No products in the cart.
Coldrex HotRem, menthol and honey lemon 5 g sachet 10 pcs
€11.01 €9.17
Out of stock
(E-mail when Stock is available)
Description
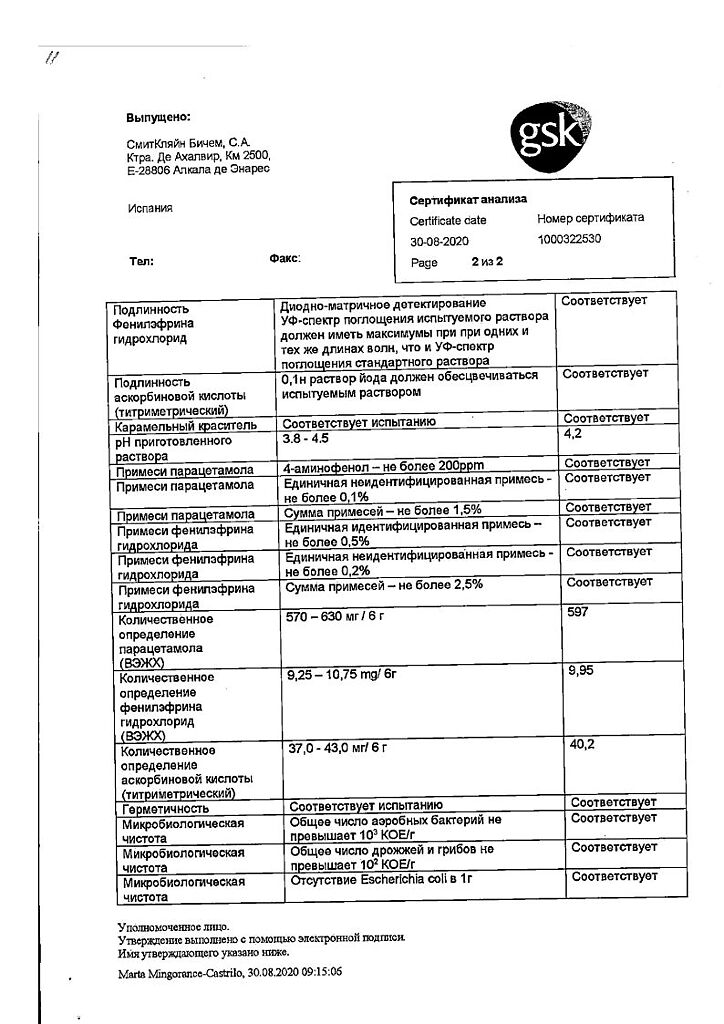
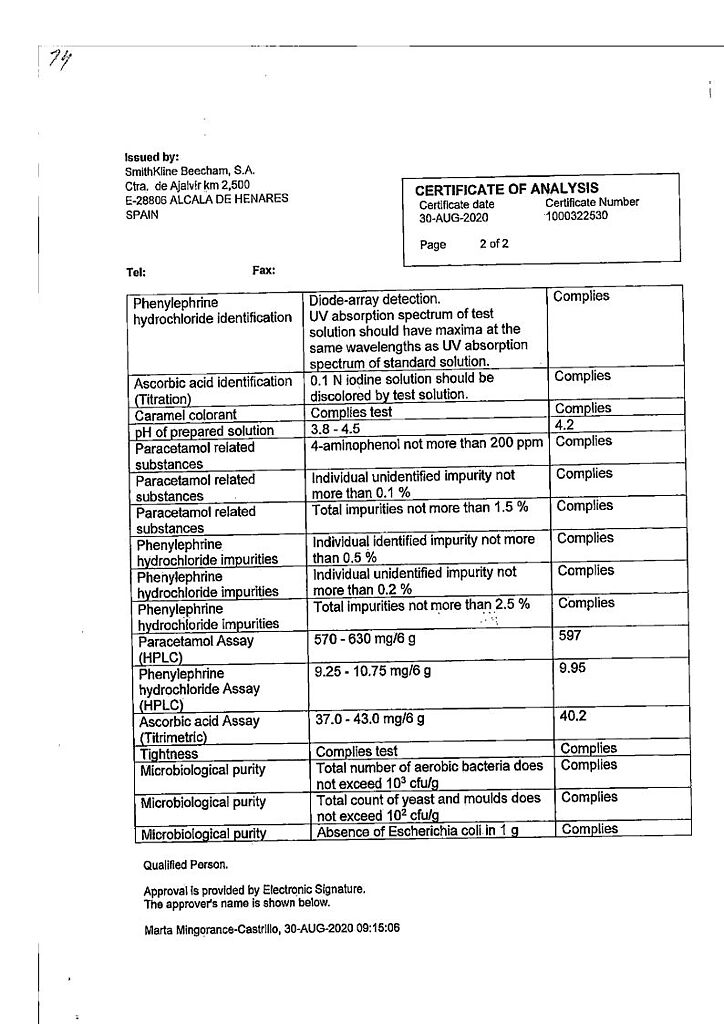
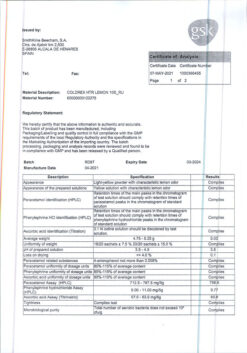
Paracetamol is an analgesic and antipyretic. Its mechanism of action is presumably the inhibition of prostaglandin synthesis, mainly in the CNS.
Paracetamol has almost no effect on the synthesis of prostaglandins in the peripheral tissues, it does not alter the water-electrolyte metabolism and does not damage the mucous membrane of the gastrointestinal tract.
Phenylephrine hydrochloride is a sympathomimetic agent intended to stimulate adrenoreceptors (primarily α-adrenoreceptors), which leads to reduction of nasal mucosa edema and facilitation of nasal breathing.
Ascorbic acid (vitamin C) replenishes the increased demand for vitamin C during “colds” and flu, especially during the initial stages of the disease.
The components of the drug do not cause drowsiness and do not disturb concentration.
Indications
Indications
How to take, the dosage
How to take, the dosage
Interaction
Interaction
Special Instructions
Special Instructions
Synopsis
Synopsis
Contraindications
Contraindications
Overdose
Overdose
Pregnancy use
Pregnancy use
Additional information
| Weight | 0.083 kg |
|---|---|
| Shelf life | 2 years. |
| Conditions of storage | The drug should be kept out of reach of children at a temperature not exceeding 30 ° C. |
| Manufacturer | SmithKlein Beecham S.A., Spain |
| Medication form | powder |
| Brand | SmithKlein Beecham S.A. |
Other forms…
Related products
Buy Coldrex HotRem, menthol and honey lemon 5 g sachet 10 pcs with delivery to USA, UK, Europe and over 120 other countries.