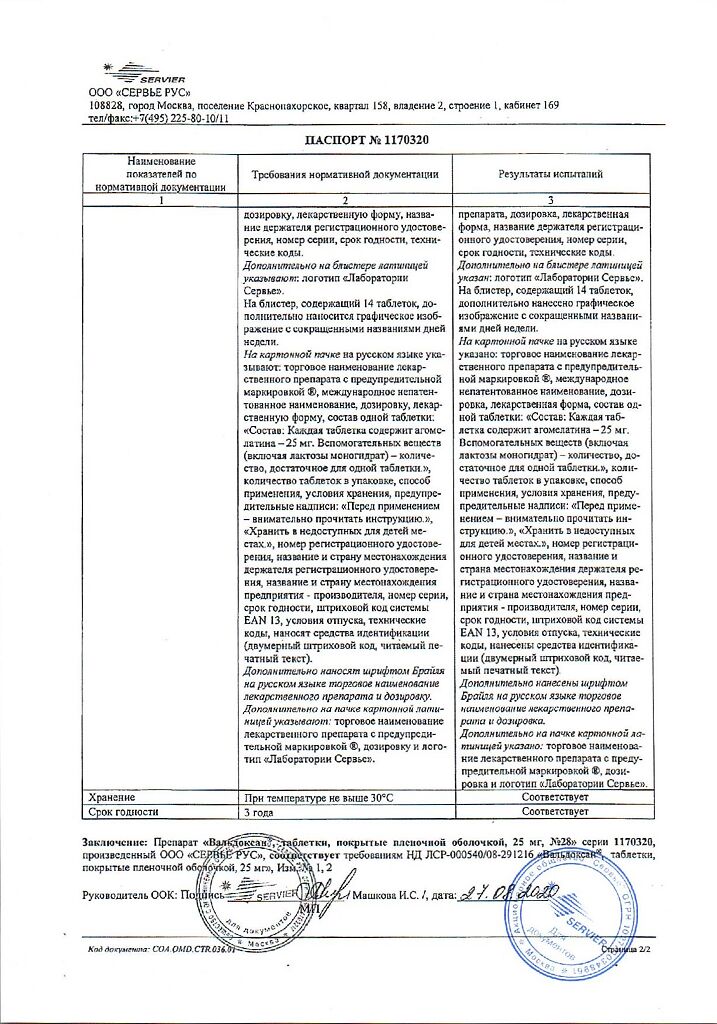
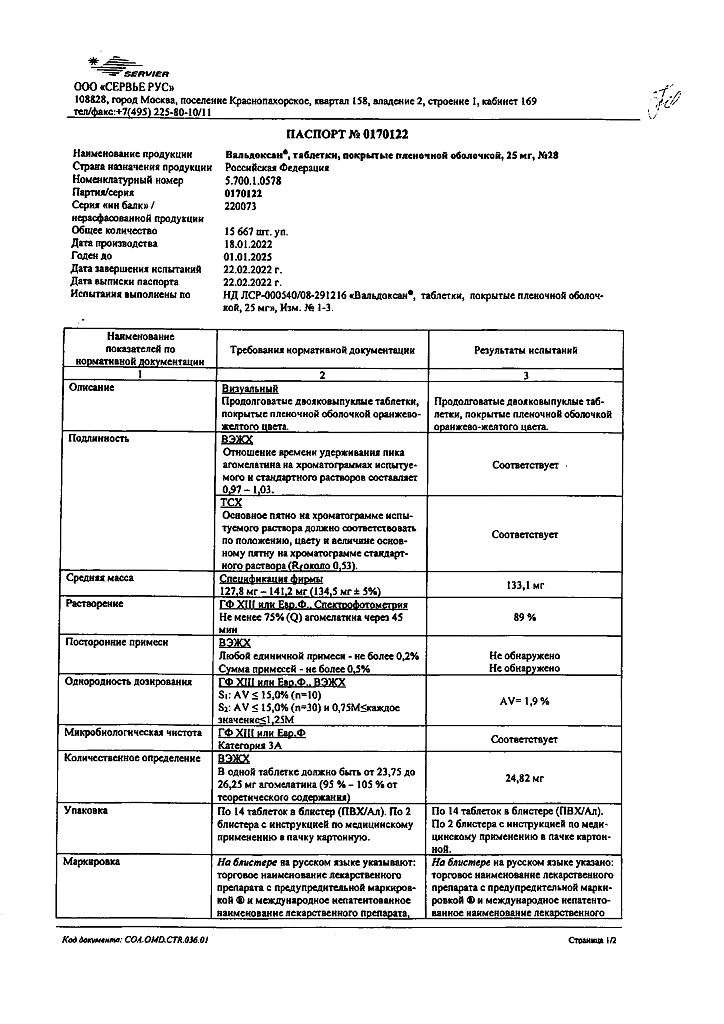
No products in the cart.
Valdoxan, 25 mg 28 pcs
€50.99 €42.49
Description
Agomelatine is a melatoninergic MT1 and MT2 receptor agonist and serotonin 5-HT2C receptor antagonist. Agomelatine is an antidepressant active in validated models of depression (acquired helplessness test, despair test, moderate chronic stress), as well as in models with desynchronization of circadian rhythms, and in experimental situations of anxiety and stress. Agomelatine has not been shown to affect monoamine uptake and has no affinity for alpha-, beta-adrenergic, histaminergic, cholinergic, dopaminergic and benzodiazepine receptors.
Agomelatine increases the release of dopamine and norepinephrine, especially in the prefrontal cortex and does not affect the concentration of extracellular serotonin. In experiments on animals with desynchronization of circadian rhythms it was shown that agomelatine restores synchronization of circadian rhythms through stimulation of melatonin receptors.
Agomelatine helps to restore normal sleep patterns.
Agomelatine short-term use (therapy for 6-8 weeks) in doses of 25-50 mg has been shown to be effective in patients with major depressive episodes.
Agomelatine has also been shown to be effective in patients with more severe forms of depressive disorder (Hamilton scale score >25). Agomelatine was also effective in initially high levels of anxiety, as well as in combinations of anxiety and depressive disorders.
The maintenance antidepressant effect of agomelatine (with a study duration of 6 months) at a dose of 25-50 mg once daily was confirmed. The results of the study confirmed the antidepressant efficacy of agomelatine, as assessed by time to relapse (p = 0.0001). The relapse rate in the group of patients taking agomelatine was 22%, while in the placebo group it was 47%.
The efficacy of agomelatine has been demonstrated in six of seven clinical trials (advantage (2 studies), or comparable efficacy (4 studies)) in heterogeneous adult patient populations with depression, compared with SSRIs/ SSRIs (sertraline, escitalopram, fluoxetine, venlafaxine or duloxetine). The antidepressant effect was assessed using the Hamilton scale (17-item version) as either the primary or secondary endpoint.
Agomelatine has no adverse effect on attention or memory; in depressed patients, agomelatine at a dose of 25 mg increases the duration of the slow sleep phase without changing the number or duration of the fast sleep phases. Agomelatine 25 mg also promotes faster onset of sleep with a decrease in heart rate and improved quality of sleep (from the first week of treatment); no daytime retardation is noted.
Agomelatine tends to reduce the frequency of sexual dysfunction (effect on arousal and orgasm).
Taking agomelatine has no effect on heart rate and blood pressure, does not cause sexual disorders, does not cause “withdrawal” syndrome (even with abrupt termination of treatment) and “addiction” syndrome. The efficacy of agomelatine at a dose of 25-50 mg once daily has been confirmed in elderly depressed patients younger than 75 years in an 8-week clinical trial. There is no confirmed evidence of a significant effect in patients 75 years of age or older.
The tolerability of agomelatine in elderly patients is comparable to that in younger patients. In a 3-week, controlled study involving patients with major depressive disorder with insufficient therapeutic effect from paroxetine (SSRIs) or venlafaxine (SSRIs), withdrawal syndrome was observed when switching from therapy with these antidepressants to agomelatine treatment.
The withdrawal syndrome occurred both after discontinuation of previously prescribed SSRIs/ SSRIs and with their gradual withdrawal, which may have been mistaken for the low efficacy of agomelatine at baseline.
The number of patients who experienced at least one symptom associated with withdrawal after one week of SSRI/ SSRI withdrawal was lower in the long-term dose reduction group (gradual dose reduction of SSRIs/ SSRIs over 2 weeks) than in the rapid dose reduction group (gradual dose reduction of SSRIs/ SSRIs over 1 week) and than in one-step withdrawal: 56.1%, 62.6%, and 79.8% of patients, respectively.
Pharmacokinetics
Absorption and bioavailability
Agomelatine is rapidly (>80%) absorbed after oral administration. Peak plasma concentrations are reached 1-2 hours after oral administration. Absolute bioavailability after therapeutic dose is low (<5%); interindividual variability is significant. Bioavailability in women is higher than in men. Bioavailability increases while taking oral contraceptives and decreases while smoking.
When administering therapeutic doses, the maximum concentration of the drug increased in proportion to the dosage. A more pronounced effect of first passage through the liver was noted with higher doses. Food intake (both regular and high-fat) had no effect on bioavailability or absorption. Interindividual variability was increased with high-fat dietary intake.
Distribution
Volume of distribution in equilibrium phase was about 35 l.
Binding to plasma proteins – 95% regardless of the drug concentration, age or presence of renal failure. In case of hepatic insufficiency a twofold increase of free fraction of the drug was noted.
Biotransformation
After oral administration agomelatine undergoes rapid oxidation, mainly due to CYP1A2 and CYP2C9 isoenzymes. CYP2C19 isoenzyme is also involved in agomelatine metabolism, but its role is less significant.
The main metabolites in the form of hydroxylated and demethylated agomelatine are inactive, rapidly bound and excreted by the kidneys.
Excretion
Excretion is rapid. The plasma elimination half-life is 1 to 2 hours. Metabolic clearance is about 1100 ml/min. Excretion is mainly by the kidneys (80%) as metabolites. The amount of unchanged drug in urine is insignificant. The kinetics does not change if the drug is repeatedly administered.
Renal impairment
In patients with severe renal impairment, the pharmacokinetic parameters were not significantly altered when agomelatine was administered in a single dose of 25 mg. Because of limited clinical experience, caution should be exercised when prescribing agomelatine in patients with moderate to severe renal impairment.
Hepatic failure
. In patients with mild (Child-Pugh Class A) and moderate (Child-Pugh Class B) chronic hepatic impairment with cirrhosis, agomelatine at a dose of 25 mg was associated with increases in plasma concentrations of 70 and 140 times, respectively, when compared to volunteers of comparable sex, age and smoking status but without hepatic impairment.
Elderly Patients
When agomelatine was administered at a dose of 25 mg to elderly patients aged 65 years and older, it was observed that the mean AUC and mean maximum concentration were 4 times and 13 times higher, respectively, in patients aged 75 years and older, compared with patients younger than 75 years. The total number of patients receiving 50 mg was too low to draw any conclusions. No dose adjustment based on age was necessary.
Raciality
There are no data on racial differences in pharmacokinetic parameters.
Indications
Indications
Depression, Aggressiveness and ConfrontationThe treatment of major depressive disorder.
Active ingredient
Active ingredient
Agomelatine
Composition
Composition
1 tablet contains:
The active ingredient:
agomelatine 25 mg.
Excipients: lactose monohydrate, magnesium stearate, corn starch, povidone, colloidal silicon dioxide, sodium carboxymethyl starch, stearic acid.
Film coating: glycerol, hypromellose, iron oxide yellow dye, macrogol 6000, magnesium stearate, titanium dioxide.
How to take, the dosage
How to take, the dosage
Orally. Tablets of the drug Valdoxan® can be taken regardless of meals. The tablet should be swallowed whole without chewing.
If the next dose of the drug is missed, at the next dose Valdoxan® is taken in the usual dose (the missed dose should not be taken). The blister containing the tablets has a calendar imprinted on it to help the patient monitor the medication.
The recommended daily dose is 25 mg (1 tablet) once before bedtime. In the absence of clinical dynamics after two weeks of treatment the dose may be increased to 50 mg (2 tablets of 25 mg) once before bedtime.
The decision to increase the dose should be made taking into account the increasing risk of increased transaminase levels. Any dose increase to 50 mg should be made on the basis of a patient-specific benefit-risk assessment and with close monitoring of liver function tests.
Interaction
Interaction
Potential effects of other drugs
Agomelatine is metabolized in the liver by 90% with cytochrome P450 1A2 (CYP1A2) and by 10% with CYP2C9/19. Therefore, any drugs whose metabolism depends on these isoenzymes may increase or decrease the bioavailability of agomelatine.
Fluvoxamine, is a strong inhibitor of the CYP1A2 isoenzyme and a moderate inhibitor of the CYP2C9 isoenzyme and significantly slows agomelatine metabolism, with an average of 60 (12 – 412) times increased agomelatine concentration. Therefore, concomitant use of agomelatine and strong CYP1A2 isoenzyme inhibitors (such as fluvoxamine, ciprofloxacin) is contraindicated. Concomitant administration of agomelatine and estrogens, which are moderate CYP1A2 isoenzyme inhibitors, leads to a several-fold increase in agomelatine concentration. Although the combined use of agomelatine and estrogens has not been associated with a worsening of the safety profile of current therapy, caution should be exercised when concomitantly prescribing agomelatine with other moderate CYP1A2 inhibitors (such as propranolol, enoxacin) until sufficient clinical experience is gained (see section “Special Precautions”).
Rifampicin, as an inducer of all three isoenzymes involved in agomelatine metabolism, may decrease the bioavailability of agomelatine.
Smoking, by inducing the CYP1A2 isoenzyme, has been shown to decrease the bioavailability of agomelatine, especially in patients who abuse smoking (>15 cigarettes/day).
Potential effects of agomelatine on other drugs
In vivo agomelatine does not induce cytochrome P450 isoenzymes. Agomelatine does not inhibit the CYP1A2 isoenzyme in vivo and other cytochrome P450 isoenzymes in vitro.
Therefore, agomelatine does not affect the concentration of drugs whose metabolism is associated with these isoenzymes.
Drugs that significantly bind to plasma proteins Agomelatine did not change the free concentration of drugs that significantly bind to plasma proteins and, in turn, they had no effect on the concentration of agomelatine.
Other drugs
There were no pharmacokinetic or pharmacodynamic interactions between agomelatine and drugs frequently used in the target patient population: benzodiazepines, lithium drugs, paroxetine, fluconazole and theophylline.
Alcohol
The use of agomelatine with alcohol is not recommended.
Electroconvulsive therapy (ECT)
There are no data on the use of agomelatine concomitantly with ECT. Because agomelatine did not contribute to seizures in animal experiments, adverse effects of concomitant use of agomelatine and ECT seem unlikely.
Special Instructions
Special Instructions
Monitoring of liver function parameters:
Hepatic impairment has been reported, including hepatic failure (exceptionally fatal or requiring liver transplantation in patients with pre-existing risk factors for liver damage), elevated liver enzymes more than 10 times the upper limit of normal, hepatitis, and jaundice in patients taking Valdoxan® during the post-registration period. Most of these abnormalities occurred in the first months of treatment. The pattern of liver damage appears to be mainly hepatocellular. Transaminase levels generally returned to normal values after discontinuation of therapy.
Caution should be exercised before initiating treatment and close monitoring during treatment for all patients, especially those with risk factors for liver disease or receiving concomitant therapy with drugs that may cause liver damage.
- Before initiating therapy
Treatment with Valdoxan® should be prescribed only after careful evaluation of the ratio of expected benefit to possible risk in patients with risk factors for hepatic impairment, such as:
- obesity/overweight/non-alcoholic fatty hepatosis, diabetes,
- alcoholism and/or alcohol abuse;
- and taking medications that may cause liver function impairment.
Prior to initiating therapy, liver function tests should be performed in all patients, and therapy should not be initiated if liver enzyme levels of AJIT and/or ACT are more than 3 times the upper limit of normal. Caution should be exercised when Valdoxan® is administered to patients with baseline elevated transaminase activity (above the upper limit of normal, but not more than 3 times the upper limit of normal).
- Periodicity of liver function tests
- Before therapy
- After:
- approximately 3 weeks,
- approximately 6 weeks later (end of the arresting period of therapy),
- approximately 12 and 24 weeks later (end of maintenance period of therapy)
- hereafter, according to the clinical situation.
- When the dose is increased, liver function should be monitored with the same frequency as at the beginning of therapy.
If serum transaminase activity increases, repeat testing within 48 hours.
- Treatment
Treatment with Valdoxan® should be discontinued immediately in case of
- appearance of symptoms and signs of possible liver dysfunction (such as dark urine, discolored stools, yellowing of skin/eyes, right upper abdominal pain, recent onset of persistent and unexplained fatigue).
- Elevation of transaminases by more than 3 times the upper limit of normal.
After discontinuation of Valdoxan® therapy liver function tests should be performed regularly until normalization of transaminase levels.
Elderly patients
Efficacy of drug administration in elderly patients (aged 75 years and older) is not established. Therefore, Valdoxan® should not be prescribed to patients in this age group.
Elderly patients with dementia
Valdoxan® should not be prescribed for treatment of major depressive episodes in elderly patients with dementia (due to lack of data on efficacy and safety of the drug in this patient group).
Patients with renal impairment
No significant changes in pharmacokinetic parameters have been observed in patients with severe renal impairment. However, there is limited experience with Valdoxan® for major depressive episodes in patients with moderate to severe renal impairment. Caution should be exercised when Valdoxan® is prescribed to these patients.
Bipolar Disorders/Mania/Hypomania
Caution should be exercised when Valdoxan® is used in patients with a history of bipolar disorders, manic or hypomanic episodes. If symptoms of mania occur, the drug should be discontinued.
Suicidal/Suicidal Behavior
With depression, there is an increased risk of suicidal ideation, self-harm, and suicide (suicide-related events). The risk persists until the onset of a distinct remission. Patients must be medically monitored until their condition improves (it may take several weeks after starting therapy before the condition improves). Clinical experience suggests that the risk of suicide may increase in the early stages of remission.
Patients with a history of suicidal events, as well as patients who had suicidal intentions prior to therapy, are at risk and should be under close medical supervision during therapy.
Results of a meta-analysis of clinical trials of antidepressants in patients with psychiatric disorders indicate an increased risk of suicidal behavior in patients under 25 years of age on antidepressants compared to placebo.
During treatment, patients, especially those at risk, should be under close medical supervision, especially at the beginning of therapy and when changing the dose of the drug. Patients (and their caregivers) should be informed to seek immediate medical attention if their condition worsens, if suicidal and unusual behavior occurs, and if suicidal ideation occurs.
Concomitant use with CYP1A2 isoenzyme inhibitors
Caution should be exercised when using agomelatine concomitantly with moderate CYP1A2 isoenzyme inhibitors (such as propranololol, enoxacin) because of the possibility of increased agomelatine concentration.
Patients with lactose intolerance
The drug should not be used in patients with lactose intolerance: lactase deficiency, galactosemia and glucose-galactose malabsorption.
Influence on driving and operating ability
There have been no studies of the effect of Valdoxan® on driving and operating ability. It should be remembered that dizziness and somnolence are frequent side effects of agomelatine.
Contraindications
Contraindications
- High sensitivity to agomelatine and/or any of the excipients of the drug.
- Hepatic failure (e.g., cirrhosis or active liver disease) or an increase in transaminase levels of more than 3 times the upper limit of normal.
- Simultaneous use of potent CYP1A2 isoenzyme inhibitors (such as fluvokeamine, ciprofloquecine).
- Children under 18 years of age (due to insufficient experience with clinical use). In children and adolescents on other antidepressants, suicidal behavior (suicide attempts and suicidal ideation) and hostility (mainly aggressiveness, conflict behavior, irritation) were observed more frequently compared to the placebo group.
The drug should not be used in patients with lactose intolerance: lactase deficiency, galactosemia and glucose-galactose malabsorption.
Precautions for use
. Patients with moderate to severe renal impairment when treating major depressive episodes, when concomitant administration of agomelatine with moderate CYP1A2 isoenzyme inhibitors (such as propranolol, enoxacin), patients with a history of manic or hypomanic episodes, patients with a history of events involving suicide, and patients who had suicidal intentions before therapy.
Patients who abuse alcohol or take medications that may cause hepatic impairment should be cautioned when prescribing the drug.
Side effects
Side effects
In clinical trials, Valdoxan® was given to more than 8,000 patients with depression.
The side effects were most often mild to moderate and were observed in the first two weeks of treatment. The most common were headache, nausea and dizziness. These side effects were usually transient and usually did not require discontinuation of treatment. Below are the side effects observed in placebo-controlled and comparative clinical trials.
The frequency of agomelatine side effects is given as the following gradation: Very common (>1/10), common (>1/100, <1/10), infrequent (>1/1000, <1/100), rare (>1/10000, <1/1000); very rare (<1/10000), unspecified frequency.
Injuries to the central nervous system:
- Very common: headache
- Often: dizziness, somnolence, insomnia.
- Infrequent: migraine, paresthesias, restless legs syndrome*.
- Rarely: akathisia*.
Gastrointestinal tract:
- Frequently: nausea, diarrhea, constipation, abdominal pain, vomiting*.
Disorders of the liver and biliary tract:
- Often: Increased AJIT and/or ACT activity (more than 3 times the upper limit of normal in 1.2% of patients taking agomelatine at a dose of 25 mg daily and in 2.6% of patients taking agomelatine at a dose of 50 mg daily, compared with 0.5% on placebo in clinical trials).
- Infrequent: increased γ-glutamyltransferase* (GGT) activity (more than 3 times the upper limit of normal)
- Rarely: hepatitis, increased alkaline phosphatase activity* (more than 3 times the upper limit of normal), liver failure*(1), jaundice*.
Skin and subcutaneous tissue:
- Infrequent: sweating, eczema, itching*, urticaria*.
- Rarely: erythematous rash, facial edema and Quincke’s edema*.
Hearing organ:
- Infrequent: tinnitus*.
Visual organ:
- Infrequent: blurred vision.
Musculoskeletal system:
- Frequently: back pain.
Renal and urinary tract disorders:
- Rarely: urinary retention*.
General disorders:
- Often: fatigue.
Mental disorders
- Often: anxiety, unusual dreams*.
- Infrequent: suicidal thoughts or suicidal behavior, agitation and related symptoms* (such as irritability and restlessness), aggressiveness*, nightmares*, mania/hypomania* (these symptoms may also be a manifestation of underlying disease), confusion*.
- Rare: Hallucinations*.
Data from (additional) examinations
- Often: weight gain*.
- Infrequent: weight loss*.
* The frequency of adverse reactions identified from spontaneous reports was estimated based on data from clinical studies.
(1) Only a few cases with fatal outcome or liver transplantation have been reported in patients with pre-existing risk factors for liver damage.
Overdose
Overdose
Data on agomelatine overdose are limited.
Symptoms: drowsiness, epigastric pain, restlessness, weakness, anxiety, agitation, tension, dizziness, cyanosis, malaise.
When the patient took agomelatine at a dose of 2450 mg, the condition normalized on its own, without cardiovascular abnormalities or changes in laboratory parameters.
Treatment: Specific antidotes for agomelatine are not known. Symptomatic treatment and monitoring in specialized departments with follow-up.
Pregnancy use
Pregnancy use
Significant experience with agomelatine in pregnant women has not shown any adverse effects on pregnancy, fetal or neonatal health. No other epidemiologic data are available at this time. In pregnancy, the drug should be used with caution and in cases where the expected benefit of therapy to the mother outweighs the potential risk to the fetus.
It is unknown whether agomelatine is excreted with breast milk in humans. Breastfeeding should be discontinued if Valdoxan should be used during lactation.
There have been no direct or indirect harmful effects of the drug on pregnancy, embryonic or fetal development, labor or postnatal development in experimental animal studies. Agomelatine and its metabolites have been shown to be excreted with breast milk in rats.
Significant experience with agomelatine in pregnant women has not shown any adverse effects on pregnancy, fetal or neonatal health. No other epidemiologic data are available at this time. In pregnancy, the drug should be used with caution and in cases where the expected benefit of therapy to the mother outweighs the potential risk to the fetus.
It is not known whether agomelatine is excreted with the breast milk in humans. Breastfeeding should be stopped if Valdoxan has to be used during lactation.
There have been no direct or indirect harmful effects of the drug on pregnancy, embryonic or fetal development, labor or postnatal development in experimental animal studies. Agomelatine and its metabolites have been shown to be excreted with breast milk in rats.
Additional information
| Weight | 0.014 kg |
|---|---|
| Shelf life | 3 years |
| Conditions of storage | No special storage conditions required |
| Manufacturer | Servier Rus LLC, Russia |
| Medication form | pills |
| Brand | Servier Rus LLC |
Related products
Buy Valdoxan, 25 mg 28 pcs with delivery to USA, UK, Europe and over 120 other countries.