No products in the cart.
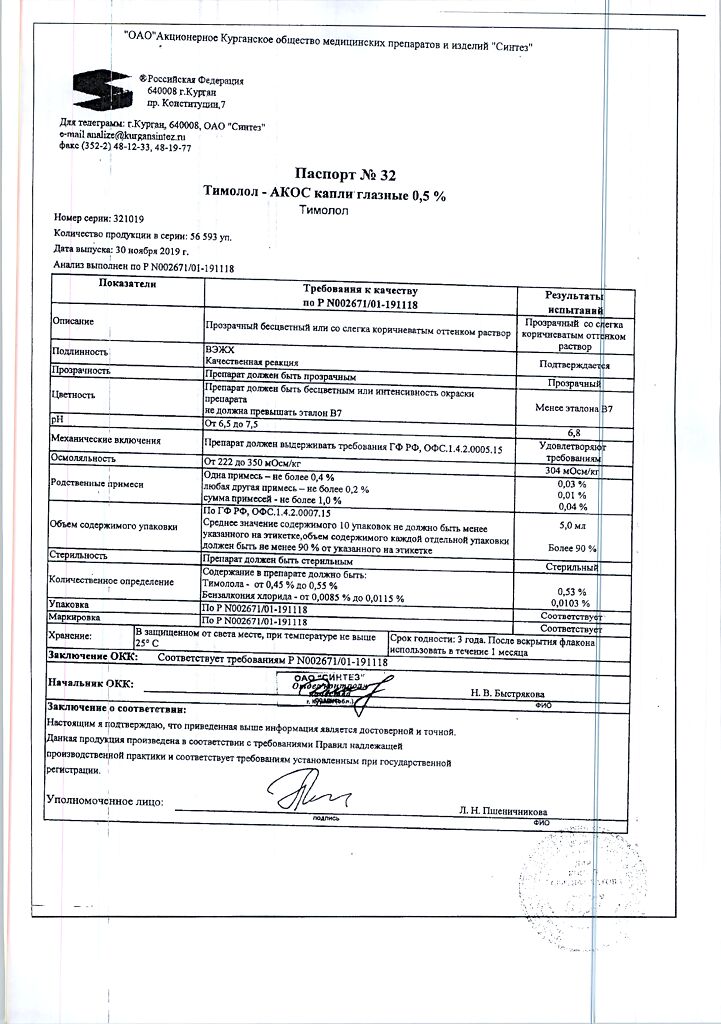
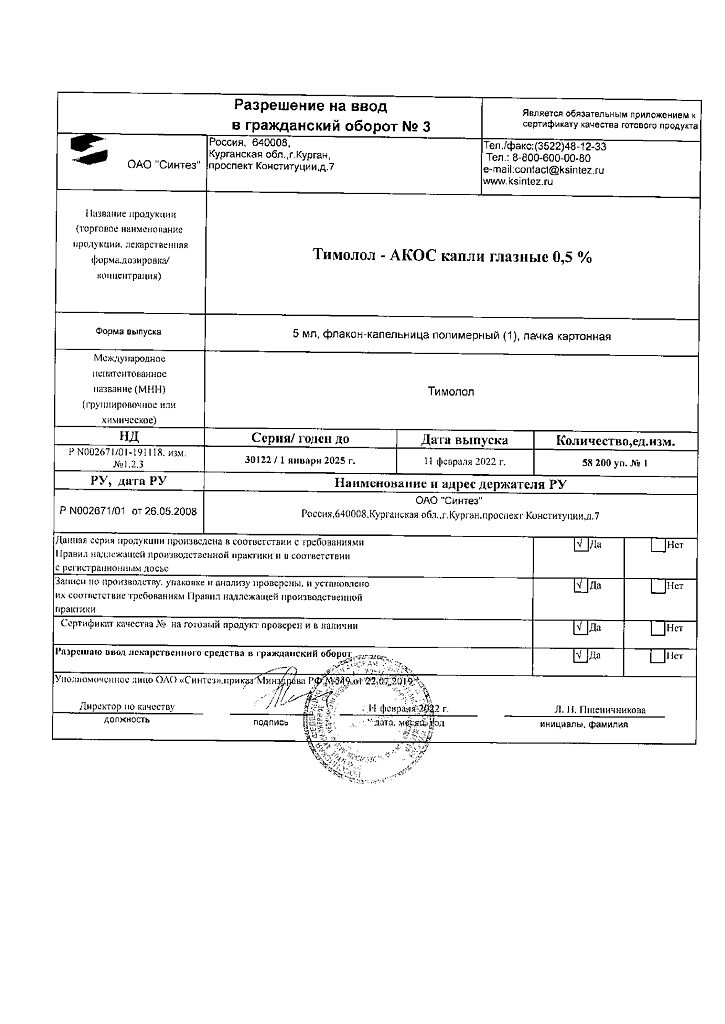
Timolol-ACOS, eye drops 0.5% 5 ml
€1.24 €1.13
Description
Timolol-ACOS is a non-selective beta-adrenoceptor blocker without sympathomimetic activity. When used topically it lowers intraocular pressure by reducing the formation of aqueous humor and slightly increasing its outflow. Decreasing intraocular pressure does not affect accommodation and pupil size, so there is no deterioration of visual acuity and quality of night vision. The action occurs 20 minutes after injection, the maximum effect – in 1-2 hours, the duration of action – 24 hours.
Pharmacokinetics
After instillation of eye drops in the moisture of the anterior chamber of the eye time to reach maximum concentration is 1-2 hours. The active substance enters the systemic bloodstream by absorption through the conjunctiva, nasal mucous membranes and the lacrimal tract. The drug is metabolized by the CYP2D6 enzyme system. Excretion of metabolites is by the kidneys. In infants the concentration of the active substance is significantly higher than its maximum concentration in adult plasma.
Indications
Indications
open-angle glaucoma;
secondary glaucoma (uveal, aphakic, post-traumatic);
acute increase in ophthalmotonus;
angle-closure glaucoma (in combination with miotics).
Pharmacological effect
Pharmacological effect
Timolol-AKOS is a non-selective beta-adrenergic receptor blocker without sympathomimetic activity. When applied topically, it lowers intraocular pressure by reducing the formation of aqueous humor and slightly increasing its outflow. By reducing intraocular pressure, it does not affect accommodation and pupil size, so there is no deterioration in visual acuity and the quality of night vision does not decrease. The effect appears 20 minutes after instillation, the maximum effect is after 1-2 hours; duration of action – 24 hours.
Pharmacokinetics
After instillation of eye drops in the moisture of the anterior chamber of the eye, the time to reach the maximum concentration is 1-2 hours. The active substance enters the systemic bloodstream through absorption through the conjunctiva, mucous membranes of the nose and lacrimal tract. The drug is metabolized by the CYP2D6 enzyme system. Excretion of metabolites by the kidneys. In newborns, the concentration of the active substance significantly exceeds its maximum concentration in the plasma of adults.
Special instructions
Special instructions
During treatment, at least once every 6 months, the function of tear secretion, the integrity of the cornea, and visual fields should be monitored.
3-4 weeks after the start of therapy, the effectiveness of the drug should be monitored.
Contact lenses should be removed before instillation and put on no earlier than 15 minutes after the procedure.
You should not put two beta blockers into your eyes at the same time.
With prolonged use of the drug, the effect may weaken.
When transferring patients to treatment with timolol maleate, refractive error correction may be required.
Impact on the ability to drive vehicles and operate machinery
Caution should be exercised when driving at night while using the drug.
Active ingredient
Active ingredient
Timolol
Composition
Composition
1 ml of eye drops contains:
Active ingredients: timolol maleate (in terms of timolol) – 5.0 mg.
Excipients: benzalkonium chloride, monosubstituted sodium phosphate dihydrate, disubstituted sodium phosphate 12-water, water for injection.
Pregnancy
Pregnancy
The use of Timolol-AKOS during pregnancy is only possible if the expected benefit to the mother outweighs the potential risk to the fetus.
Breastfeeding should be stopped while using the drug.
Contraindications
Contraindications
bronchial asthma;
acute and chronic heart failure;
cardiogenic shock;
dystrophic diseases of the cornea;
rhinitis;
sinus bradycardia;
AV block II and III degrees;
hypersensitivity to the components of the drug.
Side Effects
Side Effects
Local reactions: hyperemia of the skin of the eyelids, burning and itching in the eyes, hyperemia of the conjunctiva, lacrimation or decreased lacrimation, short-term visual acuity impairment, blepharitis, photophobia, swelling of the corneal epithelium, conjunctivitis.
With long-term use of the drug, the development of superficial punctate keratopathy (decreased transparency of the cornea), decreased sensitivity of the cornea, and ptosis is possible; rarely – diplopia.
From the cardiovascular system: decreased blood pressure, bradycardia, bradyarrhythmia, AV block, heart failure, cardiac arrest.
From the central nervous system: paresthesia, dizziness, headache, drowsiness, hallucinations, transient cerebrovascular accident, depression.
From the respiratory system: rhinitis, nasal congestion, shortness of breath, bronchospasm, pulmonary failure.
From the digestive system: nausea, vomiting, diarrhea.
Other: nosebleeds, muscle weakness, sexual dysfunction, decreased potency, collapse, allergic reactions (including urticaria).
Interaction
Interaction
Epinephrine and pilocarpine enhance the hypotensive effect of timolol.
When used simultaneously with reserpine, severe bradycardia or hypotension may develop (if it is necessary to use this combination, careful medical monitoring should be carried out).
When used simultaneously with calcium channel blockers or cardiac glycosides, AV conduction disturbances, acute left ventricular failure or arterial hypotension may occur.
Timolol-AKOS enhances the effect of muscle relaxants (therefore, the drug should be discontinued 48 hours before the intended general anesthesia with the use of peripheral muscle relaxants).
Overdose
Overdose
When applied topically, no symptoms of overdose are described. In case of accidental ingestion, the following may occur: nausea, vomiting, dizziness, headache, decreased blood pressure, bronchospasm, bradycardia.
Treatment: carry out symptomatic therapy. If necessary, isoprenaline is administered intravenously to eliminate severe bradycardia or bronchospasm, and dobutamine is administered in case of arterial hypotension.
Storage conditions
Storage conditions
Store in a place protected from light, at a temperature not exceeding 25°C.
Shelf life
Shelf life
2 years.
Manufacturer
Manufacturer
Sintez, Russia
Additional information
| Shelf life | 2 years. |
|---|---|
| Conditions of storage | Store in a place protected from light, at a temperature not exceeding 25 ° C. |
| Manufacturer | Sintez OAO, Russia |
| Medication form | eye drops |
| Brand | Sintez OAO |
Related products
Buy Timolol-ACOS, eye drops 0.5% 5 ml with delivery to USA, UK, Europe and over 120 other countries.