No products in the cart.
Xanthinol nicotinate, tablets 150 mg 60 pcs
€7.59 €6.64
Description
Pharmacotherapeutic group: Vasodilator.
ATX code: C04AD02.
Pharmacological properties
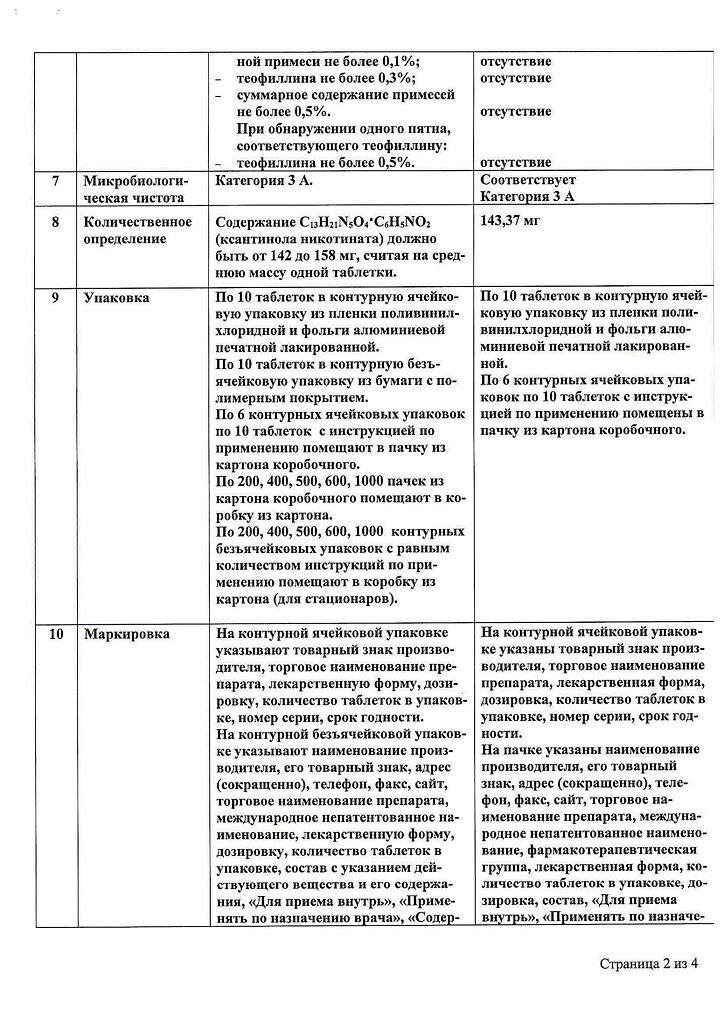
Pharmacodynamics
Xantinol nicotinate has vasodilator and antiaggregant effects. The drug combines the properties of theophylline and nicotinic acid: causes dilation of peripheral vessels, improves collateral circulation, improves oxygenation, increases oxidative phosphorylation and synthesis of adenosine triphosphate (ATP). Xanthinol nicotinate improves cerebral circulation and reduces cerebral hypoxia. The drug also activates the processes of fibrinolysis, reduces blood viscosity, improves microcirculation.
Pharmacokinetics
Xantinol nicotinate is quickly absorbed in the digestive tract. In the body it dissociates to form xanthinol and nicotinic acid. Nicotinic acid binds to blood proteins by less than 20%. Preclinical studies in mice have shown that nicotinic acid bound to a radioactive isotope accumulates in the liver, kidneys, and adipose tissue. Metabolism of nicotinic acid occurs in the liver. There are two pathways of metabolism of nicotinic acid: 1) formation of nicotinamide-adenindinucleotide (NAD) with subsequent conversion to N-methylnicotinamide and N-methyl-2-pyridone-5 -carboxamide (the main metabolic route when nicotinic acid is taken at low doses) and 2) conjugation with glycine to form nicotinic-ureic acid. Nicotinic acid is excreted in the urine, mainly as metabolites; with repeated administration up to 12% of the taken dose of nicotinic acid is excreted unchanged in the urine.
Indications
Indications
Cerebral circulation disorder, Vascular lesions in diabetes, Atherosclerosis, Raynaud’s disease, Thrombophlebitis, Trophic ulcers As part of the complex therapy in the treatment of obliterating atherosclerosis and endarteritis of the lower limbs: diabetic angiopathy; retinopathy; acute thrombophlebitis of superficial and deep veins; strombophlebitis syndrome; trophic ulcers of lower extremities; bedsores; Meniere’s syndrome; dermatoses associated with trophic disorder of vascular nature; acute violation of cerebral circulation.
Active ingredient
Active ingredient
Xanthinol nicotinate
Composition
Composition
1 tablet contains
Active substance:
Csantinol nicotinate – 150.0 mg
Additional substances:
lactose – 15.7 mg;
potato starch – 51.3 mg;
calcium stearate – 2.3 mg;
povidone (polyvinylpyrrolidone low molecular weight medical) – 10.7 mg
How to take, the dosage
How to take, the dosage
Used orally after a meal, starting with 150 mg three times a day, if necessary, the dose is increased to 300-600 mg three times a day. Tablets are taken without chewing. As the patient’s condition improves, the dose is reduced to 150 mg 2-3 times a day. The course of treatment usually lasts at least two months. Repeated course is possible after doctor’s consultation.
Interaction
Interaction
To avoid a sharp fall in blood pressure, the drug should not be used simultaneously with hypotensive agents. It should not be combined with therapy with monoamine oxidase inhibitors (MAO), strophantine.
Special Instructions
Special Instructions
Xanthinol nicotinate causes a “nicotine-like” syndrome accompanied by hyperaesthesia of the mucous membranes of the nose and mouth (aggravates the sensitivity of olfactory and taste receptors) due to which the smell and taste of alcoholic beverages taken during therapy are perceived sharper and more perverse.
Information on the possible effect of the drug for medical use on the ability to operate vehicles, machinery
Higher doses of the drug may cause dizziness, weakness, arterial hypotension and therefore one should refrain from driving and operating machinery.
Synopsis
Synopsis
Tablets are white or almost white, flat-cylindrical, beveled and ribbed.
Contraindications
Contraindications
Acute heart failure, including acute myocardial infarction; IIB-III degree chronic heart failure; mitral stenosis; arterial hypotension; acute bleeding; acute gastric and 12-gastrointestinal ulcer; acute renal failure; glaucoma; childhood; individual hypersensitivity to the components of the preparation, congenital lactose intolerance, lactose deficiency, glucose-galactose malabsorption.
Side effects
Side effects
Xanthinol nicotinate may cause dizziness, decreased blood pressure, general weakness, fever, tingling and reddening of the upper body skin, especially the neck and head, nausea, anorexia, diarrhea. These symptoms usually disappear after 10-20 minutes, do not require special treatment and discontinuation of therapy with this drug. If there are undesirable effects not described in these instructions and the above side effects increase during therapy with this drug, the attending physician should be informed immediately.
The drug causes changes in glucose tolerance, increased activity of “hepatic” transaminases, alkaline phosphatase, hyperuricemia when administered in high doses for a long time.
Overdose
Overdose
The drug is mildly toxic. Acute overdose of xanthinol nicotinate is accompanied by arterial hypotension, general weakness, dizziness, tachycardia, abdominal pain and vomiting. If symptoms of overdose occur, symptomatic therapy is prescribed.
Pregnancy use
Pregnancy use
It is contraindicated in the first trimester. In the second and third trimesters of pregnancy and during lactation, the drug is allowed only in the presence of absolute indications and under careful medical supervision.
Additional information
| Weight | 0.200 kg |
|---|---|
| Shelf life | 4 years. Do not use after the expiration date printed on the package. |
| Conditions of storage | In a dry, light-protected place at a temperature not exceeding 25o C. Keep out of reach of children. |
| Manufacturer | Usolye SCFZ, Russia |
| Medication form | pills |
| Brand | Usolye SCFZ |
Related products
Buy Xanthinol nicotinate, tablets 150 mg 60 pcs with delivery to USA, UK, Europe and over 120 other countries.