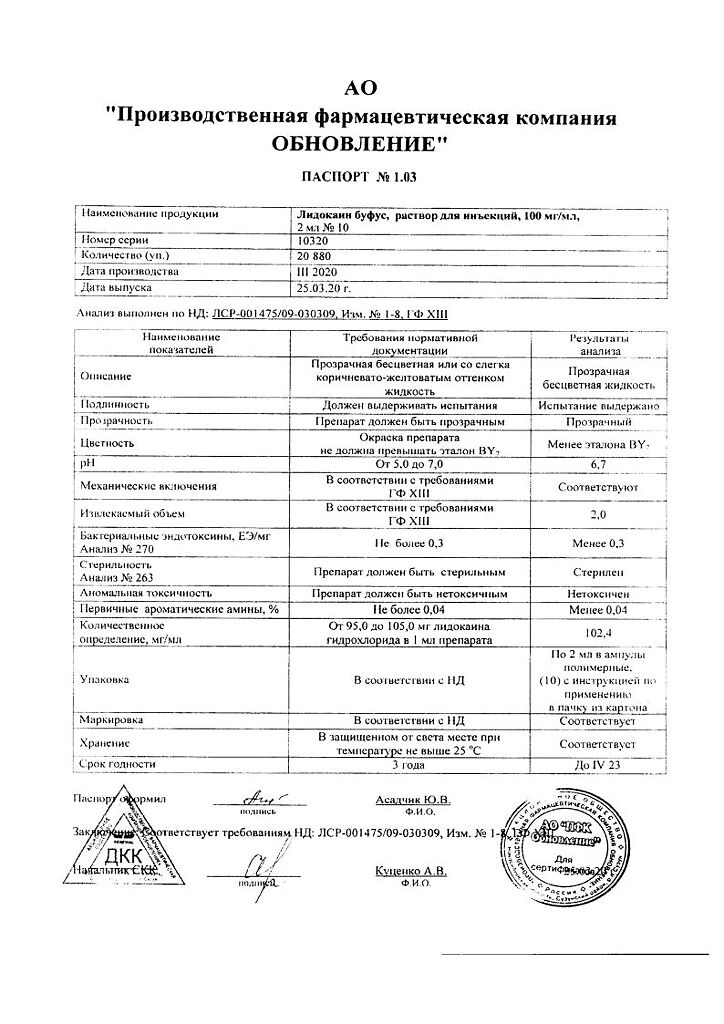
-
×


-
×

-
×


-
×


-
×

-
×


-
×

-
×

-
×


-
×

Subtotal: €469.21