to take, the dosage
How to take, the dosage
Overly, once a day, after breakfast, with a small amount of water.
Oedematous syndrome of different genesis, including. In chronic heart failure, liver, lung and kidney disease
The usual therapeutic dose is 5 mg orally once daily. If necessary, the dose should be gradually increased to 20-40 mg once daily, in some cases up to 200 mg per day. The drug is prescribed for a long period or until the edema disappears.
Arterial hypertension
The initial dose is 2.5 mg (1/2 tablet of 5 mg) once daily. If necessary, the dose may be increased to 5 mg once daily.
Elderly patients do not need dose adjustment.
Interaction
Interaction
Increases the concentration and risk of nephro- and ototoxic effects of cephalosporins, aminoglycosides, chloramphenicol, etacrynic acid, cisplatin, amphotericin B (due to competitive renal excretion).
Increases the efficacy of diazoxide and theophylline, decreases the efficacy of hypoglycemic agents, allopurinol.
Presor amines and thorasemide mutually decrease the effectiveness.
Drugs that block the tubular secretion increase the serum concentration of thorasemide.
Concomitant use of glucocorticosteroids, amphotericin B increases the risk of hypokalemia, with cardiac glycosides – increased risk of glycoside intoxication due to hypokalemia (for high and low polar) and lengthening of the elimination half-life (for low polar).
Reduces renal clearance of lithium drugs and increases the possibility of intoxication. Non-steroidal anti-inflammatory drugs and sucralfate decrease diuretic effect due to inhibition of prostaglandin synthesis, disturbance of plasma renin activity and aldosterone excretion.
It increases hypotensive effect of hypotensive drugs, neuromuscular blockade of depolarizing muscle relaxants (suxamethonium) and decreases effect of nondepolarizing muscle relaxants (tubocurarin).
Simultaneous use of high doses of salicylates during treatment with thorasemide increases the risk of their toxicity (due to competitive renal excretion).
Serial or simultaneous use of thorasemide with angiotensin-converting enzyme inhibitors (ACE) or angiotensin II receptor antagonists may lead to a strong decrease of blood pressure. This can be avoided by reducing the dose of thorasemide or temporarily cancelling it.
Concomitant use of probenecid or methotrexate may reduce the effectiveness of thorasemide (same secretion pathway). On the other hand, thorasemide may lead to decreased renal elimination of these drugs.
Concomitant use of cyclosporine and thorasemide increases the risk of gouty arthritis due to the fact that cyclosporine may cause impaired renal excretion of urate and thorasemide may cause hyperuricemia.
It has been reported that patients at high risk of nephropathy taking oral thoracemide were more likely to have impaired renal function when administering radiopaque agents than patients at high risk of nephropathy who received intravenous hydration before the administration of radiopaque agents.
Special Instructions
Special Instructions
Patients with hypersensitivity to sulfonamides and sulfonylurea derivatives may have cross-sensitivity to the drug Diuver.
Patients receiving high doses of Diuver over a long period of time are recommended to have a diet with a sufficient content of table salt and use potassium preparations to avoid development of hyponatremia, metabolic alkalosis and hypokalemia.
An increased risk of the water-electrolyte balance disorders is noted in patients with renal insufficiency. During the course of treatment it is necessary to monitor periodically the concentration of plasma electrolytes (including sodium, calcium, potassium, magnesium), acid-base status, residual nitrogen, creatinine, uric acid and if necessary perform the appropriate corrective therapy (with higher frequency of patients with frequent vomiting and parenterally administered fluids).
In case of the appearance or increase of azotemia and oliguria in patients with severe progressive renal disease it is recommended to suspend treatment.
The dosage regimen of patients with ascites due to liver cirrhosis should be performed in in-patient settings (disorders of water-electrolyte balance may lead to hepatic coma). This category of patients requires regular monitoring of plasma electrolytes.
Patients with diabetes or reduced glucose tolerance require periodic monitoring of glucose concentration in blood and urine.
Patients who are unconscious, with prostatic hyperplasia, ureteral narrowing require diuresis control due to possible acute urine retention.
During treatment patients should refrain from driving vehicles and engaging in other potentially dangerous activities that require high concentration and quick psychomotor reactions.
Synopsis
Synopsis
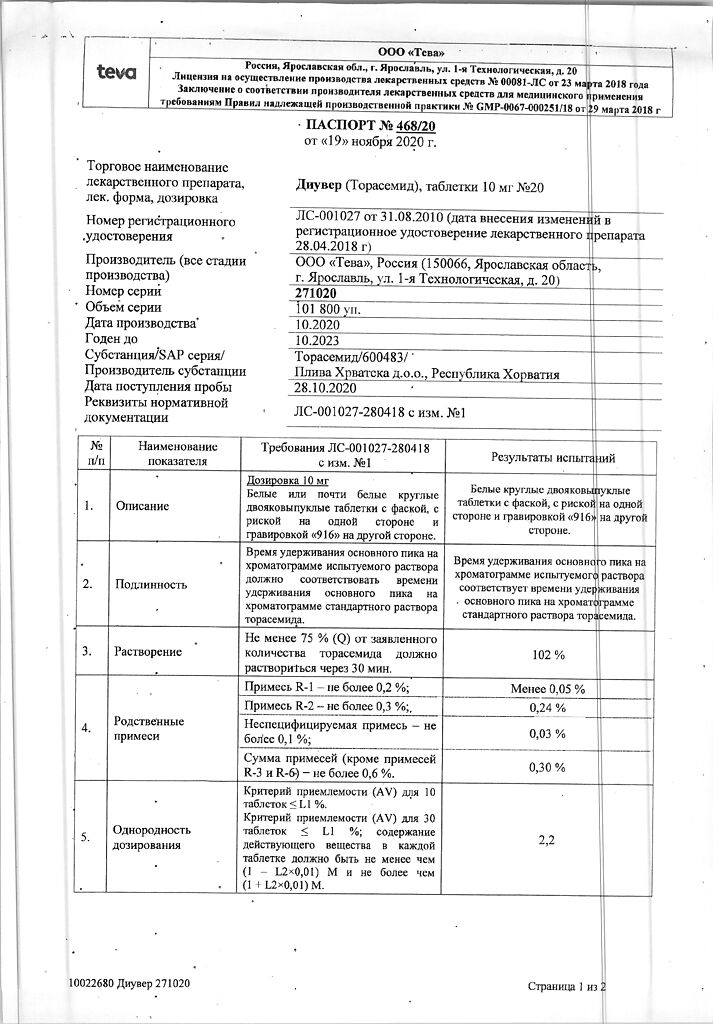
Dosage of 5 mg:
white or almost white round biconvex tablets with a bevel on one side and engraved “915” on the other side.
Dosage 10 mg:
white or almost white round biconvex tablets, beveled, with a rib on one side and engraving “916” on the other side.
Contraindications
Contraindications
Hypersensitivity to thorasemide or to any of the drug components; in patients with allergy to sulfonamides (sulfonamide antimicrobials or sulfonylureas); renal failure with anuria; hepatic coma and precoma; marked hypokalemia; marked hyponatremia; hypovolemia (with or without arterial hypotension) or dehydration; acute glomerulonephritis; decompensated aortic and mitral stenosis, hypertrophic obstructive cardiomyopathy; increased central venous pressure (over 10 mm Hg).
Lactose intolerance, lactase deficiency or glucose-galactose malabsorption.
Cautions
. Arterial hypotension, stenotic atherosclerosis of cerebral arteries, hypoproteinemia, predisposition to hyperuricemia, urinary outflow disorders (benign prostatic hyperplasia, urethral narrowing or hydronephrosis), ventricular arrhythmia in anamnesis, acute myocardial infarction (increased risk of cardiogenic shock), diarrhea, pancreatitis, diabetes (decreased glucose tolerance), hepatorenal syndrome, gout, anemia, pregnancy.
Side effects
Side effects
Water-electrolyte and acid-base balance: hyponatremia, hypochloremia, hypokalemia, hypomagnesemia, hypocalcemia, metabolic alkalosis. Symptoms indicative of the development of electrolyte and acid-base state disorders may include headache, confusion, seizures, tetany, muscle weakness, heart rhythm disorders and dyspeptic disorders; hypovolemia and dehydration (more common in elderly patients), which may lead to hemoconcentration with a tendency to develop thrombosis.
Cardiovascular system disorders: excessive decrease in blood pressure, orthostatic hypotension, collapse, tachycardia, arrhythmias, decreased circulating blood volume.
Metabolic side: Hypercholesterolemia, hypertriglyceridemia; transient increase in blood creatinine and urea concentrations; increase in blood uric acid concentration, which may cause or worsen manifestations of gout; decrease in glucose tolerance (possible manifestation of latent diabetes).
Urinary system disorders: oliguria, acute urinary retention (e.g., in prostatic hyperplasia, narrowing of the urethra, hydronephrosis); interstitial nephritis, hematuria, decreased potency.
Gastrointestinal tract disorders: nausea, vomiting, diarrhea, intrahepatic cholestasis, increased activity of “liver” enzymes, acute pancreatitis.
Central nervous system, hearing: disorder of hearing, usually reversible, and/or tinnitus, especially in patients with renal failure or hypoproteinemia (nephrotic syndrome), paresthesias.
Skin side: cutaneous itching, urticaria, other rashes or bullous skin lesions, erythema polymorphicum, exfoliative dermatitis, purpura, fever, vasculitis, eosinophilia, photosensitization; severe anaphylactic or anaphylactoid reactions up to shock, which so far have only been described after intravenous administration.
Peripheral blood: thrombocytopenia; leukopenia; agranulocytosis, aplastic or hemolytic anemia.
Overdose
Overdose
Symptoms: Excessive diuresis accompanied by decreased circulating blood volume and blood electrolyte imbalance, followed by a marked decrease in blood pressure, drowsiness and confusion, collapse. Gastrointestinal disorders may be observed.
Treatment: There is no specific antidote. Provocation of vomiting, gastric lavage, activated charcoal. Treatment is symptomatic, dose reduction or discontinuation of the drug and simultaneously replenishment of the blood circulation and parameters of water-electrolyte balance and acid-base status under control of serum concentrations of electrolytes, hematocrit, symptomatic treatment.
Hemodialysis is ineffective.
Pregnancy use
Pregnancy use
Torasemide has no teratogenic effect and fetotoxicity, penetrates through the placental barrier, causing water-electrolyte metabolism disorders and thrombocytopenia in the fetus.
The drug Diuver during pregnancy can be used only if the benefit to the mother exceeds the possible potential risk to the fetus, only under medical supervision and only in the minimum dose.
It is unknown whether thoracemide penetrates into the breast milk. If it is necessary to use Diuver during lactation, breastfeeding should be stopped.
Similarities
Similarities
Trigrim, Torasemide-SZ, Lotonel, Torasemide
Additional information
| Weight | 0.043 kg |
|---|---|
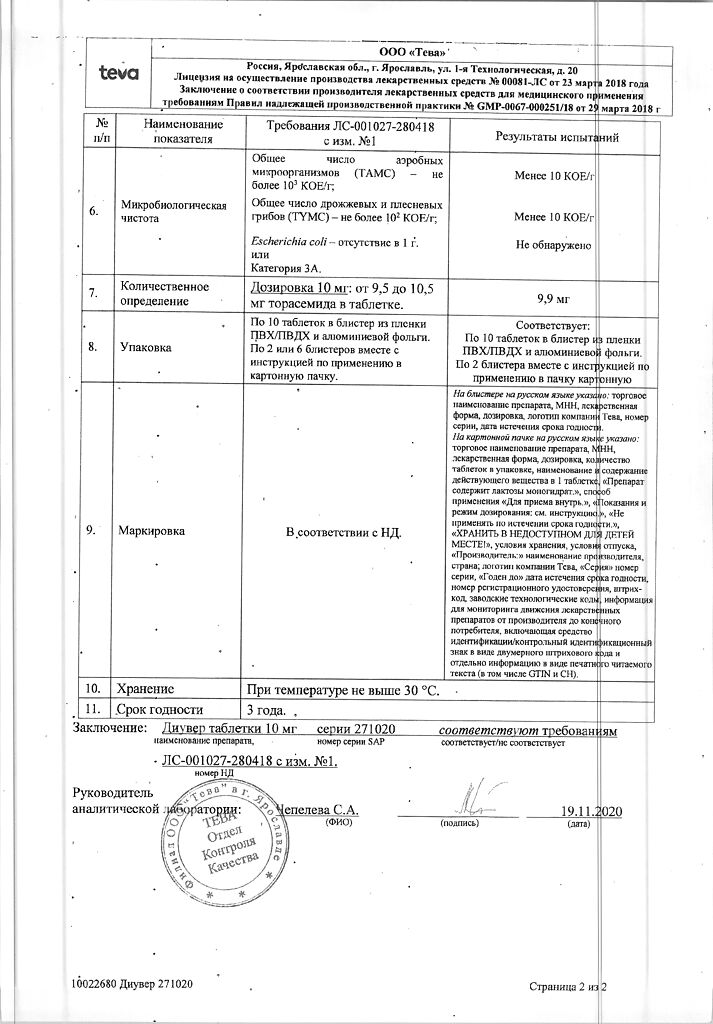
| Shelf life | 3 years. Do not use after the expiration date. |
| Conditions of storage | At a temperature not exceeding 25°C. Keep out of reach of children! |
| Manufacturer | Teva LLC, Russia |
| Medication form | pills |
| Brand | Teva LLC |
Related products
Buy Diuver, tablets 10 mg 20 pcs with delivery to USA, UK, Europe and over 120 other countries.