No products in the cart.
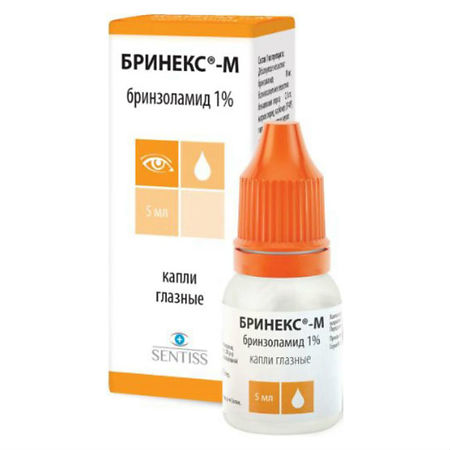
Brinex-M, eye drops 1% 5 ml
€23.43 €19.52
Description
Increased intraocular pressure is the main risk factor for optic nerve damage and narrowing of the visual field.
Brinzolamide is an inhibitor of carboanhydrase II. Due to inhibition of carboanhydrase II formation of bicarbonate ions is slowed down with subsequent reduction of sodium transport in the fluid which leads to decrease of intraocular fluid production in the ciliary body of the eye. This results in a decrease in intraocular pressure (IOP).
Pharmacokinetics:
When applied topically, brinzolamide penetrates the systemic bloodstream. Brinzolamide is adsorbed in erythrocytes as a result of selective binding. A metabolite, N-dezethyl brinzolamide, is formed, which also binds to carboangidase and accumulates in erythrocytes. In the presence of brinzolamide, the metabolite binds primarily to carboanhydrase I.
In plasma concentrations of brinzolamide and its metabolite are below the limit of quantification (<10 ng/ml). The elimination half-life is 111 days. Binding to plasma proteins is about 60%. Brinzolamide is mainly excreted unchanged in the urine. The main metabolite (N-dezethylbrinzolamide) and low concentrations of other metabolites (N-desmethoxypropyl and O-desmethyl) are also detected in the urine.
Indications
Indications
Lowering intraocular pressure in:
- open-angle glaucoma,
- ophthalmohypertension.
As monotherapy in patients who do not respond to β-blockers or in patients for whom β-blockers are contraindicated.
As adjunctive therapy to β-blockers or prostaglandin analogues.
Active ingredient
Active ingredient
Brinzolamide
Composition
Composition
Composition of 1 ml of the drug:
Active ingredient: brinzolamide 10 mg
Excipients:
benzalkonium chloride 0.1 mg,
dinatrium edetate dihydrate 0.1 mg,
sodium chloride 2.5 mg,
tiloxapol 0.25 mg,
mannitol 33.0 mg,
carbomer (974P) 4.2 mg,
5M sodium hydroxide solution to pH 7.5,
1M hydrochloric acid solution to pH 7.5,
water for injection to 1 ml.
How to take, the dosage
How to take, the dosage
Topically. Shake the bottle for 15 seconds before use.
Put 1 drop in the conjunctival sac twice a day.
The dose should not exceed 1 drop in the affected eye three times a day.
In order to decrease systemic absorption of the drug it is recommended to squeeze tear ducts at the inner corner of the eye or close eyelids for 2 minutes. As a result the risk of systemic adverse reactions is reduced and local activity is increased.
To prevent contamination of the drip tip and the suspension it is necessary to be careful not to touch the eyelids, the surrounding areas or other surfaces with the drip tip of the bottle. The vial should be kept tightly closed.
If changing to another ophthalmic anti-glaucoma drug and switching to brinzolamide, the other drug should be discontinued and brinzolamide started the next day.
If more than one topical ophthalmic drug is used, the drugs should be injected at least 5 minute intervals. Eye ointments should be applied last.
If a dose is missed, the next dose should be taken at the scheduled time.
Special patient groups
Older age
No dose adjustment is required.
Renal and hepatic impairment
Studies of brinzolamide eye drops in patients with hepatic impairment have not been conducted, so the drug is not recommended for use in such patients.
Studies of the use of brinzolamide eye drops in patients with acute renal failure (creatinine clearance <30 ml/min) or in patients with hyperchloremic acidosis have not been conducted. Since brinzolamide and its main metabolite are mainly excreted by the kidneys, brinzolamide is contraindicated in these patients (see section Contraindications).
Pediatric use
It is not recommended for use in children under 18 years old, since the safety and effectiveness of the drug in children and adolescents has not yet been established.
Interaction
Interaction
Specific studies on the interaction of Brinex®-M eye drops with other drugs have not been conducted.
Simultaneous use with oral carboanhydrase inhibitors is not recommended because of the possibility of increased systemic adverse reactions.
Salicylates at high doses increase the risk of systemic adverse reactions.
In clinical trials, brinzolamide was used concomitantly with prostaglandin analogues and timolol ophthalmic drugs without evidence of adverse interactions. Interactions between brinzolamide and myotics or adrenergic agonists during co-therapy of glaucoma have not been evaluated.
Brinzolamide is a carboanhydrase inhibitor and, although administered topically, is absorbed systemically. Acid-base imbalance has been reported when combined with oral carboanhydrase inhibitors. Assessment of interaction potential is necessary.
Cytochrome P-450 isoenzymes involved in metabolism of brinzolamide: CYP3A4 (major), CYP2A6, CYP2C8 and CYP2C9. CYP3A4 inhibitors such as ketoconazole, itraconazole, clotrimazole, ritonavir and troleandomycin are expected to inhibit the metabolism of brinzolamide by the CYP3A4 isoenzyme.
Caution should be exercised when co-administering brinzolamide with CYP3A4 inhibitors. Accumulation of brinzolamide is unlikely because it is excreted mainly by the kidneys. Brinzolamide is not an inhibitor of cytochrome P-450 isoenzymes.
Special Instructions
Special Instructions
The bottle should be shaken before use.
The bottle should be closed after each use.
Do not touch the pipette tip to any surface. The drug contains benzalkonium chloride preservative which can be absorbed by the contact lenses. The lenses shall be taken off and put back on not earlier than 15-20 minutes after instillation of the drug.
Systemic effects
Brinzolamide is a sulfonamide and carbohydrase inhibitor of local use but can be absorbed systemically. Sensitization to sulfonamides may develop if the drug is repeatedly prescribed in violation of the instructions for its use.
In case of serious adverse reactions or with signs of hypersensitivity the drug should be discontinued.
Acid-base balance disruption has been reported when combined with oral carboanhydrase inhibitors.
Brinzolamide has not been studied in premature infants (gestational age less than 36 weeks) or in children less than one week of age.
Patients with abnormalities or significant renal tubular immaturity may be treated with brinzolamide only after careful risk/benefit assessment, as there is a risk of metabolic acidosis.
Oral carboanhydrase inhibitors may impair the ability to perform tasks requiring mental activity and/or physical coordination.
Concomitant therapy
There is a possibility of increasing the known systemic effects of carboanhydrase inhibitors in patients when oral carboanhydrase inhibitor and brinzolamide are used together. Their combined use has not been studied and is not recommended (See section Interaction).
Initial evaluation of the combined use of brinzolamide with timolol during adjective therapy of glaucoma was performed. In addition, the IOP-lowering effects of brinzolamide during coadministration with the prostaglandin analog hervoprost were studied. There are no long-term data on the use of brinzolamide as an adjunct to hervoprost therapy.
There is limited experience with brinzolamide in the treatment of patients with pseudoexfoliative glaucoma or pigmentary glaucoma. Caution should be exercised when treating such patients, and close monitoring of intraocular pressure (IOP) is recommended.
Brinzolamide has not been studied in patients with narrow angle glaucoma and is not recommended for use in such patients.
The possible role of brinzolamide in corneal endothelial function has not been investigated in patients with corneal disorders (especially in patients with low endothelial cell counts).
In particular, the use of brinzolamide in patients who wear contact lenses has not been studied; close monitoring of these patients when using brinzolamide is recommended because carboanhydrase inhibitors can affect corneal hydration and wearing contact lenses may increase corneal risk. Close monitoring of patients with corneal lesions, such as patients with diabetes or corneal dystrophy, is recommended.
There is evidence that benzalkonium chloride, which is commonly used as a preservative in ophthalmic products, causes pitting keratopathy and/or toxic ulcerative keratopathy.
Because Brinex®-M contains benzalkonium chloride, close monitoring is required when the drug is used frequently or prolonged in patients with dry eye syndrome or corneal damage.
Benzalkonium chloride can also cause eye irritation and is known to discolor soft contact lenses. Contact of the drug with soft contact lenses should be avoided. Patients should be advised to remove contact lenses before using Brinex®-M and wait at least 15 minutes after administering the dose before putting on contact lenses.
Potential withdrawal syndrome after discontinuing brinzolamide treatment has not been studied; IOP-lowering effects are expected to last 5-7 days.
Influence on the ability to drive vehicles and machines:
If the patient’s vision is temporarily reduced after using the drug, it is not recommended to drive vehicles and engage in activities that require increased attention and reaction until it is restored.
Contraindications Cautions:
Contraindications
Hypersensitivity to the active ingredient or any of the excipients;
Hypersensitivity to sulfonamides;
severe renal insufficiency;
hyperchloremic acidosis.
Caution should be exercised when using the drug in patients with risk of renal failure because of the possible development of metabolic acidosis.
The effect of brinzolamide on corneal endothelial function in patients with corneal disorders (especially in patients with low endothelial cell counts) has not been studied. Therefore, close monitoring of these patients is recommended when using brinzolamide.
Caution should be exercised when treating patients with pseudoexfoliative glaucoma or pigmentary glaucoma, and close monitoring of IOP is recommended.
The use of the drug has not been studied in patients with closed-angle glaucoma, with severe liver disease, so this patient should be prescribed with caution.
.
Side effects
Side effects
The frequency of adverse reactions listed below is defined using the following frequency categories: very common (≥1/10); common (≥1/100 and <1/10); infrequent (≥1/1000 and <1/100); rare (≥1/10000 and <1/1000); very rare (<1/10000); with unknown frequency (which cannot be estimated based on available data). In each frequency group, adverse reactions are presented in order of frequency.
Data on adverse reactions were obtained from clinical trials and from post-registration spontaneous reports.
Infections and infections:
Infrequent: nasopharyngitis, pharyngitis, sinusitis.
With unknown frequency: rhinitis.
On the hematopoietic system:
Infrequent: decrease in the number of red blood cells, increase in plasma chloride.
Immune system:
With unknown frequency: hypersensitivity.
Metabolism:
With unknown frequency: decreased appetite.
Mental disorders:
Infrequently: apathy, depression, depressed mood, decreased libido, nightmares, nervousness.
Rarely: insomnia.
Nervous system disorders:
Infrequent: motor dysfunction, amnesia, dizziness, paresthesia, headache.
Rarely: memory disturbances, somnolence.
With unknown frequency: tremor, hypoesthesia, aguesia.
Visual organ:
Frequently: blurred vision, eye irritation, eye pain, sensation of a foreign body in the eye, eye hyperemia.
Infrequent: Corneal erosion, keratitis, pitting keratitis, keratopathy, ocular accumulation, corneal staining, corneal epithelial defect, corneal epithelial integrity disorders, blepharitis, itching of the eye, conjunctivitis, conjunctival edema, meibomitis, photophobia, dry eye syndrome, allergic conjunctivitis, pterygium, sclera pigmentation, asthenopia, eye discomfort, unusual feeling in the eye, dry keratoconjunctivitis, subconjunctival cysts, conjunctival injection, itching of eyelids, discharge from the eye, crust on the eyelid edges, lacrimation, pigment accumulation in the cornea during diagnostic tests to confirm corneal epithelial integrity disorders.
Rarely: corneal edema, diplopia, decreased visual acuity, photopsia, hypoesthesia, periorbital edema, increased intraocular pressure, changes in the ratio of excavation diameters to the optic disc diameter.
With unknown frequency: corneal disorders, visual impairment, allergic eye manifestations, madarosis, eyelid disorders, eyelid erythema.
Hearing organ and labyrinth:
Rarely: ringing in the ears.
With unknown frequency: dizziness.
Cardiovascular system disorders:
Infrequent: cardiorespiratory distress syndrome, bradycardia, palpitations.
Rarely: angina pectoris, impaired heart rate.
With unknown frequency: arrhythmia, tachycardia, hypertension, increased blood pressure, decreased blood pressure, increased heart rate.
Respiratory, chest and mediastinal disorders:
Infrequently: shortness of breath, nasal bleeding, oropharyngeal pain, pharyngeal pain, throat irritation, upper respiratory cough syndrome, runny nose, sneezing.
Rarely: bronchial hyperactivity, feeling of congestion in the upper respiratory tract, sinus mucosa swelling, nasal congestion, cough, nasal dryness.
With unknown frequency: bronchial asthma.
Gastrointestinal tract:
Frequently: dysgeusia.
Infrequent: esophagitis, diarrhea, nausea, vomiting, dyspepsia, upper abdominal pain, abdominal discomfort, stomach discomfort, flatulence, frequent bowel emptying, gastrointestinal disorders, decreased sensitivity in the mouth, paresthesia in the mouth, dry mouth.
Hepatobiliary system disorders:
With unknown frequency: change of liver function test results.
Skin and subcutaneous fat:
Infrequent: rash, maculopapular rash, feeling of skin tightness;
Rarely: Urticaria, hair loss, generalized itching;
With unknown frequency: dermatitis, erythema.
Musculoskeletal system and connective tissue:
Infrequent: back pain, muscle cramps, myalgia.
With unknown frequency: arthralgia, pain in the extremities.
Renal and urinary tract function:
Infrequent: pain in the renal area.
With unknown frequency: pollakiuria.
Reproductive system disorders:
Infrequent: erectile dysfunction.
General disorders:
Infrequent: pain, chest discomfort, increased fatigue, unusual sensations.
Rarely: chest pain, anxiety, asthenia, irritability.
With unknown frequency: peripheral edema, malaise.
Injuries, poisoning and complications during instillation
Infrequent: sensation of foreign body in the eye.
Description of individual adverse reactions:
Dysgeusia (bitter or unusual taste in the mouth after instillation of the drug) was the most common systemic adverse reaction associated with the use of brinzolamide during clinical trials. This is probably due to the passage of the eye drops into the nasopharynx through the nasopharyngeal canal.
Nasolacral occlusion or gentle eyelid closure after instillation may help reduce the frequency of this effect (see Dosage and administration).
Brinzolamide is a sulfonamide and a carboanhydrase inhibitor with systemic absorption.
Unwanted gastrointestinal and nervous system reactions as well as hematologic, renal, and metabolic adverse effects are usually associated with the systemic action of carboanhydrase inhibitors. A similar type of adverse reactions that may be associated with oral carboanhydrase inhibitors may also occur with topical administration.
No unexpected adverse reactions have been observed when using brinzolamide in conjunction with hervoprost. Adverse reactions observed during co-therapy were related to each of the active substances.
Overdose
Overdose
There are no reports of overdose symptoms when administered topically. When administering the drug orally there may be electrolyte imbalance, development of acidosis, as well as disorders of the nervous system.
The electrolyte levels (especially potassium) and pH of the blood should be monitored.
Pregnancy use
Pregnancy use
Fertility
According to the results of animal studies no data have been obtained on the effect of brinzolamide on fertility. No studies have been conducted to evaluate the effect of brinzolamide solution for topical ophthalmic use on fertility in humans.
Pregnancy
There are no reliable data on the use of brinzolamide in pregnant women. Animal studies have reported reproductive toxicity after systemic administration.
Brinzolamide is not recommended for use during pregnancy and by women of childbearing age who do not use contraception.
Breastfeeding
It is unknown whether brinzolamide / its metabolites are excreted into the breast milk after instillation into the conjunctival cavity. Animal studies have demonstrated excretion of minimal levels of brinzolamide into breast milk after oral administration.
Due to the possibility of side effects in the infant during treatment of the mother with the drug, a decision should be made to stop breastfeeding or to discontinue treatment, taking into account the importance of treatment to the mother and the importance of breastfeeding to the child.
Similarities
Similarities
Azopt, Brinzopt
Additional information
| Weight | 0.034 kg |
|---|---|
| Shelf life | 2 years. |
| Conditions of storage | At a temperature of 4 to 30 ° C. Store out of the reach of children. |
| Manufacturer | Sentiss Pharma Pvt.Ltd, India |
| Medication form | eye drops |
| Brand | Sentiss Pharma Pvt.Ltd |
Related products
Buy Brinex-M, eye drops 1% 5 ml with delivery to USA, UK, Europe and over 120 other countries.