No products in the cart.
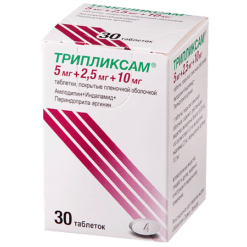
Triplixam, 5 mg+1.25 mg+5 mg 30 pcs.
€23.14 €19.29
Description
Triplixam® is a combination drug that includes three antihypertensive components, each of which complements the action of the others in controlling blood pressure in patients with arterial hypertension. Amlodipine is a “slow” calcium channel blocker (CMCB), a dihydropyridine derivative, indapamide is a sulfonamide diuretic, perindopril arginine is an inhibitor of the enzyme that converts angiotensin I into angiotensin II (ACE inhibitor)
Indications
Indications
As therapy in patients with arterial hypertension when BP decreases on the background of taking amlodipine, indapamide and perindopril at the same doses.
Active ingredient
Active ingredient
Amlodipine, Indapamide, Perindopril
How to take, the dosage
How to take, the dosage
Orally, 1 tablet once a day, preferably in the morning before a meal.
Special Instructions
Special Instructions
All precautions associated with the administration of individual components of the drug should be considered when using their fixed combination in Triplixam®.
Amlodipine
Chronic heart failure
Treatment of patients with chronic heart failure should be performed with caution.
During amlodipine use in patients with chronic heart failure class III and functional class according to NYHA classification pulmonary edema may develop. Slow” calcium channel blockers, including amlodipine, should be used with caution in patients with chronic heart failure due to possible increase of risk of cardiovascular adverse events and mortality.
In patients with severe chronic heart failure (functional class IV according to NYHA classification) treatment should be started with lower doses and under close medical supervision.
Patients with arterial hypertension and coronary heart disease should not stop taking beta-adrenoblockers: ACE inhibitor should be used together with beta-adrenoblockers.
Hypertensive crisis
The efficacy and safety of amlodipine in hypertensive crisis has not been established.
Indapamide
Hepatic encephalopathy
In the presence of liver function abnormalities thiazide and thiazide-like diuretics can lead to hepatic encephalopathy. In this case, the use of the diuretic should be stopped immediately.
Photosensitivity
There have been reported the cases of photosensitivity reactions during the use of thiazide and thiazide-like diuretics (see section “Side effects”). In case of photosensitivity reaction during the drug administration, the treatment should be discontinued. If it is necessary to continue the therapy with diuretics it is recommended to protect skin from sunlight or artificial ultraviolet rays.
Plasma calcium ion content
Thiazide and thiazide-like diuretics may decrease renal excretion of calcium ions and lead to a slight and temporary increase in plasma calcium ions. Severe hypercalcemia may result from previously undiagnosed hyperparathyroidism. In such cases, diuretics should be discontinued and parathyroid gland function should be studied (see section “Side effects”).
Uric acid
Patients with elevated plasma concentrations of uric acid during therapy may experience an increase in the frequency of gout attacks.
Perindopril
Potassium-saving diuretics, potassium preparations, potassium-containing salt substitutes and dietary supplements
It is not recommended to use perindopril concomitantly with potassium-saving diuretics, potassium preparations, potassium-containing salt substitutes, and dietary supplements (see “Interaction with other food supplements”). section “Interaction with other medicinal products”).
Double blockade of the renin-angiotensin-aldosterone system (RAAS)
There is evidence of an increased risk of arterial hypotension, hyperkalemia and renal dysfunction (including acute renal failure) when using ACE inhibitors simultaneously with ARA II or aliskiren. Therefore, dual RAAS blockade as a result of combination of ACE inhibitor with ARA II or aliskiren is not recommended (see sections “Interaction with other medicinal products” and “Pharmacodynamics”). If double blockade is necessary, it should be performed under the strict supervision of a specialist with regular monitoring of renal function, plasma electrolyte levels and BP. ACE inhibitors should not be used simultaneously with ARA II in patients with diabetic nephropathy.
Neutropenia/agranulocytosis/thrombocytopenia/anemia
There are reports of neutropenia/agranulocytosis, thrombocytopenia and anemia during ACE inhibitors administration. In patients with normal renal function and in the absence of other aggravating factors neutropenia rarely develops. Perindopril should be used with particular caution in patients with systemic connective tissue diseases, during immunosuppressant therapy, allopurinol or procainamide, or in their combination, especially in patients with impaired renal function.
Some of these patients had severe infections, in some cases resistant to intensive antibiotic therapy. When perindopril is administered to such patients, it is recommended that the leukocytes in the blood be monitored periodically, and patients should report any signs of infections (e.g., sore throat, fever) to the physician (see section “Side effects”).
Hypersensitivity/angioneurotic edema
When taking ACE inhibitors, including perindopril, in rare cases the development of angioedema of the face, extremities, lips, tongue, vocal cleft and/or larynx may be observed. This may occur during any period of therapy. If symptoms develop, the drug should be stopped immediately and the patient should be observed until the signs of oedema have completely disappeared. If only the face and lips are affected, the swelling usually resolves on its own, although antihistamines may be used to treat the symptoms.
Angioedema accompanied by laryngeal edema may be fatal. Swelling of the tongue, vocal cleft or larynx may cause airway obstruction, in which case intensive therapy should be begun immediately. If such symptoms occur, an epinephrine (adrenaline) solution of 1:1000 (0.3-0.5 ml) should be administered immediately and/or the airway should be secured. The patient should be under medical supervision until the symptoms completely and persistently disappear.
In patients of the Negro race there was a higher incidence of angioedema against the background of ACE inhibitors administration compared to other races.
Patients with a history of Quincke’s edema not associated with ACE inhibitors administration may be at increased risk of developing it when taking the drug (see section “Contraindications”). There have been reports of rare cases of angioedema of the intestine during therapy with ACE inhibitors. Patients have reported abdominal pain as an isolated symptom or in combination with nausea and vomiting, in some cases without preceding angioedema of the face and with normal C1-esterase levels. The diagnosis was established by abdominal computed tomography, ultrasound, or at the time of surgery. Symptoms went away after discontinuation of ACE inhibitors. Therefore, in patients with abdominal pain receiving ACE inhibitors, the possibility of angioedema of the bowel should be considered when making a differential diagnosis.
Anaphylactoid reactions during desensitization
There are separate reports about the development of prolonged life-threatening anaphylactoid reactions in patients receiving ACE inhibitors during desensitization therapy with venom of hymenopteran insects (bees, wasps). ACE inhibitors should be used with caution in patients with a history of severe allergic reactions or susceptibility to allergic reactions undergoing desensitization procedures, and ACE inhibitor use should be avoided in patients receiving immunotherapy with venom of hymenopteran insects. However, anaphylactoid reaction can be avoided by temporarily withdrawing the ACE inhibitor at least 24 hours before the desensitization procedure.
Anaphylactoid reactions during LDL apheresis
In rare cases, life-threatening anaphylactoid reactions may develop in patients receiving ACE inhibitors during LDL apheresis with dextran sulfate. To prevent anaphylactoid reactions, ACE inhibitor therapy should be temporarily discontinued before each apheresis procedure.
Hemodialysis
Anaphylactoid reactions have been reported in patients receiving ACE inhibitors when performing hemodialysis using high flow membranes (such as AN69®). Therefore, it is advisable to use a membrane of another type or use an antihypertensive agent of another pharmacotherapeutic group.
Pregnancy
ACE inhibitors are contraindicated during pregnancy. If continued therapy with ACE inhibitors is necessary, patients should switch to other types of antihypertensive therapy with an established safety profile for administration during pregnancy. If pregnancy occurs, ACE inhibitors should be discontinued immediately and if necessary, alternative antihypertensive therapy should be started (see sections “Contraindications” and “Use during pregnancy and lactation”).
Cough
Dry cough may occur during ACE inhibitor therapy. Cough persists for a long time against the background of the drugs of this group and disappears after their withdrawal. If a patient has a dry cough, one should remember about the possible iatrogenic nature of this symptom. If the physician believes that ACE inhibitor therapy is necessary for the patient, continuation of the drug may be considered.
Mitral stenosis/aortic stenosis/hypertrophic obstructive cardiomyopathy
ACE inhibitors should be prescribed with caution in patients with left ventricular outflow tract obstruction.
Ethnic differences
Perindopril, like other ACE inhibitors, apparently has a less pronounced hypotensive effect in patients of the Negro race compared with other races. It is possible that this difference is due to the fact that patients with arterial hypertension of the Negro race are more likely to have low renin activity.
Surgery/General Anesthesia
The use of ACE inhibitors in patients undergoing surgery with general anesthesia may result in a marked decrease in BP, especially when using general anesthesia agents that have an antihypertensive effect.
It is recommended if possible to stop taking long-acting ACE inhibitors, including perindopril, one day before surgery.
Patients with renovascular hypertension
The method of renovascular hypertension treatment is revascularization. However, the use of ACE inhibitors has a beneficial effect in patients both awaiting surgical intervention and when surgical intervention is not possible.
When Triplixam® is used in patients with existing or suspected renal artery stenosis, treatment should be started in hospital with low doses with constant monitoring of renal status and blood potassium levels, because such patients may develop functional renal failure, which disappears when therapy is stopped.
Atherosclerosis
The risk of arterial hypotension exists in all patients, but special caution should be observed when using the drug in patients with coronary heart disease and insufficiency of cerebral circulation. In such patients the treatment should be started with low doses of the drug.
Perindopril/indapamide.
Lithium preparations
Simultaneous use of the combination of perindopril and indapamide with lithium preparations is not recommended (see.
Arterial hypotension and water-electrolyte imbalance
The presence of initial hyponatremia is associated with a risk of sudden development of arterial hypotension (especially in patients with renal artery stenosis). Therefore, when monitoring patients, attention should be paid to possible symptoms of dehydration and decreased plasma electrolyte content, such as after diarrhea or vomiting. Such patients need regular monitoring of plasma electrolytes. Severe arterial hypotension may require intravenous injection of 0.9% sodium chloride solution.
Transient arterial hypotension is not a contraindication for continuation of therapy. After the recovery of the blood circulation and blood pressure it is possible to resume the therapy using low doses of the combination or using the components of the drug in monotherapy.
All diuretics can cause hyponatremia which sometimes leads to serious complications. Hyponatremia at the initial stage may not be accompanied by clinical symptoms, so regular laboratory control is necessary. More frequent monitoring of sodium ions content is indicated in elderly patients and patients with liver cirrhosis (see sections “Side effects” and “Overdose”).
Patients with diabetes
In patients with type 1 diabetes (risk of spontaneous increase in potassium ions), treatment should be started with lower doses and under close medical supervision.
In patients with diabetes receiving hypoglycemic agents for oral administration or insulin, regular monitoring of plasma glucose concentration is required during the first month of therapy. Blood glucose levels should be monitored in patients with diabetes, especially in the presence of hypokalemia.
Amlodipine/perindopril
Liver failure
In rare cases cholestatic jaundice occurs during ACE inhibitors administration. With the progression of this syndrome, fulminant necrosis of the liver develops, sometimes with a fatal outcome. The mechanism of this syndrome is unclear. If jaundice or significant increase in “hepatic” enzymes activity occurs in patients taking ACE inhibitors, the ACE inhibitor should be stopped and a physician should be consulted (see section “Side effects”).
In patients with impaired liver function T½ and AUC of amlodipine are increased. Amlodipine should be started at the lowest dose and precautions should be observed, both at the beginning of treatment and when increasing the dose. In patients with severe hepatic impairment, the dose should be increased gradually, ensuring close monitoring of the clinical condition.
Triplixam® has not been studied in patients with hepatic impairment. Given the effect of each component of the drug individually, Triplixam® is contraindicated in patients with severe hepatic impairment and also requires special caution when prescribed to patients with moderate to mild hepatic impairment.
Amlodipine/indapamide/perindopril
Renal impairment
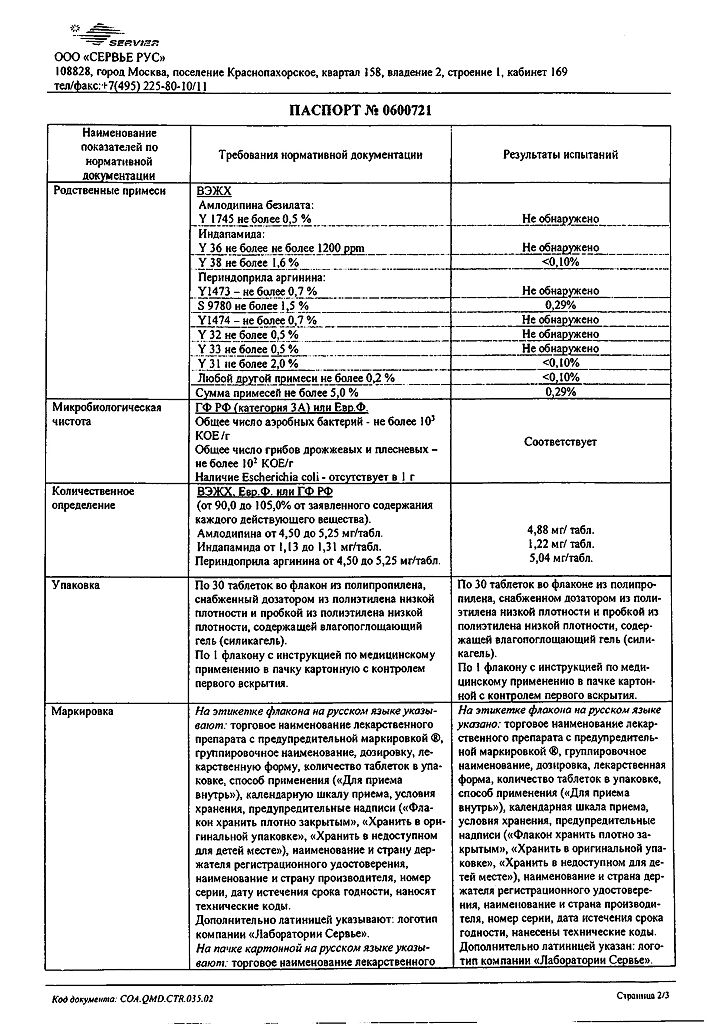
The drug is contraindicated in patients with severe renal insufficiency (CK less than 30 ml/min) (see section “Contraindications.
In patients with moderate renal insufficiency (CKI 30-60 ml/min), Triplixam® is contraindicated in dosages containing 10 mg of perindopril and 2.5 mg of indapamide (i.e. Triplixam® doses of 5 mg + 2.5 mg + 10 mg and 10 mg + 2.5 mg + 10 mg).
In some patients with arterial hypertension without previous obvious renal dysfunction there may appear laboratory signs of functional renal insufficiency during therapy. In this case, treatment with the drug should be discontinued with the further possibility to resume combined therapy using low doses of the drug, or to use the drug components in monotherapy mode. Such patients require regular monitoring of serum potassium ions and creatinine – 2 weeks after the start of therapy and every 2 months thereafter. Renal insufficiency occurs more frequently in patients with severe chronic heart failure or initial renal dysfunction, including renal artery stenosis.
Triplixam® is not recommended in patients with bilateral renal artery stenosis or artery stenosis of the only functioning kidney.
There is a risk of arterial hypotension and/or renal failure (in the presence of chronic heart failure, dehydration and decreased plasma electrolyte content, etc.In some pathological conditions significant activation of RAAS may be observed, especially in cases of severe hypovolemia and blood plasma electrolyte reduction (due to salt-free diet or long-term diuretics use), in patients with initially low blood pressure, renal artery stenosis (including bilateral), chronic heart failure or liver cirrhosis with edema and ascites.
RAAS blockade by ACE inhibitors may be accompanied by a sharp decrease in BP and/or increase in plasma creatinine concentration indicating the development of functional renal insufficiency. These phenomena are more often observed when taking the first dose of the drug or during the first two weeks of therapy. Sometimes these conditions develop acutely and the time of their onset may vary. In such cases, it is recommended to resume therapy starting with lower doses, gradually increasing them. In patients with CHD and cerebrovascular disease a sharp decrease of BP may result in myocardial infarction or stroke.
Thiazide and thiazide-like diuretics are fully effective only in patients with normal or slightly impaired renal function (creatinine concentration in blood plasma of adult patients is below 25 mg/l or 220 µmol/l). In elderly patients creatinine level should be assessed taking into account age, body weight and sex.
At the beginning of diuretic treatment patients due to hypovolemia and hyponatremia may have temporary decrease of glomerular filtration rate and increase of urea and creatinine concentration in blood plasma. This transient functional renal failure is not dangerous in patients with unchanged renal function, but in patients with underlying renal insufficiency, its severity may increase. Patients with renal insufficiency can take amlodipine in standard doses. Changes in plasma concentrations of amlodipine do not correlate with the degree of renal failure.
No special studies on the use of Triplixam® in renal failure have been conducted. When using Triplixam® in renal insufficiency, the effects noted for individual drug components should be considered.
Plasma potassium ion content
Combined therapy with indapamide, perindopril and amlodipine does not prevent hypokalemia, especially in patients with diabetes or renal insufficiency. As in the case of using other antihypertensive agents in combination with a diuretic, regular monitoring of plasma potassium ions is necessary.
Hyperkalemia may develop in some patients during treatment with ACE inhibitors, including perindopril. Risk factors for hyperkalemia are renal insufficiency, impaired renal function, advanced age (>70 years), diabetes mellitus, some concomitant conditions (dehydration, acute cardiac decompensation, metabolic acidosis), simultaneous use of potassium-saving diuretics (such as spironolactone, eplerenone, triamterene, amiloride), potassium drugs or potassium-containing salt substitutes, and use of other drugs that contribute to the increase in plasma potassium ions (eg, heparin). The use of dietary supplements/potassium preparations, potassium-saving diuretics, potassium-containing salt substitutes can lead to a significant increase in blood potassium content, especially in patients with reduced renal function. Hyperkalemia can lead to serious, sometimes fatal heart rhythm disturbances. If combined administration of the above mentioned drugs is necessary, the treatment should be carried out with caution, with regular monitoring of serum potassium ions (see section “Interaction with other medicinal products”). Therapy with thiazide and thiazide-like diuretics is associated with the risk of hypokalemia. Hypokalemia (less than 3.4 mmol/l) should be avoided in the following categories of high-risk patients: elderly patients and/or emaciated patients (even if they do not receive combined drug therapy), patients with cirrhosis with edema and ascites, patients with coronary heart disease, chronic heart failure. Hypokalemia in these patients increases the toxic effect of cardiac glycosides and increases the risk of arrhythmia.
Patients with prolonged QT interval are also included into risk group.
Hypokalemia, like bradycardia, contributes to the development of severe heart rhythm disorders, particularly pirouette-type polymorphic ventricular tachycardia, which may be fatal. In all cases described above, regular monitoring of the content of potassium ions in blood plasma is necessary. The first measurement of potassium ions content should be done during the first week of the therapy.
If hypokalemia is detected, the appropriate treatment should be prescribed.
Elderly patients
Before the beginning of the drug administration renal function and the content of potassium ions in plasma should be assessed. At the beginning of therapy the drug dose is adjusted taking into account the degree of BP reduction, especially in cases of decreased circulating blood volume (CBV) and electrolyte loss. Such measures allow to avoid sharp decrease of BP.
In elderly patients the dose should be increased with caution (see sections “Dosage and administration” and “Pharmacokinetics”).
Influence on the ability to drive vehicles, mechanisms
Due to the possibility of weakness and dizziness during the use of Triplixam® one should be careful when driving vehicles and operating other technical devices that require high concentration and quick psychomotor reactions.
Synopsis
Synopsis
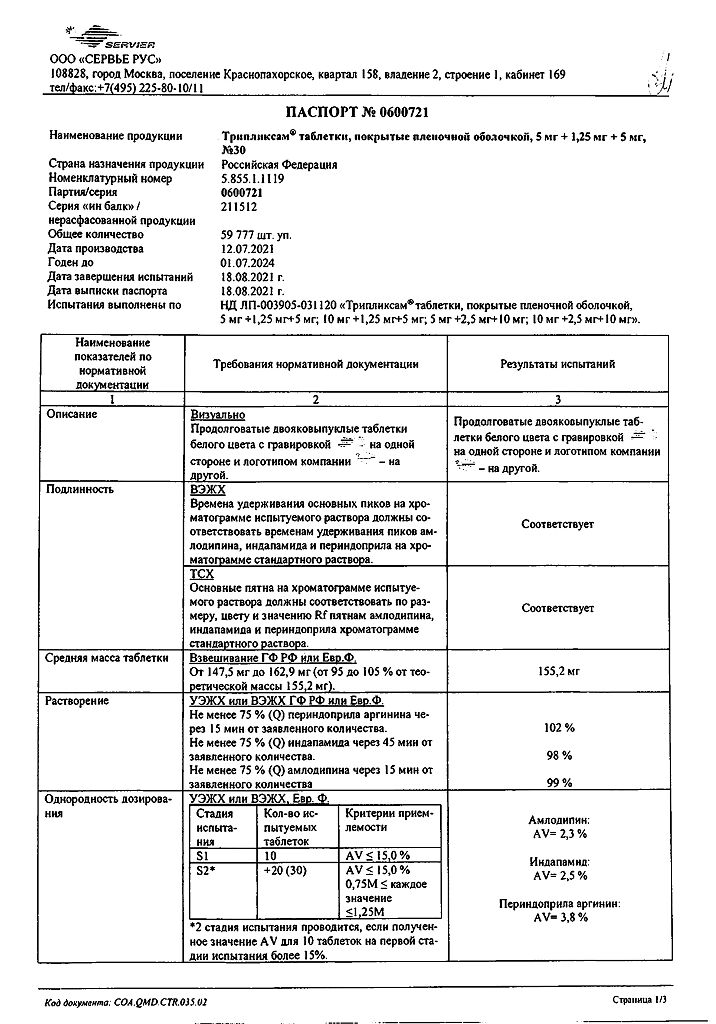
1 tablet 5 mg + 1.25 mg + 5 mg: contains the active ingredients amlodipine besylate 6.935 mg, corresponding to 5.0 mg amlodipine, 1.25 mg indapamide and perindopril arginine 5 mg.
Contraindications
Contraindications
Hypersensitivity to the active and excipients contained in the drug, sulfonamide derivatives, dihydropyridine derivatives, other ACE inhibitors, any other substances contained in the drug;
Patients on hemodialysis;
Untreated heart failure in decompensation stage;
Severe renal insufficiency (creatinine clearance (CK) less than 30 ml/min);
Moderate renal insufficiency (creatinine clearance (CK) less than 60 ml/min) for dosage of perindopril/indapamide 10 mg/2.5 mg combination (ieе. Triplixam® 5 mg + 10 mg and Triplixam® 10 mg + 2.5 mg + 10 mg);
Angioedema (Quincke’s edema) while taking ACE inhibitors in past history (see section “Cautionary information”);
Hereditary/idiopathic angioedema;
Pregnancy (see sect. Pregnancy (see Section “Administration during pregnancy and breastfeeding”);
Breastfeeding (see Section “Administration during pregnancy and breastfeeding”). Severe hepatic insufficiency;
Hypokalemia;
Severe arterial hypotension (systolic BP <90 mm Hg).Systolic BP less than 90 mm Hg.);
Shock (including cardiogenic shock;
Left ventricular outflow tract obstruction (e.g., clinically significant aortic stenosis);
Hemodynamically unstable heart failure after acute myocardial infarction;
Concomitant use with aliskiren-containing drugs in patients with diabetes mellitus or impaired renal function (glomerular filtration rate (GFR) < 60 ml/min/1.73m2 body surface area) (see Sections “Interaction with Aliskiren-containing Drugs. Interaction with other medicinal products” and “Pharmacodynamics”);
Bilateral stenosis of the renal arteries, stenosis of the artery of the only kidney;
Concomitant use with drugs that can cause polymorphic ventricular tachycardia of the “pirouette” type;
Concomitant use with drugs that prolong the QT interval;
Concomitant use with potassium-saving diuretics, potassium and lithium preparations, in patients with high plasma potassium content;
Age under 18 years (effectiveness and safety is not established).
Side effects
Side effects
The most common adverse reactions reported during treatment with perindopril, indapamide and amlodipine as monotherapy were: Dizziness, headache, paresthesias, vertigo, somnolence, visual disturbances, tinnitus, palpitations, blood “rushes” to the facial skin, decreased BP (and effects associated with hypotension), cough, shortness of breath gastrointestinal disorders (abdominal pain, constipation, diarrhea, perversion of taste, nausea, dyspepsia, vomiting), skin itching, rash, maculopapular rash, muscle cramps, swollen ankles, asthenia, edema and fatigue.
Overdose
Overdose
There is no information about overdose of Triplixam®.
Similarities
Similarities
Co-Dalneva
Additional information
| Weight | 0.030 kg |
|---|---|
| Shelf life | 2 years. |
| Conditions of storage | At a temperature not exceeding 25°C. Keep out of reach of children. |
| Manufacturer | Servier Rus LLC, Russia |
| Medication form | pills |
| Brand | Servier Rus LLC |
Other forms…
Related products
Buy Triplixam, 5 mg+1.25 mg+5 mg 30 pcs. with delivery to USA, UK, Europe and over 120 other countries.