No products in the cart.
Ultrix Quadri, 0.5 ml/dose 0.5 ml
€1.00
Out of stock
(E-mail when Stock is available)
Description
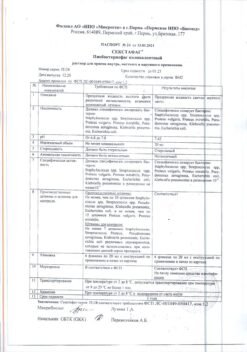
Pharmacotherapeutic group.
MIBP vaccine.
The ATX code: J07BB02.
Inactivated purified split influenza vaccine. Prevents disease caused by influenza virus types A and B.
Induces the formation of humoral antibodies to hemagglutinins that neutralize influenza viruses. Seroprotective level of antibodies is usually achieved within 7-10 days after vaccine administration. The duration of postvaccinal immunity to homologous or related strains varies from 6 to 12 months.
Indications
Indications
Active annual prophylactic immunization against seasonal influenza in children aged 6 years and older, adolescents and adults up to 60 years of age.
The vaccine is especially indicated:
- People at high risk for illness and complications from influenza:
– persons frequently ill with acute respiratory viral infections;
– Persons suffering from chronic somatic diseases, including diseases and malformations of the cardiovascular, respiratory and central nervous systems, chronic kidney diseases, metabolic diseases, diabetes, chronic anemia, allergic diseases (except allergies to chicken proteins), congenital or acquired immunodeficiency, including those infected with human immunodeficiency virus. - People who by virtue of their educational or professional activities have a high risk of influenza or infecting other persons:
– employees of medical and educational institutions, transport, utilities and social services, police, military personnel, etc.
.
How to take, the dosage
How to take, the dosage
Vaccination is carried out annually in the autumn-winter period. Vaccination is possible at the beginning of the epidemic rise of influenza.
The vaccine is administered intramuscularly in a dose of 0.5 ml once in the deltoid muscle (the upper third of the outer surface of the shoulder).
The drug is not suitable for use in syringes/vials with compromised integrity or labeling, with changes in physical properties (color, transparency), with the presence of foreign particles in the solution, with expired shelf life, violation of storage conditions.
Opening of syringes / bottles and vaccination shall be carried out in strict compliance with aseptic and antiseptic rules. The drug in the opened syringes/vials shall not be stored.
Interaction
Interaction
The vaccine can be used simultaneously with inactivated and live vaccines of the National Preventive Immunization Calendar (except for tuberculosis vaccines) and inactivated vaccines of the Preventive Immunization Calendar for epidemiological indications (except for anti-rabies). Contraindications for each vaccine used must be taken into account; the drugs should be injected into different parts of the body with different syringes.
Vaccines can be administered against the background therapy of the underlying disease. Vaccination of patients who received immunosuppressive therapy (glucocorticosteroids, cytotoxic drugs, radiotherapy) may be less effective.
Special Instructions
Special Instructions
The used and unused syringes and vials shall be disposed in accordance with the requirements of applicable law.
Influence on the ability to drive vehicles, mechanisms
The drug does not affect the ability to drive vehicles or engage in other potentially dangerous activities requiring increased concentration and quick psychomotor reactions.
Contraindications
Contraindications
- allergic reactions to previous flu vaccines;
- allergic reactions to chicken protein and other vaccine components;
- acute febrile, acute infectious and non-infectious diseases, conditions or exacerbation of chronic disease;
- age under 18 years of age;
In case of non-serious acute respiratory infections and acute intestinal diseases, the vaccination is carried out after normalization of the body temperature of the vaccinated; with chronic diseases in the acute stage the vaccination is carried out after convalescence (remission).
With caution.
Do not administer intravenously!
Before vaccination the immunized must be examined by a doctor (paramedic) with mandatory thermometry. If the body temperature is higher than 37 ° C the vaccination is not carried out.
The rooms where the vaccination is carried out must be equipped with antishock therapy. The vaccinated person must be under the supervision of a health care worker for 30 minutes after vaccination.
Side effects
Side effects
The undesired phenomena presented below are listed according to organ and organ system involvement and frequency of occurrence. The frequency of occurrence is defined as follows: very frequently (≥1/10), frequently (≥1/100 and < 1/10), infrequently (≥1/1000 and < 1/100), rarely (≥1/10000 and < 1/1000), very rarely (< 1/10000, including individual cases).
Nervous system disorders: Often: headache.
Skin and subcutaneous tissue disorders: Infrequent: increased sweating.
Musculoskeletal and connective tissue disorders: Infrequent: arthralgia, myalgia.
General disorders and reactions at the injection site: Often: pain, hyperemia at the injection site.
From “infrequent” to “frequent”: thickening, swelling and itching at the injection site.
Infrequent: fever, chills, weakness.
These adverse reactions may develop on the day of vaccination; usually disappear independently in 1-3 days and do not require treatment.
Despite the lack of clinical data, we can not exclude the possibility of development of neurological disorders and allergic reactions characteristic of influenza vaccines (including immediate reactions to chicken protein and other vaccine components).
The patient should be informed about the need to inform the doctor of any pronounced or not specified in these instructions adverse reactions.
Pregnancy use
Pregnancy use
Experience in the use of influenza inactivated vaccines shows that the vaccination of women during breastfeeding has no toxic effects on the child.
The final decision on the vaccination of pregnant and breastfeeding women should be made by a doctor individually, taking into account the risk of influenza infection and possible complications caused by influenza disease. The safest time to vaccinate pregnant women is in the second and third trimesters of pregnancy.
Additional information
| Weight | 0.020 kg |
|---|---|
| Shelf life | 1 year. The drug with expired shelf life cannot be used. |
| Conditions of storage | Store at 2 to 8 ° C. Do not freeze. Keep out of reach of children. |
| Manufacturer | FORT LLC, Russia |
| Medication form | solution |
| Brand | FORT LLC |
Related products
Buy Ultrix Quadri, 0.5 ml/dose 0.5 ml with delivery to USA, UK, Europe and over 120 other countries.