No products in the cart.
Detralex, 500 mg 60 pcs
€49.44 €41.20
Description
Detralex® has venotonic and angioprotective properties.
The drug reduces venous distensibility and venous stasis, reduces capillary permeability and increases their resistance. The results of clinical studies confirm the pharmacological activity of the drug with respect to indicators of venous hemodynamics.
A statistically significant dose-dependent effect of the drug Detralex® has been demonstrated for the following venous plethysmographic parameters: venous capacity, venous distensibility, and time of venous emptying. The optimal dose/effect ratio is observed with the administration of 2 tablets.
Detralex® increases venous tone: venous occlusion plethysmography has been shown to decrease venous emptying time. In patients with signs of severe microcirculatory disturbances a statistically significant increase in capillary resistance as assessed by angiostereometry after therapy with Detralex® was observed (statistically significant) compared to placebo.
Therapeutic efficacy of Detralex® has been proven in the treatment of chronic diseases of lower limb veins and in the treatment of hemorrhoids.
Pharmacokinetics
The main excretion of the drug is in the feces. About 14% of the administered amount of the drug is excreted with urine on average.
The T1/2 is 11 hours.
The drug undergoes active metabolism, which is confirmed by the presence of phenolic acids in the urine.
Indications
Indications
The drug Detralex® is indicated for use in adults for the treatment of symptoms of chronic venous diseases (elimination and relief of symptoms).
• Treatment of symptoms of venous-lymphatic insufficiency:
– pain;
– spasms of the lower extremities;
– feeling of heaviness and fullness in the legs;
– “fatigue” of the legs.
• Treatment of manifestations of venous-lymphatic insufficiency:
– swelling of the lower extremities;
– trophic changes in the skin and subcutaneous tissue;
– venous trophic ulcers of the legs.
• Symptomatic treatment of acute and chronic hemorrhoids.
If there is no improvement or you feel worse during the course of use
drug, you need to consult a doctor.
Pharmacological effect
Pharmacological effect
Detralex® has venotonic and angioprotective properties.
The drug reduces the distensibility of veins and venous stagnation, reduces capillary permeability and increases their resistance. The results of clinical studies confirm the pharmacological activity of the drug in relation to indicators of venous hemodynamics.
A statistically significant dose-dependent effect of the drug Detralex® was demonstrated for the following venous plethysmographic parameters: venous capacity, venous distensibility, venous emptying time. The optimal dose/effect ratio is observed when taking 2 tablets.
Detralex® increases venous tone: using venous occlusion plethysmography, a decrease in the time of venous emptying was shown. In patients with signs of severe microcirculation disorders, there is a (statistically significant) increase in capillary resistance, assessed by angiostereometry, compared with placebo, after therapy with Detralex®.
The therapeutic effectiveness of the drug Detralex® has been proven in the treatment of chronic diseases of the veins of the lower extremities, as well as in the treatment of hemorrhoids.
Pharmacokinetics
The main excretion of the drug occurs in the feces. On average, about 14% of the administered amount of the drug is excreted in the urine.
T1/2 is 11 hours.
The drug undergoes active metabolism, which is confirmed by the presence of phenolic acids in the urine.
Special instructions
Special instructions
Driving vehicles and working with machinery
The drug Detralex® does not affect or has no significant effect on the ability to drive vehicles and machines.
Active ingredient
Active ingredient
Purified micronized flavonoid fraction (diosmin, flavonoids in terms of hesperidin)
Composition
Composition
The active ingredient is a purified micronized flavonoid fraction (diosmin + flavonoids in terms of hesperidin).
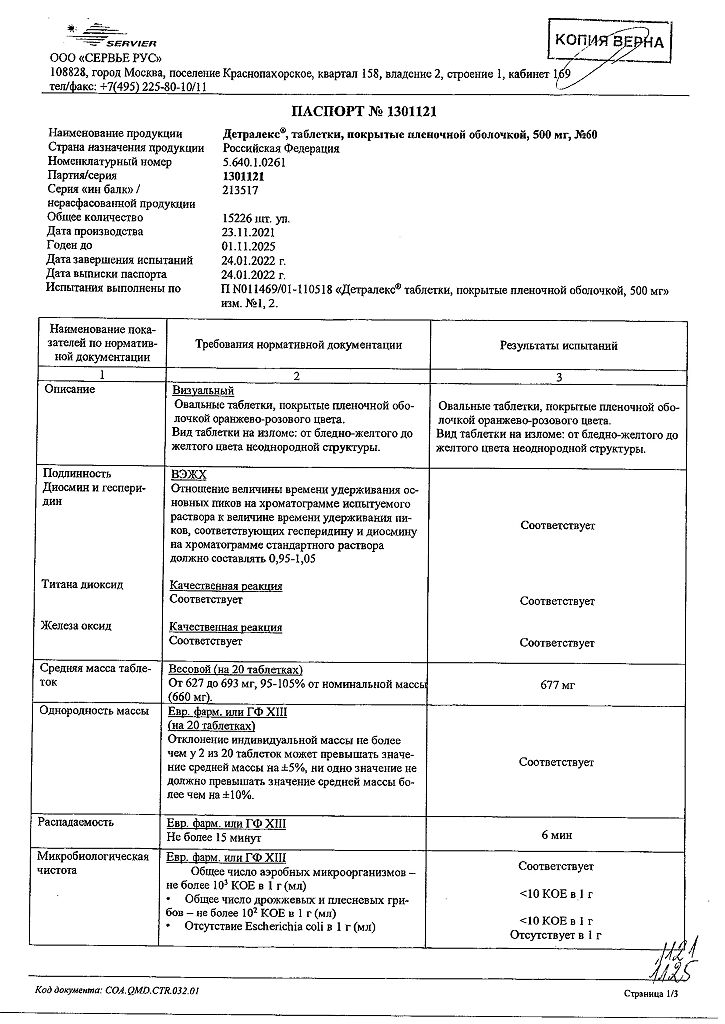
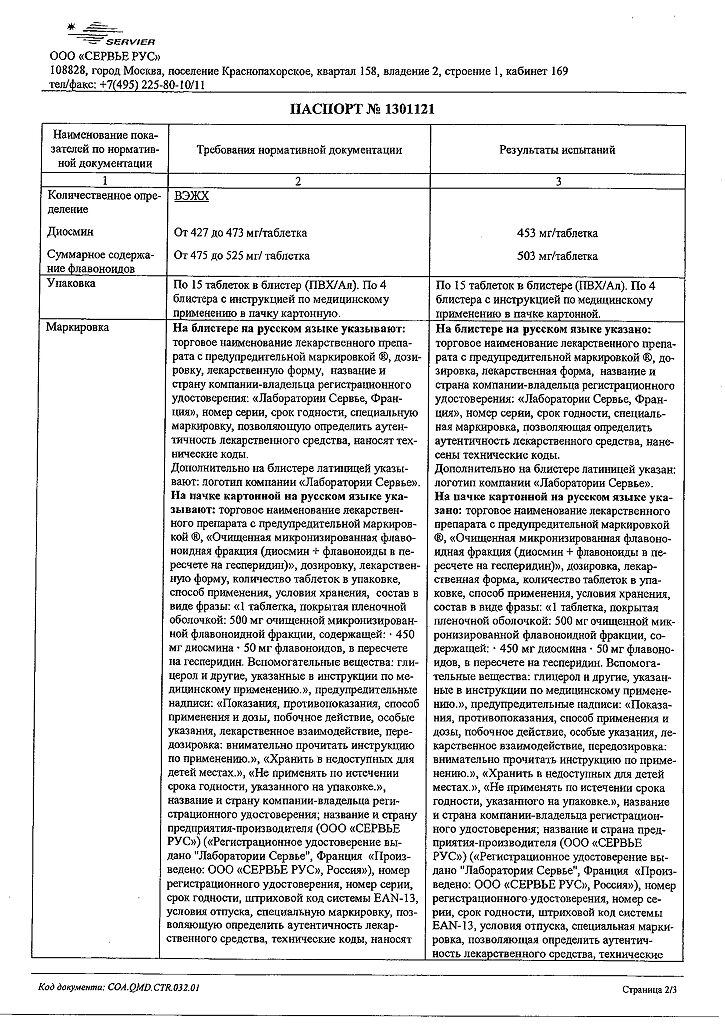
Each film-coated tablet contains 500 mg of purified micronized flavonoid fraction, consisting of diosmin (90%) 450 mg and flavonoids, calculated as hesperidin (10%) 50 mg.
Other ingredients (excipients) are: purified water, gelatin, magnesium stearate, microcrystalline cellulose, sodium carboxymethyl starch, talc, macrogol 6000, sodium lauryl sulfate, premix for film coating
orange-pink color, consisting of: glycerol, magnesium stearate, macrogol 6000, hypromellose, red iron oxide dye, titanium dioxide, yellow iron oxide dye.
Pregnancy
Pregnancy
If you are pregnant or breastfeeding, think you may be pregnant, or are planning to become pregnant, consult your doctor or pharmacist before taking this drug.
Pregnancy
As a precautionary measure, it is preferable not to take Detralex® during pregnancy.
Breastfeeding
Breastfeeding is not recommended during treatment as it is not known whether the drug passes into breast milk.
Contraindications
Contraindications
Do not take Detralex® if you are allergic to the purified micronized flavonoid fraction or any of the other components of the drug (listed in section 6 of the leaflet).
Special instructions and precautions
Before taking Detralex®, consult your doctor or pharmacist.
Tell your doctor if you have any of the following conditions:
In the treatment of hemorrhoids, the use of Detralex® does not replace the specific treatment of other diseases of the rectum and anal canal. When using the drug on your own, do not exceed the maximum duration and recommended doses,
specified in section 3 “Taking Detralex®”. If symptoms of hemorrhoids persist after the recommended course of therapy, you should be examined by a proctologist, who will select further therapy.
In the presence of venous circulation disorders, the maximum effect of treatment is ensured by a combination of therapy with additional therapeutic and preventive measures/healthy (balanced) lifestyle: it is advisable to avoid
long exposure to the sun, long periods of standing, and reduction of excess body weight is also recommended. Walking and, in some cases, wearing special stockings helps improve blood circulation.
Children and teenagers
Do not give the drug to children aged 0 to 18 years, since the safety and effectiveness of Detralex® in children and adolescents have not been established.
No data available.
Side Effects
Side Effects
Like all medicines, Detralex® can cause side effects, although not everyone gets them.
Stop taking Detralex® and seek immediate medical attention if any of the following signs of an allergic reaction occur with an unknown frequency (frequency cannot be determined based on available data):
• wheezing, swelling of the face or tongue, intense itching or severe skin rash (angioedema).
Other possible adverse reactions that may occur when taking Detralex®:
Common (may affect up to 1 in 10 people):
• diarrhea;
• feeling of pain or discomfort (heaviness, feeling of fullness, fullness, early satiety) in the abdomen (dyspepsia);
• nausea;
• vomiting.
Uncommon (may affect up to 1 in 100 people):
• inflammatory disease of the colon mucosa (colitis).
Rare (may affect up to 1 person in 1000):
• dizziness;
• headache;
• general malaise;
• skin rash;
• skin itching;
• urticaria.
Unknown (frequency cannot be determined based on available data):
• abdominal pain,
• isolated swelling of the face, lips, eyelids.
Interaction
Interaction
Tell your doctor or pharmacist if you are taking, have recently taken, or may start taking any other medications.
Storage conditions
Storage conditions
Keep the drug out of the reach of children and so that the child cannot see it.
Do not take the drug after the expiration date (shelf life) indicated on the carton and on each blister. The expiration date is the last day of the given month.
Store at a temperature not exceeding 30 °C.
Do not throw the drug into the sewer. Ask your pharmacist how to dispose of (destroy) a drug that is no longer needed. These measures will protect the environment.
Shelf life
Shelf life
4 years
Manufacturer
Manufacturer
Servier/Servier Rus LLC, Russia
Additional information
| Shelf life | 4 years |
|---|---|
| Conditions of storage | No special storage conditions required |
| Manufacturer | Servier Rus LLC, Russia |
| Medication form | pills |
| Brand | Servier Rus LLC |
Other forms…
Related products
Buy Detralex, 500 mg 60 pcs with delivery to USA, UK, Europe and over 120 other countries.