No products in the cart.
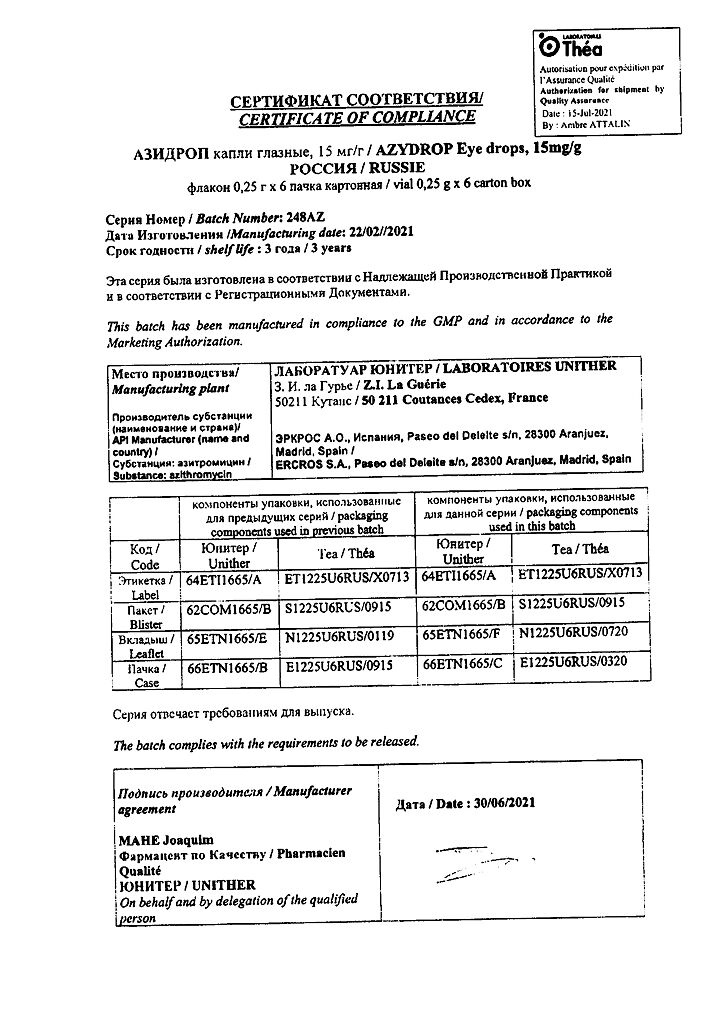
Azidrop, eye drops 15 mg/g 0.25 g 6 pcs
€21.76 €18.13
Description
An antimicrobial drug for topical use. Azithromycin is a second generation macrolide antibiotic of azalide group. It inhibits protein synthesis in bacteria by binding to 50S-subunit of ribosomes and prevents translocation of peptides.
The mechanism of resistance
There are three mechanisms of resistance for different types of bacteria to macrolides: due to modification of the target of action, modification of the antibiotic or due to active release of the antibiotic from the microbial cell by means of transport systems (efflux). Different efflux systems have been described for bacteria. An important efflux system for Streptococci is encoded by the mef gene and leads to macrolide-limited resistance (M-phenotype). Modification of the target of action controlled by erm encoded methylase (MLSB-phenotype) leads to cross-resistance to different classes of antibiotics.
Cases of cross-resistance to erythromycin, azithromycin, other macrolides and lincosamide and streptogramine B have been described for Streptococcus pneumoniae, β-haemolytic group A streptococci, Enterococcus spp. and Staphylococcus aureus, including methicillin-resistant S. aureus (MRSA). Constitutive mutants in inducibly resistant strains with erm(A) or erm(C) can be isolated in vitro at low frequencies of approximately 10-7COU in the presence of azithromycin.
Borderline value
The following are the minimum inhibitory concentrations (MICs) for microorganisms for these indications.
It should be noted that the borderline MIC values and spectrum of action presented below are for systemic use. These MICs cannot be used for topical treatment with the drug in the form of eye drops due to different concentrations and physicochemical conditions that may affect the overall activity of the drug at the site of action.
According to EUCAST (European Committee on Antibiotic Susceptibility Testing), the following MIC limit values are defined for azithromycin:
Haemophilus influenzaeS ≤ 0.12mg/L and R >4 mg/LMogachella catarrhalisS ≤ 0.5 mg/L and R>0.5 mg/LNeisseria gonorrhoeaeS ≤ 0.25 mg/L and R>0.5 mg/LStarhooloccus spp.S ≤ 1.0 mg/L and R>2.0 mg/LSthealthoccus pneumoniaeS ≤ 0.25 mg/L and R>0.5 mg/LSthealthoccus A, B, C, GS ≤ 0.25 mg/L and R>0.5 mg/L
EUCAST allows the use of erythromycin to determine the sensitivity of other specified bacterial species to azithromycin.
The incidence of acquired resistance for individual species may vary depending on the geographic region and over time. Therefore, it is advisable to have local information about resistance, especially when treating severe infections. Specialist advice should be sought, if necessary, when the local incidence of resistance is such that the efficacy of the drug, at least for some types of infection, is questionable.
Clinical trial data
- Thoracomatous conjunctivitis caused by Chlamydia trachomatis
. A randomized, double-blind, 2-month comparison study of Azidrop with oral administration of a single dose of azithromycin was conducted to treat active trachoma in 670 children aged 1-10 years. The primary efficacy variable was clinical cure at day 60, i.e., no active trachoma TF0 (according to the simplified WHO trachoma severity classification). At day 60, the rate of clinical cure with Azidrop, administered 1 drop 2 times daily for 3 days, was no lower (96.3%) than with oral azithromycin (96.6%).
The clinical efficacy of Azidrop, administered 1 drop 2 times daily for 3 days, for the treatment and prevention of trachoma in the entire population (from birth) in Northern Cameroon (112,000 subjects) was evaluated in a multicenter, open-label, noncomparative phase IV study. Treatment was administered in 3 periods of 1 year duration. The primary efficacy criterion was the prevalence of active trachoma, i.e., trachomatous follicular inflammation or marked trachomatous inflammation (TF+TI0 or TF+TI+). For the analysis, a clinical evaluation of trachoma was performed each year in 2,400 children aged ≥1 and < 10 years, selected by random cluster sampling. The prevalence of active trachoma (TF+TI0 or TF+TI+) was observed in 31.1% before instillation of Azidrop at “year 0” and decreased to 6.3% (year 1), 3.1% (year 2), and 3.1% (year 3). Overall, no serious adverse reactions to the study drug were observed in the population.
- Purulent bacterial conjunctivitis
A randomized, blinded comparative study of Azidrop, used 1 drop twice daily for 3 days, with tobramycin (eye drops, 0.3%), applied 1 drop every 2 h for 2 days and then 4 times/day for 5 days, to treat purulent bacterial conjunctivitis in 1,043 patients (ITT group), including 109 children under 11 years of age, 5 of whom were newborns (0 to 27 days), 38 infants, and toddlers (28 days to 23 months). According to the protocol, the population (n=471) (RR group), included 16 infants and toddlers and did not include newborns. The clinical trial was conducted in different regions of Europe, North Africa, and India. The primary efficacy variable represented clinical cure at day 9 in the RR group and was defined as 0 points for bulbar conjunctival injection and for purulent discharge. At day 9, the rate of clinical cure with azidrop (87.8%) was no lower than with tobramycin (89.4%). The microbiological efficacy of azithromycin was comparable to that of tobramycin.
- Children’s population
The efficacy and safety of Azidrop in children and adolescents under 18 years of age were demonstrated in a randomized, investigator-masked study comparing tobramycin in 282 study patients diagnosed with purulent bacterial conjunctivitis (including 148 patients in the 0-day-< 24-month subgroup). Patients received either Azidrop (1 drop 2 times daily for 3 days) or tobramycin, eye drops, 0.3% (1 drop every 2 h for 2 days and then 4 times/day for 5 days). The main criterion for efficacy was clinical cure of the more affected eye on day 3 in patients with positive bacteriologic test results on day 0. In patients using Azidrop, clinical cure of the more affected eye on day 3 was superior (47%) to that in patients using tobramycin(28%). At day 7, 89% of patients treated with Azidrop were completely cured, whereas among patients treated with tobramycin, there was a complete cure in 78% of cases.
There were no statistical differences between the treated groups with respect to bacteriological resolution at day 7. Azidrop, administered 1 drop 2 times daily for 3 days, was well tolerated in all age groups in a large group study of children.
The adverse reactions noted in children were also seen in adults; no new adverse reactions were noted in children. Moreover, no age-related clinical problems have been noted. The short duration of therapy with azithromycin (1.5% eye drops), the small number of instillations required, and the ease of instilling the drops in children have been accepted by both children and parents.
Pharmacokinetics
Azithromycin is not detectable in the blood of patients after instillation of Azidrop eye drops for the treatment of bacterial conjunctivitis at the recommended dose (detection limit: 0.0002 µg/ml).
Pharmacokinetic studies have only been performed in adults
Indications
Indications
Treatment of conjunctivitis caused by pathogens sensitive to the drug, incl. Chlamydia trachomatis, in adults and children over 1 year of age.
Pharmacological effect
Pharmacological effect
Antimicrobial drug for topical use. Azithromycin is a second generation macrolide antibiotic from the azalide group. Inhibits bacterial protein synthesis by binding to the 50S ribosomal subunit and preventing peptide translocation.
Mechanism of resistance
Three mechanisms of resistance to macrolides are noted for different types of bacteria: due to modification of the target of action, modification of the antibiotic, or due to the active release of the antibiotic from the microbial cell using transport systems (efflux). Various efflux systems have been described for bacteria. An important efflux system for streptococci is encoded by the mef gene and leads to macrolide-limited resistance (M phenotype). Modification of the target of action, controlled by the erm-encoded methylase (MLSB phenotype), leads to cross-resistance to different classes of antibiotics.
Cases of cross-resistance to erythromycin, azithromycin, other macrolides and lincosamide and streptogramin B have been described for Streptococcus pneumoniae, group A β-hemolytic streptococci, Enterococcus spp. and Staphylococcus aureus, including methicillin-resistant S. aureus (MRSA). Constitutive mutants in inducibly resistant strains with erm(A) or erm(C) can be isolated in vitro at low frequencies of approximately 10-7 CFU in the presence of azithromycin.
Borderline value
Below are the minimum inhibitory concentrations (MICs) for microorganisms for these indications.
It should be noted that the MIC breakpoints and spectrum of activity presented below are for systemic use. MIC data cannot be used in the case of topical treatment with the drug in the form of eye drops due to different concentrations and physicochemical conditions that may affect the overall activity of the drug at the site of action.
In accordance with EUCAST (European Committee on Antimicrobial Susceptibility Testing), the following MIC breakpoints are defined for azithromycin:
Haemophilus influenzaeS ≤ 0.12 mg/l and R >4 mg/lMoraxella catarrhalisS ≤ 0.5 mg/l and R>0.5 mg/lNeisseria gonorrhoeaeS ≤ 0.25 mg/l and R>0.5 mg/lStaphylococcus spp.S ≤ 1.0 mg/l and R>2.0 mg/lStreptococcus pneumoniaeS ≤ 0.25 mg/l and R>0.5 mg/lStreptococcus A, B, C, GS ≤ 0.25 mg/l and R>0.5 mg/l
EUCAST allows the use of erythromycin to determine the sensitivity of other species of specified bacteria to azithromycin.
The frequency of acquired resistance for individual species may vary depending on geographic region and time. Therefore, it is desirable to have local information on resistance, especially when treating severe infections. If necessary, specialist advice should be sought when the local incidence of resistance is such that the effectiveness of the drug, at least for some types of infection, is doubtful.
Clinical trial data
Trachomatous conjunctivitis caused by Chlamydia trachomatis
A randomized, double-blind, 2-month comparative study of Azidrop with single-dose oral azithromycin was conducted for the treatment of active trachoma in 670 children aged 1–10 years. The primary efficacy variable was clinical cure at day 60, i.e. absence of active trachoma TF0 (according to the simplified WHO classification of trachoma severity). On day 60, the frequency of clinical cure with Azidrop, used 1 drop 2 times a day for 3 days, was no lower (96.3%) than when taking azithromycin orally (96.6%).
The clinical efficacy of Azidrop, used 1 drop 2 times a day for 3 days, for the treatment and prevention of trachoma in the entire population (from birth) in Northern Cameroon (112,000 subjects) was assessed in a multicenter, open-label, non-comparative phase IV study. Treatment was carried out in 3 periods lasting 1 year. The primary criterion for effectiveness was the prevalence of active trachoma, i.e. trachomatous follicular inflammation or severe trachomatous inflammation (TF+TI0 or TF+TI+). For the analysis, clinical assessment of trachoma was performed each year on 2400 children aged ≥1 and <10 years selected by cluster random sampling. The prevalence of active trachoma (TF+TI0 or TF+TI+) was observed in 31.1% of cases before Azidrop instillation in "year 0" and decreased to 6.3% (1 year), 3.1% (2 year) and 3.1% (3 year). Overall, no serious adverse reactions to the study drug were observed in the population.
Purulent bacterial conjunctivitis
A randomized, blinded comparative study of Azidrop, used 1 drop 2 times a day for 3 days, with tobramycin (eye drops, 0.3%), used 1 drop every 2 hours for 2 days, and then 4 times a day for 5 days, was conducted for the treatment of purulent bacterial conjunctivitis in 1043 patients (ITT group), including 109 children in under 11 years of age, 5 of whom were newborns (0 to 27 days), 38 infants and toddlers (28 days to 23 months). The per-protocol population (n=471) (RR group) included 16 infants and toddlers and did not include neonates. The clinical study was conducted in different regions of Europe, North Africa and India. The primary efficacy variable was clinical cure on day 9 in the RR group and was defined as 0 for bulbar conjunctival injection and for purulent discharge. On day 9, the clinical cure rate with Azidrop (87.8%) was no lower than with tobramycin (89.4%). The microbiological effectiveness of azithromycin was comparable to that of tobramycin.
Children’s population
The efficacy and safety of Azidrop in children and adolescents under 18 years of age was demonstrated in a randomized, investigator-blinded study compared with tobramycin in 282 study patients diagnosed with purulent bacterial conjunctivitis (including 148 patients in the 0 days – < 24 months subgroup). Patients received either Azidrop (1 drop 2 times a day for 3 days) or tobramycin eye drops, 0.3% (1 drop every 2 hours for 2 days, and then 4 times a day for 5 days). The primary outcome measure was clinical cure of the more affected eye on day 3 in patients with positive culture results on day 0. In patients treated with Azidrop, clinical cure of the more affected eye on day 3 was superior (47%) compared to patients treated with tobramycin (28%). On day 7, 89% of patients treated with Azidrop were completely cured, while among patients treated with tobramycin, complete cure was observed in 78% of cases.
There were no statistical differences between treatment groups regarding bacteriological resolution at day 7. Azidrop, administered 1 drop 2 times a day for 3 days, was well tolerated in all age groups in a study of a large group of children.
Adverse reactions noted in children were also found in adults; no new adverse reactions were identified in children. Moreover, no age-related clinical problems were noted. The short duration of azithromycin therapy (eye drops 1.5%), the small number of instillations required, and the ease of instillation of drops in children were taken into account by both children and parents.
Pharmacokinetics
After instillation of Azidrop eye drops for the treatment of bacterial conjunctivitis at the recommended dose, azithromycin is not detected in the blood of patients (detection limit: 0.0002 μg/ml).
Pharmacokinetic studies were conducted in adults only
Special instructions
Special instructions
The drug should not be injected or swallowed, nor should it be administered by peri- or intraocular injection.
If an allergic reaction to the drug develops, treatment should be stopped.
When treating with other ophthalmic drugs simultaneously, Azidrop should be instilled last, 15 minutes after instillation of the other drug.
The patient should be informed that eye drops should not be instilled after completion of therapy on day 3, even if residual signs of bacterial conjunctivitis persist.
Patients suffering from bacterial conjunctivitis should not wear contact lenses.
Cases of fulminant hepatitis, which could potentially lead to life-threatening liver failure, have been reported with systemic use of azithromycin. When using the drug in ophthalmology, such a risk does not exist, since the systemic effect of the active substance is extremely insignificant.
Use in pediatrics
Comparative studies of the effectiveness and safety of the drug for trachomatous conjunctivitis in children under 1 year of age have not been conducted. However, taking into account the experience of clinical use in children of this age group with trachomatous conjunctivitis and taking into account the experience of treating newborns with purulent bacterial conjunctivitis with Azidrop, there are no safety concerns and differences in the pathological process that would exclude the use of Azidrop eye drops in children under 1 year of age for this indication.
According to current international recommendations for the treatment of diseases of the eye and genitourinary tract, which are highly likely to be transmitted to newborns, systemic therapy is necessary for non-trachomatous conjunctivitis caused by Chlamydia trachomatis and conjunctivitis caused by Neisseria gonorrhoeae.
In newborns and children under 3 months of age, systemic infections (such as pneumonia and bacteremia) caused by Chlamydia trachomatis may be accompanied by conjunctivitis. If such conditions are suspected, systemic therapy is necessary.
The drug is not intended for the prevention of bacterial conjunctivitis in newborns.
Impact on the ability to drive vehicles and operate machinery
The effect on the ability to drive vehicles and machinery has not been studied.
Transient blurred vision may occur after using eye drops. Patients who experience transient blurred vision after using eye drops are not recommended to drive vehicles or operate machinery until vision is restored.
Active ingredient
Active ingredient
Azithromycin
Composition
Composition
1 g of eye drops contains:
active ingredient:
azithromycin dihydrate 15 mg, which corresponds to the content of azithromycin 14.3 mg,
excipients:
medium chain triglycerides up to 1 g.
Pregnancy
Pregnancy
Since systemic exposure to azithromycin is negligible, no negative effects of the drug are expected during pregnancy. The use of the drug Azidrop, eye drops, in pregnant women is possible.
There is limited evidence that azithromycin is excreted in breast milk, but given the low doses and low systemic availability, the dose exposed to newborns is extremely small. Thus, the use of Azidrop during breastfeeding is acceptable.
Data from animal studies have not confirmed the effect of azithromycin on fertility in men and women. There are no results from human studies. Since the systemic exposure of azithromycin to the body is insignificant, the drug is not expected to affect fertility.
Contraindications
Contraindications
Individual hypersensitivity to azithromycin and other macrolide antibiotics, as well as components of the drug Azidrop; age less than 1 year.
Side Effects
Side Effects
Adverse reactions observed in clinical trials and post-marketing studies
From the immune system: uncommon (≥ 1/1000,
From the organ of vision: very often (≥ 1/10) – ocular discomfort (itching, burning, tingling) after instillation of the drug; often (≥1/100,
Adverse reactions observed in post-marketing studies
The inclusion of these adverse reactions is based on post-marketing data. Frequency is based on 3/X, where X is the total sample size summed across all relevant clinical studies, which at 3/879 results in the category “uncommon.”
From the immune system: uncommon (≥1/1000,
From the organ of vision: uncommon (≥1/1000,
The profile of adverse reactions in children corresponds to the adult population; no side effects were identified. The safety profile was also similar between pediatric groups.
Interaction
Interaction
Interaction studies of Azidrop with all specific drugs have not been conducted.
Due to the absence of detectable concentrations of azithromycin in blood plasma when instilled with Azidrop eye drops (see Pharmacokinetics section), interactions are not expected with any of the drugs that interact with azithromycin when administered orally.
Overdose
Overdose
There is no information on cases of overdose.
The single-use package contains azithromycin in sufficient quantities to treat both eyes, but not enough to cause adverse reactions following accidental intravenous administration or ingestion of the solution.
Storage conditions
Storage conditions
Store at a temperature not exceeding 25°C. Keep out of the reach of children.
Shelf life
Shelf life
18 months
Manufacturer
Manufacturer
Laboratory Unitere, France
Additional information
| Shelf life | 18 months |
|---|---|
| Conditions of storage | Store at a temperature not exceeding 25°C. Keep out of reach of children. |
| Manufacturer | Laboratoire Uniter, France |
| Medication form | eye drops |
| Brand | Laboratoire Uniter |
Related products
Buy Azidrop, eye drops 15 mg/g 0.25 g 6 pcs with delivery to USA, UK, Europe and over 120 other countries.