No products in the cart.
DexaTobropt, eye drops 5 ml
€12.25 €10.21
Description
A combination drug for topical use in ophthalmology with anti-inflammatory and antibacterial action.
Tobramycin is a broad-spectrum antibiotic of the aminoglycoside group. It is active against Gram-positive and Gram-negative microorganisms: Staphylococcus spp. (including Staphylococcus aureus, Staphylococcus epidermidis, including penicillin-resistant strains); Streptococcus spp. some group A beta-haemolytic species, some non-haemolytic species and some strains of Streptococcus pneumoniae), Pseudomonas aeruginosa, Escherichia coli, Klebsiella pneumoniae, Enterobacter aerogenes, Proteus mirabilis, Morganella morganii, most species of Proteus vulgaris, Haemophilus influenzae, Haemophilus aegyptius, Moraxella lacunata, Acinetobacter calcoaceticus (Herellea vaginacola), and some Neisseria spp.
Dexamethasone is a GCS. It has a pronounced anti-inflammatory, anti-allergic and desensitizing effect. Dexamethasone actively suppresses inflammatory processes by inhibiting the release of inflammatory mediators by eosinophils, migration of mast cells and reducing capillary permeability. It does not have mineralocorticoid activity.
Pharmacokinetics
After topical administration, tobramycin is absorbed through the cornea and conjunctiva. A very small amount reaches the aqueous humor. Tobramycin reaches the systemic bloodstream in very low concentrations. When applied topically, systemic absorption of dexamethasone is low. After injection into the eye, it penetrates well into the corneal epithelium and conjunctiva, aqueous humor of the eye. Maximum activity is reached within 10-20 min after application of the drug.
The distribution of the drug was observed in plasma, bile, liver, kidney and renal excretion after 30 min of administration.
Indications
Indications
Infectious eye diseases:
blepharoconjunctivitis,
blepharitis,
conjunctivitis,
keratitis (without damage to the epithelium);
Treatment and prevention of inflammatory phenomena in the postoperative period after cataract extraction.
Pharmacological effect
Pharmacological effect
Combined drug for local use in ophthalmology with anti-inflammatory and antibacterial effects.
Tobramycin is a broad-spectrum antibiotic from the aminoglycoside group. Active against gram-positive and gram-negative microorganisms: Staphylococcus spp. (including Staphylococcus aureus, Staphylococcus epidermidis, including strains resistant to penicillin); Streptococcus spp. (including some beta-hemolytic species of group A, some non-hemolytic species and some strains of Streptococcus pneumoniae), Pseudomonas aeruginosa, Escherichia coli, Klebsiella pneumoniae, Enterobacter aerogenes, Proteus mirabilis, Morganella morganii, most species of Proteus vulgaris, Haemophilus influenzae, Haemophilus aegyptius, Moraxella lacunata, Acinetobacter calcoaceticus (Herellea vaginacola), as well as some species of Neisseria spp.
Dexamethasone – GCS. It has a pronounced anti-inflammatory, antiallergic and desensitizing effect. Dexamethasone actively suppresses inflammatory processes, inhibiting the release of inflammatory mediators by eosinophils, the migration of mast cells and reducing capillary permeability. Does not have mineralocorticoid activity.
Pharmacokinetics
After topical application, tobramycin is absorbed through the cornea and conjunctiva. A very small amount reaches the aqueous humor. Tobramycin reaches the systemic circulation in very low concentrations. When applied topically, systemic absorption of dexamethasone is low. After instillation into the eye, it penetrates well into the epithelium of the cornea and conjunctiva, the aqueous humor of the eye. Maximum activity is achieved within 10-20 minutes after using the drug.
Distribution of the drug was observed in blood plasma, bile, liver, kidneys and renal excretion after 30 minutes from the moment of administration.
Special instructions
Special instructions
DexaTobropt is for topical use only (not for injection).
When using drugs containing corticosteroids for more than 10 days, intraocular pressure should be regularly measured.
When prescribing DexaTobropt simultaneously with aminoglycoside antibiotics for systemic use, it is necessary to monitor the concentration of tobramycin in the blood serum, which should not exceed 8 mcg/ml.
If allergic reactions develop, use of the drug should be discontinued.
Because The eye drops contain benzalkonium chloride as a preservative, which may cause irritation and is known to discolor soft contact lenses. Therefore, patients should remove contact lenses before using DexaTobropt and should be informed to wait 15 minutes after installing DexaTobropt before inserting contact lenses.
Impact on the ability to drive vehicles and operate machinery
There have been no reports of adverse effects of the drug on the ability to drive vehicles or engage in other activities that require concentration and speed of psychomotor reactions. However, given that the patient temporarily loses clarity of vision after the application, it is not recommended to drive vehicles or work with complex equipment immediately after instilling the drug.
Active ingredient
Active ingredient
Dexamethasone, Tobramycin
Composition
Composition
Eye drops in the form of a suspension of white or almost white color, there may be a sediment that quickly dissipates with gentle shaking.
1 ml
dexamethasone
1 mg
tobramycin
3 mg
Excipients:
tyloxapol – 0.5 mg,
hypromellose (HPMC 4000 cP) – 5 mg,
sodium sulfate – 12 mg,
sodium chloride – 3.3 mg,
disodium edetate dihydrate – 0.1 mg,
benzalkonium chloride – 0.1 mg,
sodium hydroxide 1M solution or sulfuric acid 1M solution – up to pH 5.0-6.0,
purified water – up to 1 ml.
Pregnancy
Pregnancy
There is no sufficient experience with the use of the drug during pregnancy and lactation (breastfeeding).
The use of DexTobropt during pregnancy is possible only if the expected effect of therapy for the mother exceeds the potential risk to the fetus.
Breastfeeding should be stopped during treatment.
Use in pediatrics
At the moment, the safety and effectiveness of the drug in children have not been established. Contraindicated for children and adolescents under 18 years of age.
Contraindications
Contraindications
viral eye diseases (including keratitis caused by Herpes simplex, chicken pox);
fungal eye diseases;
mycobacterial eye infections;
conditions after removal of a foreign body from the cornea;
hypersensitivity to the components of the drug.
Side Effects
Side Effects
swelling, itching of the eyelids, conjunctival hyperemia.
increased intraocular pressure (with long-term use). With continuous use for more than 3 months, the development of corneal mycoses is possible. Possible slowdown of regeneration processes, development of posterior subcapsular cataracts, and secondary infection.
headache, increased blood pressure.
Interaction
Interaction
No drug interactions have been established.
When used with other local ophthalmic drugs, the interval between their use should be at least 5 minutes.
Storage conditions
Storage conditions
In a dry place, protected from light, at a temperature not exceeding 25 °C
Shelf life
Shelf life
3 years
Manufacturer
Manufacturer
K.O.Rompharm Company S.R.L., Romania
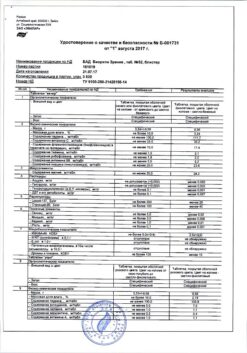
Additional information
| Shelf life | 3 years |
|---|---|
| Conditions of storage | In a dry, light-protected place at a temperature not exceeding 25 °C |
| Manufacturer | C.O.Rompharm Company S.R.L., Romania |
| Medication form | eye drops |
| Brand | C.O.Rompharm Company S.R.L. |
Related products
Buy DexaTobropt, eye drops 5 ml with delivery to USA, UK, Europe and over 120 other countries.