No products in the cart.
Ciprofloxacin Reneval, eye drops 0.3% 10 ml
€2.04 €1.70
Description
Ciprofloxacin is a broad-spectrum antimicrobial from the group of fluoroquinolones. It has a bactericidal effect. The drug inhibits bacterial enzyme DNA-gyrase, as a result DNA replication and synthesis of bacterial cell proteins are disrupted. Ciprofloxacin acts both on multiplying microorganisms and on those which are in the quiescent phase.
The following bacteria are sensitive to Ciprofloxacin:
Gram-negative aerobic bacteria: Escherichia coli, Salmonella spp, Shigella spp, Citrobacter spp, Klebsiella spp, Enterobacter spp, Proteus mirabilis, Proteus vulgaris, Serratia marcescens, Hafnia alvei, Edwardsiella tarda, Providencia spp, Morganella mordarii, Vibrio spp, Yersinia spp.
Other Gram-negative bacteria: Haemophilus spp, Pseudomonas aeruginosa, Moraxella catarrhalis, Aeromona, spp, Pasteurella multocida, Plesiomonas shigelloides, Campylobacter jejuni, Neissenia spp.
Some intracellular pathogens: Legionella pneumophila, Brucella spp, Chlamydia trachomatis, Listeria monocytogenes, Mycobacterium tuberculosis, Mycobacterium kansasii, Mycobacterium aviumintracellulare.
Gram-positive aerobic bacteria: Staphylococcus spp. (S.haemolyticus, S.hominis, S.saprophyticus), Streptococcus spp. (St. pyogenes, St. agalactiae).
The sensitivity of the bacteria Streptococcus pneumoniae, Enterococcus faecalis is moderate. Most staphylococci resistant to methicillin are also resistant to Ciprofloxacin.
Corynebacterium spp, Bacteroides fragilis, Pseudomonas cepacia, Pseudomonas maltophilia are resistant to the drug.
Ureaplasma, Clostridium difficile.
The action of the drug against Treponema pallidum has not been studied sufficiently.
Indications
Indications
Eye infections, Conjunctivitis, Blepharitis, Inflammatory eye diseasesInfectious and inflammatory eye diseases (acute and subacute conjunctivitis, blepharitis, blepharoconjunctivitis, keratitis, keratoconjunctivitis, bacterial corneal ulcer, meibomitis, acute dacryocystitis, infective lesions after eye injury), pre- and postoperative prevention of infectious complications in ophthalmosurgery.
Active ingredient
Active ingredient
Ciprofloxacin
Composition
Composition
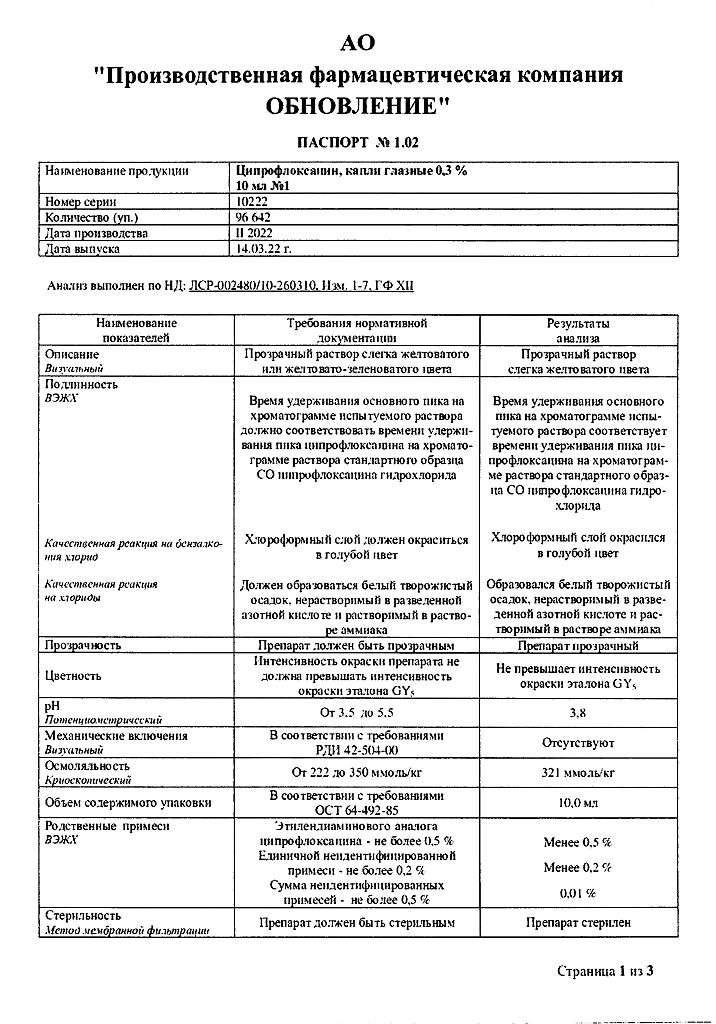
1 ml of the solution contains:
active ingredient:
ciprofloxacin hydrochloride
(ciprofloxacin) – 3 mg
excipients:
dimethoate (trilon B) – 0.5 mg
mannitol (mannitol) – 46 mg
sodium acetate – 0.5 mg
acetic acid – 0.0015 ml
benzalkonium chloride – 0.1 mg
water for injection – up to 1 ml
How to take, the dosage
How to take, the dosage
Topically. The drug is injected 1-2 drops into the conjunctival sac. Frequency of instillation depends on the severity of the inflammatory process.
In acute bacterial conjunctivitis, simple, scaly and ulcerous blepharitis, meibomitis the drug is prescribed 4 to 8 times a day depending on the severity of the disease. The course of treatment is from 5 to 14 days.
In case of keratitis the drug is prescribed in 1 drop at least 6 times a day; with positive effect the maximum course of treatment depending on the severity of corneal damage is about 2-4 weeks.
Interaction
Interaction
The interaction of Ciprofloxacin drops with other drugs during concomitant use has not been identified.
There is information that systemic use of some quinolones leads to increased plasma concentrations of theophylline, affects caffeine metabolism and enhances the effect of oral anticoagulants such as warfarin and its derivatives. a temporary increase in serum creatinine was reported in patients who received cyclosporine together with systemic use of ciprofloxacin.
Ciprofloxacin is incompatible with alkaline solutions.
Special Instructions
Special Instructions
The solution in the form of eye drops is not intended for intraocular injections.
When using other ophthalmic drugs the interval between their administration should
be at least 15 minutes.
Immediately after the use of the preparation the perception of vision blurring is possible which can cause difficulties while driving vehicles and operating mechanisms; it is recommended to start working (driving) 15 minutes after the use of the preparation.
During the period of the drug treatment it is not recommended to wear contact lenses.
After opening the bottle the drug should not be used for more than 14 days.
Contraindications
Contraindications
Hypersensitivity to the components of the drug, viral and fungal eye lesions, pregnancy, breast-feeding period, children under 1 year.
With caution:
The solution in the form of eye drops is not intended for intraocular injections.
When using other ophthalmic medications, the interval between injections should be at least 15 minutes.
Side effects
Side effects
Allergic reactions, itching and burning in the eye, mild pain and hyperemia of the conjunctiva, nausea, rarely – eyelid edema, photophobia, lacrimation, foreign body sensation in the eye, unpleasant taste in the mouth immediately after injection, reduced visual acuity, appearance of white crystalline precipitate in patients with corneal ulcer, keratitis, keratopathy, corneal infiltration, development of superinfection.
.
Overdose
Overdose
There are no data on overdose of the drug. In case of accidental ingestion there are no specific symptoms. Nausea, vomiting, diarrhea, headache, fainting, anxiety are possible.
Treatment: standard first aid measures, adequate intake of fluids into the body, creation of an acidic urine reaction to prevent crystalluria.
Similarities
Similarities
Ciprofloxacin, Ciprolet, Cifran, Ciprofloxacin, Cipromed, Cifran OD, Ciprofloxacin Ecocifol, Ciprofloxacin, Cipromed, Ciprofloxacin Teva
Additional information
| Weight | 0.014 kg |
|---|---|
| Shelf life | 3 years. |
| Conditions of storage | In a light-protected place at a temperature of 15 to 25 ° C. Do not freeze. Store out of the reach of children. |
| Manufacturer | Update PFC AO, Russia |
| Medication form | eye drops |
| Brand | Update PFC AO |
Related products
Buy Ciprofloxacin Reneval, eye drops 0.3% 10 ml with delivery to USA, UK, Europe and over 120 other countries.