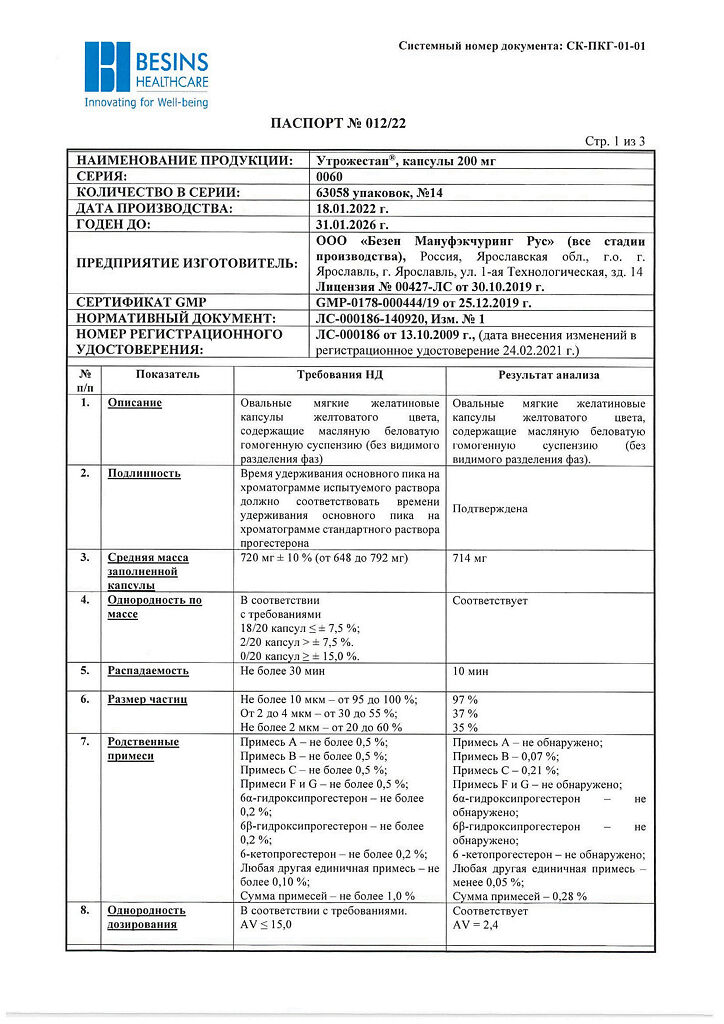
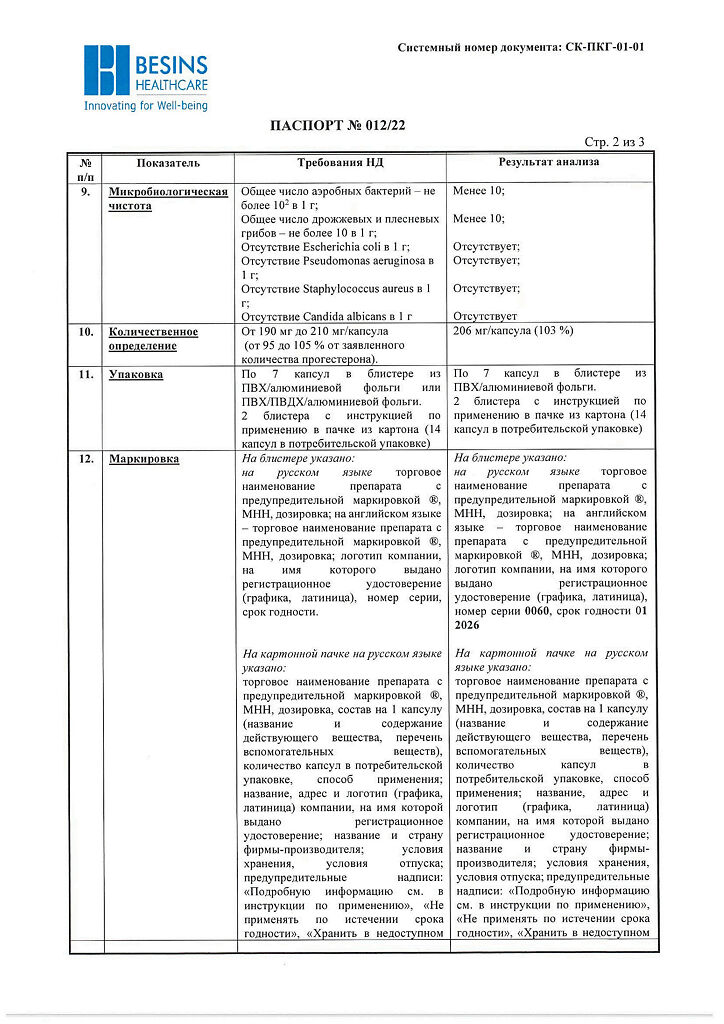
No products in the cart.
Utrogestan, 200 mg capsules 14 pcs
€15.84 €13.53
Description
Pharmacotherapeutic group: gestagen
ATX code: G03DA04
Pharmacological Properties
Pharmacodynamics
The active ingredient of Utrogestan® is progesterone, which is identical to the natural hormone of the corpus luteum of the ovary. It binds to receptors on the surface of target cells and penetrates the nucleus where, by activating DNA, it stimulates RNA synthesis. It promotes transition of uterine mucosa from the phase of proliferation caused by the follicular hormone estradiol to the secretory phase, and after fertilization to a state required for development of a fertilized egg.
It reduces the excitability and contractility of uterine muscles and fallopian tubes. Promotes the formation of normal endometrium. Stimulates the development of mammary gland terminal elements and induces lactation.
By stimulating protein lipase, increases fat reserves; increases glucose utilization; by increasing the concentration of basal and stimulated insulin, promotes glycogen accumulation in the liver; increases production of gonadotropic hormones of the pituitary; reduces azotemia, increases nitrogen excretion by the kidneys.
Pharmacokinetics
When taken orally.
Indications
Indications
Premenstrual syndrome, Threatened miscarriage, Mastopathy, Infertility, Climax, Threatened premature birthProgesterone deficiency in women:
Actually:
– threatened abortion or prevention of habitual abortion due to progesterone insufficiency;
– infertility due to luteal insufficiency;
– premenstrual syndrome;
– menstrual cycle disorders due to ovulation disorders or anovulation;
– cystic fibrosis mastopathy;
– menopausal transition period;
– menopausal hormone therapy (MHT) in peri- and postmenopause (in combination with oestrogen-containing medications).
For intravaginal use:
– MGT in case of progesterone deficiency in non-functioning (absent) ovaries (egg donation);
– Prevention (prevention) of premature births in women from the risk group (with shortened cervix and/or with the history of premature births and/or premature rupture of membranes);
– support of the luteal phase in preparation for in vitro fertilization;
– support of the luteal phase in a spontaneous or induced menstrual cycle;
premature menopause;
– MGT (in combination with estrogen-containing drugs);
– infertility due to luteal insufficiency;
– threatened abortion or prevention of habitual abortion due to progesterone insufficiency.
Active ingredient
Active ingredient
Progesterone
Composition
Composition
for 1 capsule:
Active substance:
Progesterone micronized 100 or 200 mg.
Associates:
Sunflower oil 149 mg/298 mg,
Soybean lecithin 1 mg/2 mg;
The capsule – gelatin 76.88 mg/153.76 mg,
Glycerin 31.45 mg/62.9 mg,
Titanium dioxide 1.67 mg/3.34 mg.
Description:
The 100 mg capsules are round, the 200 mg capsules are oval, soft shiny yellowish gelatin capsules containing an oily whitish homogeneous suspension (no visible phase separation).
How to take, the dosage
How to take, the dosage
Orally
The drug is taken orally in the evening before bedtime with water.
In most cases of progesterone deficiency, the daily dose of Utrogestan® is 200-300 mg divided into two doses (200 mg in the evening before bedtime and 100 mg in the morning, if necessary).
In threatened abortion or to prevent habitual abortion due to progesterone deficiency: 200-600 mg daily in the first and second trimesters of pregnancy. Further administration of Utrogestan® is possible by prescription of a physician on the basis of the evaluation of the clinical data of a pregnant woman.
In case of luteal phase failure (premenstrual syndrome, cystic fibrosis, dysmenorrhea, menopausal transition) the daily dose is 200 or 400 mg taken for 10 days (usually from the 17th to the 26th day of the cycle).
In MGT in perimenopause against a background of estrogen administration, Utrogestan® is used 200 mg daily for 12 days. At MGT in postmenopause in a continuous regimen Utrogestan® is used in a dose of 100-200 mg from the first day of use of oestrogen-containing drugs. The dose is adjusted individually.
Intravaginal
The capsules are placed deep in the vagina.
Prevention (prevention) of preterm birth in women at risk (with shortened cervix and/or a history of preterm birth and/or premature rupture of membranes): the usual dose is 200 mg before bedtime, from the 22nd to the 34th week of pregnancy.
The complete absence of progesterone in women with non-functioning (absent) ovaries (egg donation): on the background of estrogen therapy by 100 mg per day on days 13 and 14 of the cycle, then by 100 mg 2 times a day from day 15 to 25 of the cycle, from day 26, and in case of pregnancy determination the dose increases by 100 mg a day every week, reaching a maximum of 600 mg a day, divided into 3 doses. This dose can be used for up to 60 days.
Luteal phase support during an in vitro fertilization cycle: 200 to 600 mg daily starting on the day of chorionic gonadotropin injection during the first and second trimesters of pregnancy.
Luteal phase support in spontaneous or induced menstrual cycle in infertility associated with corpus luteum dysfunction: it is recommended to use 200-300 mg daily starting from the 17th day of the cycle for 10 days, in case of delayed menstruation and pregnancy diagnosis the treatment should be continued.
In cases of threatened abortion or to prevent habitual abortion arising against the background of progesterone insufficiency: 200-400 mg daily in 2 doses daily in I and II trimesters of pregnancy.
Interaction
Interaction
In oral administration
Progesterone increases the effect of diuretics, hypotensive drugs, immunosuppressants, anticoagulants.
Concomitant use with agents inducers of microsomal liver enzymes CYP3A4, such as barbiturates, antiepileptic drugs (phenytoin, carbamazepine), rifampicin, phenylbutazone, spironolactone, griseofulvin is accompanied by acceleration of progesterone metabolism in the liver.
Concomitant administration of progesterone with some antibiotics (penicillins, tetracyclines) may lead to a decrease in its effectiveness due to impaired intestinal hepatic recirculation of sex hormones due to changes in intestinal microflora.
The degree of severity of these interactions may vary from patient to patient; therefore, it is difficult to predict the clinical effects of these interactions.
Ketoconazole may increase the bioavailability of progesterone. Progesterone can increase the concentration of ketoconazole and cyclosporine.
Progesterone may decrease the effectiveness of bromocriptine.
Progesterone may cause a decrease in glucose tolerance, thereby increasing the need for insulin or other hypoglycemic drugs in diabetic patients.
Progesterone bioavailability may be decreased in female patients who smoke and excessive alcohol consumption.
In intravaginal use
Interaction of progesterone with other drugs in intravaginal use has not been evaluated. Concomitant use of other drugs administered intravaginally should be avoided to avoid impairing the release and absorption of progesterone.
Special Instructions
Special Instructions
The drug Utrogestan® should not be used for contraceptive purposes.
The drug should not be taken with food, since eating increases the bioavailability of progesterone.
The preparation Utrogestan® should be used with caution in patients with diseases and conditions, which may be aggravated by fluid retention (arterial hypertension, cardiovascular diseases, chronic renal failure, epilepsy, migraine, bronchial asthma); in patients with diabetes, liver function disorders of mild and moderate severity, photosensitivity.
Patients with a history of depression should be monitored, and in case of severe depression the drug should be discontinued.
Utrogestan® contains soy lecithin, which may cause hypersensitivity reactions (urticaria and anaphylactic shock).
Patients with concomitant cardiovascular diseases or with their history should also be periodically monitored by a physician.
The use of Utrogestan® after the first trimester of pregnancy may cause the development of cholestasis.
During long-term treatment with progesterone regular medical examinations (including liver function tests) should be performed; treatment should be discontinued if there are deviations from normal liver function tests or cholestatic jaundice.
Progesterone use may decrease glucose tolerance and increase the need for insulin and other hypoglycemic drugs in diabetic patients.
If amenorrhea occurs during treatment it is necessary to exclude the presence of pregnancy.
If the treatment course is started too early at the beginning of the menstrual cycle, especially before the 15th day of the cycle, there may be shortening of the cycle and/or acyclic bleeding. In case of acyclical bleeding the drug should not be used until the cause has been determined, including histological examination of the endometrium.
If there is a history of chloasma or the tendency to develop it, the patients are recommended to avoid UV-exposure.
More than 50% of spontaneous abortions in early pregnancy are caused by genetic disorders. In addition, the cause of spontaneous abortions in early pregnancy may be infectious processes and mechanical damage. Administration of Utrogestan® in these cases may result only in delayed rejection and evacuation of a non-viable fetal egg.
Utrogestan® administration for prevention of threatened abortion is justified only in cases of progesterone deficiency.
If MHT is performed with oestrogens during perimenopause, the use of Utrogestan® is recommended for at least 12 days of the menstrual cycle.
In case of continuous MHT in postmenopause it is recommended to use the drug from the first day of estrogen intake.
MHT increases the risk of venous thromboembolism (deep vein thrombosis or pulmonary embolism), risk of ischemic stroke and coronary heart disease.
Because of the risk of thromboembolic complications the drug should be discontinued in case of: visual disorders such as vision loss, exophthalmus, double vision, retinal vascular lesions; migraine; venous thromboembolism or thrombotic complications, regardless of their location.
If there is a history of thrombophlebitis, the patient should be closely monitored.
When using Utrogestan® with estrogen-containing drugs it is necessary to refer to the instructions for their use concerning the risks of venous thromboembolism.
Results of the Women Health Initiative Study (WHI) suggest a small increase in breast cancer risk when estrogen-containing medications and synthetic gestagens are used together for longer than 5 years. It is unknown whether there is an increased risk of breast cancer in postmenopausal women when MHT is given with estrogen-containing medications in combination with progesterone.
Results of the WHI study also found an increased risk of dementia when starting MHT over the age of 65.
Before starting MHT and regularly during it, women should be examined to identify contraindications to MHT. If there are clinical indications, a breast examination and gynecological examination should be performed.
Progesterone use may affect the results of some laboratory tests, including indicators of liver function, thyroid function; coagulation parameters; pregnandiol concentration.
Influence on the ability to drive vehicles and mechanisms
When using the drug orally, caution should be exercised when driving vehicles and engaging in other potentially dangerous activities that require increased concentration and rapid psychomotor reactions.
Contraindications
Contraindications
Hypersensitivity to progesterone or any of the excipients of the drug; deep vein thrombosis, thrombophlebitis; thromboembolic disorders (pulmonary embolism, myocardial infarction, stroke), intracranial hemorrhage or history of these conditions/diseases; vaginal bleeding of unclear genesis; incomplete abortion; porphyria; established or suspected malignancies of the breast and genital organs; severe liver disease (including cholestatic jaundice, hepatitis, Dubin-Johnson syndrome, Rotor, malignant liver tumors) at present or in anamnesis; childhood age below 18 years (effectiveness and safety not established); period of breast-feeding.
With caution:
Cardiovascular disease, arterial hypertension, chronic renal failure, diabetes mellitus, bronchial asthma, epilepsy, migraine, depression, hyperlipoproteinemia, mild to moderate liver function disorders; photosensitivity.
The drug should be used with caution during the second and third trimesters of pregnancy.
Side effects
Side effects
The following adverse events noted with oral administration of the drug are distributed by frequency of occurrence according to the following gradation: frequently >1/100, < 1/10; infrequently >1/1000, < 1/100; rarely >1/10000, < 1/1000; very rarely < 1/10000.
Genital and mammary gland disorders:often – menstrual irregularities, amenorrhea, acyclic bleeding; infrequent – mastodynia.
Mental side:very rarely – depression.
Nervous system disorders:often – headache; infrequently – drowsiness, transient dizziness.
Gastrointestinal disorders:often – bloating; infrequent – vomiting, diarrhea, constipation; rarely – nausea.
Hepatic and biliary tract disorders: infrequent – cholestatic jaundice.
From the immune system:very rarely – urticaria.
Skin and subcutaneous tissue: infrequently – itching, acne; very rarely – chloasma.
Drowsiness, transient dizziness are possible, usually 1-3 hours after taking the drug. These side effects can be reduced by reducing the dose, using the drug before bedtime, or switching to the vaginal route of administration.
These adverse effects are usually the first signs of an overdose. Drowsiness and/or transient dizziness have been observed, particularly in cases of concomitant hypoestrogenism. Reducing the dose or restoring higher estrogenicity immediately eliminates these phenomena without reducing the therapeutic effect of progesterone.
If treatment begins too early (in the first half of the menstrual cycle, especially before day 15), menstrual cycle shortening or acyclic bleeding may occur.
Recorded menstrual cycle changes, amenorrhea or acyclic bleeding are common with all progestagens.
Application in clinical practice
The following adverse events with oral progesterone have been noted in postmarketing use: Insomnia; premenstrual syndrome; breast tension; vaginal discharge; joint pain; hyperthermia; increased sweating at night; fluid retention; body weight changes; acute pancreatitis; alopecia, hirsutism; changes in libido; thrombosis and thromboembolic complications (if MHT is combined with estrogen-containing medications); increased BP.
The drug contains soy lecithin, which may cause hypersensitivity reactions (urticaria and anaphylactic shock).
Vaginal administration There have been reports of individual cases of local intolerance reactions of the drug components (in particular soy lecithin) in the form of hyperemia of the vaginal mucosa, burning, itching, oily discharge.
Systemic adverse effects have not been noted with intravaginal administration at the recommended doses, such as drowsiness or dizziness (observed with oral administration of the drug).
Overdose
Overdose
Symptoms: drowsiness, transient dizziness, euphoria, menstrual cycle shortening, dysmenorrhea.
In some patients the average therapeutic dose may be excessive due to the existing or emerged unstable endogenous progesterone secretion, special sensitivity to the drug or too low concentration of estradiol.
Treatment: If drowsiness or dizziness occurs, reduce the daily dose or prescribe taking the drug before bedtime for 10 days of the menstrual cycle; if the menstrual cycle is shortened or there is “smeared” bloody discharge, it is recommended that the start of treatment be moved to a later day of the cycle (for example, the 19th instead of the 17th); in perimenopause and with MHT in postmenopause, make sure that the estradiol concentration is optimum.
In case of overdose, symptomatic treatment is administered if necessary.
Similarities
Similarities
Utrogestan, Progesterone, Progestogel, Iprozhin, Prajisan, vaginal progesterone gel 90mg
Additional information
| Weight | 0.022 kg |
|---|---|
| Shelf life | 3 years. Do not use after the expiration date. |
| Conditions of storage | Store at a temperature not exceeding 25 ° C. Store out of the reach of children. |
| Manufacturer | Bezen Manufacturing Rus LLC, Russia |
| Medication form | capsules |
| Brand | Bezen Manufacturing Rus LLC |
Other forms…
Related products
Buy Utrogestan, 200 mg capsules 14 pcs with delivery to USA, UK, Europe and over 120 other countries.