No products in the cart.
Alfagan R, eye drops 0.15% 5 ml
€1.00
Out of stock
(E-mail when Stock is available)
Description
Pharmgroup: Antiglaucoma drug.
Pharmacological action: Brimonidine is a selective alpha2-adrenomimetic with stimulating effect on alpha2-adrenoreceptors. When used in the dosage form of eye drops 0.15% maximum decrease in intraocular pressure (IOP) is achieved in 2 hours. The hypotensive effect of brimonidine is provided by reducing the formation and increasing the outflow of intraocular fluid through the uveoscleral pathway.
Indications
Indications
Open angle glaucoma.
Ocular hypertension (in monotherapy or in combination with other drugs that lower IOP).
Pharmacological effect
Pharmacological effect
Pharmaceutical group: Antiglaucoma drug.
Pharmaceutical action: Brimonidine is a selective alpha2-adrenergic agonist that has a stimulating effect on alpha2-adrenergic receptors. When used in the dosage form of eye drops 0.15%, the maximum reduction in intraocular pressure (IOP) is achieved after 2 hours. The hypotensive effect of brimonidine is achieved by reducing the formation and increasing the outflow of intraocular fluid along the uveoscleral pathway.
Special instructions
Special instructions
Use for liver dysfunction
With caution: liver failure.
Use for renal impairment
With caution: renal failure (creatinine clearance below 40 ml/min).
Impact on the ability to drive vehicles and operate machinery
Taking Alphagan R may be accompanied by episodes of weakness and drowsiness in some patients. If the patient’s work involves potentially hazardous activities, such as driving vehicles, he must be warned in advance about a possible decrease in concentration and speed of psychomotor reactions, and recommended to refrain from these types of activities.
Active ingredient
Active ingredient
Brimonidine
Composition
Composition
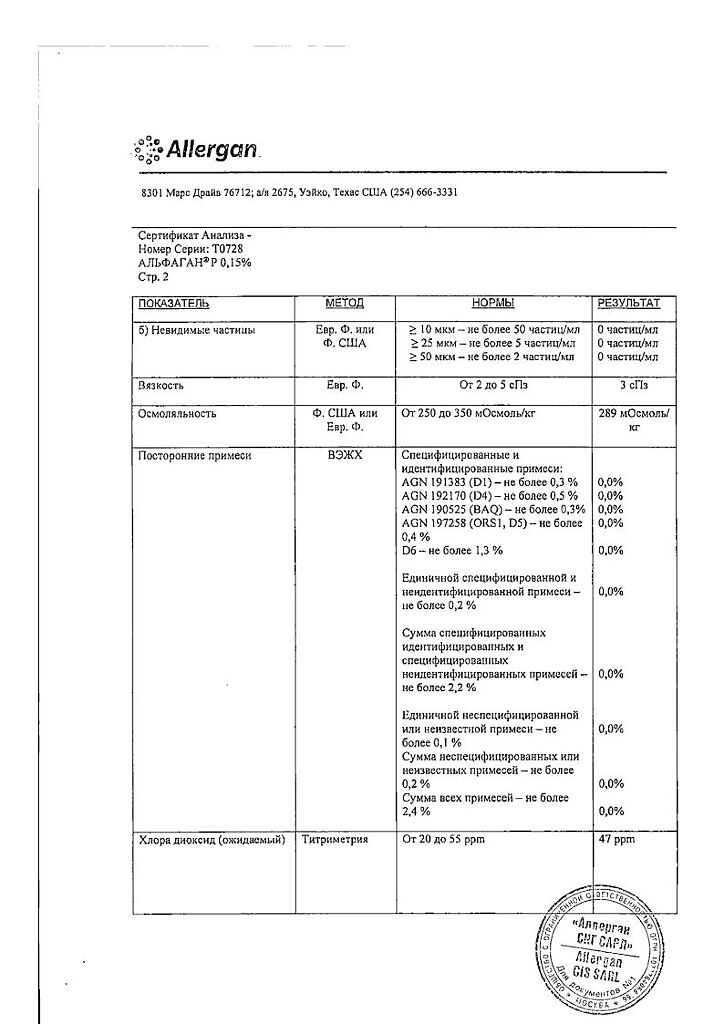
1 ml – brimonidine tartrate 1.5 mg
auxiliary components: stabilized oxy-chloro complex (sodium chloride, sodium chlorate, chlorine dioxide), sodium carmellose, sodium chloride, potassium chloride, calcium chloride dihydrate, magnesium chloride hexahydrate, boric acid, sodium tetraborate decahydrate, hydrochloric acid or sodium hydroxide, water.
Pregnancy
Pregnancy
Pregnancy
Preclinical studies did not reveal any effect on reproductive function.
However, it has been established that brimonidine penetrates the placental barrier and is found in small amounts in fetal blood plasma. No harmful effect on the fetus has been established.
There have been no controlled studies in pregnant women.
During pregnancy, ALPHAGAN R should be used with extreme caution, only in cases where the expected benefit to the mother significantly outweighs the possible risk to the fetus.
Lactation
Animal studies have shown that brimonidine tartrate passes into breast milk. Breastfeeding should be stopped while using the drug.
Use in children
Contraindicated in children under 2 years of age, with caution in children aged 2 to 7 years.
Contraindications
Contraindications
hypersensitivity to brimonidine and other components of the drug;
simultaneous therapy with MAO inhibitors;
children under 2 years of age, low body weight (up to 20 kg);
breastfeeding period.
With caution: orthostatic hypotension, heart failure, cerebrovascular diseases leading to cerebrovascular insufficiency, renal failure (creatinine clearance below 40 ml/min), liver failure, depression, Raynaud’s syndrome, thromboangiitis obliterans, children aged 2 to 7 years.
Side Effects
Side Effects
The frequency of side effects identified during the studies was assessed as follows: very often (>10%), often (>1% and 0.1% and 0.01% and
Clinical studies of the drug Alphagan R revealed the following side effects:
From the organ of vision: very often – allergic conjunctivitis, conjunctival hyperemia, itching of the mucous membrane of the eyes and skin of the eyelids; often – burning sensation, conjunctival folliculosis or follicular conjunctivitis, local allergic reactions of the eyes (including keratoconjunctivitis), blepharitis, blepharoconjunctivitis, blurred vision, cataracts, conjunctival edema, conjunctival hemorrhage, conjunctivitis, retention lacrimation, mucous discharge from the eyes, dryness and irritation of the mucous membrane of the eyes, pain, swelling of the eyelids, redness of the eyelids, sensation of a foreign body in the eyes, keratitis, eyelid lesions, photosensitivity, superficial punctate keratopathy, lacrimation, loss of visual fields, functional disorders of the vitreous, hemorrhages in the vitreous, floaters in the vitreous and decreased visual acuity; infrequently – corneal erosion, barley;
From the side of the central nervous system: often – headache, drowsiness, insomnia, dizziness.
From the cardiovascular system: often – increased blood pressure; infrequently – decreased blood pressure.
From the respiratory system: often – bronchitis, cough, shortness of breath; uncommon – dry nasal mucosa, apnea.
From the gastrointestinal tract: often – gastrointestinal disorders – dyspepsia, dryness of the oral mucosa.
From the skin and subcutaneous fat: often – rash.
Infectious and parasitic diseases: often – flu-like syndrome, infectious disease (chills and respiratory infection), rhinitis, sinusitis, incl. infectious.
Laboratory indicators: often – hypercholesterolemia.
Others: often – general allergic reactions, asthenia, fatigue; infrequently – a perversion of taste.
In children, the following were noted: apnea, bradycardia, decreased blood pressure, hypothermia, muscle hypotension.
In the post-marketing period, additional reports of the following side effects have been received:
From the organ of vision: frequency unknown – iritis, sicca keratoconjunctivitis, miosis;
From the central nervous system: depression;
From the cardiovascular system: bradycardia, tachycardia.
From the gastrointestinal tract: nausea.
From the skin and subcutaneous fat: local skin reactions (erythema, itching of the eyelids, swelling of the face, rash and vasodilation of blood vessels in the skin of the eyelids and face).
Interaction
Interaction
No studies have been conducted to study drug interactions with Alphagan R, however, with its simultaneous use, the possibility of enhancing the effect of drugs that depress the central nervous system (alcohol, barbiturates, opium derivatives, sedatives, general anesthetics) should be taken into account. Given the ability of drugs from the alpha-adrenergic agonist group to reduce blood pressure and heart rate, caution should be exercised when using antihypertensive drugs and cardiac glycosides simultaneously.
Due to the known decrease in the severity of the hypotensive effect of clonidine (alpha2-adrenergic agonist) when used together with tricyclic antidepressants, a possible decrease in the effectiveness of the drug Alphagan R cannot be ruled out during concomitant treatment with tricyclic antidepressants.
Alphagan R should be used with caution with tricyclic antidepressants, which may affect the metabolism of amines and their distribution in the vascular bed.
Overdose
Overdose
Symptoms: if the drug is accidentally taken orally, the following may occur: depression of the central nervous system, drowsiness, depression and fainting, decreased blood pressure, bradycardia, decreased body temperature, cyanosis of the skin, apnea.
Treatment: if symptoms of overdose are detected, symptomatic therapy and monitoring of airway patency are necessary.
Storage conditions
Storage conditions
At a temperature not exceeding 25 °C
Shelf life
Shelf life
2 years.
Manufacturer
Manufacturer
Allergan Pharmaceuticals Ireland, Ireland
Additional information
| Shelf life | 2 years. |
|---|---|
| Conditions of storage | At a temperature not exceeding 25 °C |
| Manufacturer | Allergan Pharmaceuticals Ireland |
| Medication form | eye drops |
| Brand | #Н/Д |
Related products
Buy Alfagan R, eye drops 0.15% 5 ml with delivery to USA, UK, Europe and over 120 other countries.