No products in the cart.
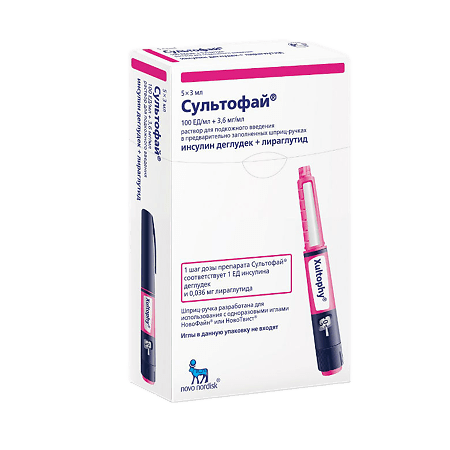
Sultophy, 100 units/ml+3, 6 mg/ml 3 ml cartridges in syringe pens 5 pcs
€1.00
Out of stock
(E-mail when Stock is available)
Description
Sultofay is a combination drug that consists of insulin degludec and liraglutide (produced by recombinant DNA biotechnology using Saccharomyces cerevisiae strain), which have complementary mechanisms of action to improve glycemic control.
Insulin degludec is a basal insulin that after p/k injection forms soluble multi-hexamers that form a depot in the subcutaneous fatty tissue, from where there is continuous and slow absorption of insulin into the bloodstream, providing ultra-long, flat and stable hypoglycemic effect of the drug with low interdaily variability. Insulin degludec binds specifically to the human insulin receptor, providing the same pharmacological effect as human insulin.
The hypoglycemic action of insulin degludek is due to increased tissue glucose utilization after insulin binds to muscle and fat cell receptors and simultaneously inhibits glucose entry from the liver.
Liraglutide is an analog of human glucagon-like peptide-1 (GFP-1) with 97% homology to endogenous human GFP-1, which binds to and activates the GFP-1 receptor. The long-term action profile of liraglutide when administered by injection is provided by three mechanisms: self-association, which results in delayed absorption of the drug; binding to albumin; and higher resistance to dipeptidyl peptidase-4 (DPP-4) and neutral endopeptidase (NEP), which leads to increased T1/2 of the drug from the blood plasma.
The action of liraglutide is due to a specific interaction with GFP-1 receptors and improves glycemic control by reducing blood glucose concentrations on an empty stomach and after a meal. Liraglutide stimulates insulin secretion and reduces excessively high glucagon secretion in a glucose-dependent way. When blood glucose concentration increases, insulin secretion is stimulated and glucagon secretion is inhibited.
On the contrary, during hypoglycemia liraglutide decreases insulin secretion and does not inhibit glucagon secretion. The mechanism of blood glucose concentration reduction is also associated with a slight delay in gastric emptying. Liraglutide reduces body weight and fat tissue mass through mechanisms that cause decreased feelings of hunger and decreased energy intake.
GPP-1 is a physiological regulator of appetite and caloric intake, and GPP-1 receptors are located in several brain regions involved in appetite regulation.
In animal studies, peripheral administration of liraglutide resulted in drug uptake in specific brain regions, including the hypothalamus, where liraglutide, through specific activation of GFP-1 receptors, increased satiety signals and decreased hunger signals, thereby leading to weight loss.
The drug has a stable pharmacodynamic profile with a duration of action reflecting the combination of the individual action profiles of insulin degludec and liraglutide, which allows the drug to be administered once daily at any time, regardless of meals. The drug improves glycemic control through persistent reduction of blood glucose concentration on an empty stomach and after meals.
The reduction of postprandial glucose concentration has been confirmed in a 4-hour sub-study with standard breakfast in patients with poor glycemic control on metformin monotherapy or on therapy with metformin combined with pioglitazone.
The drug improves pancreatic β-cell function as shown in the homeostatic model of pancreatic β-cell function assessment (NOMA-β).
Indications
Indications
Sultofail is indicated to achieve glycemic control in adults with type 2 diabetes in combination with oral hypoglycemic agents.
Active ingredient
Active ingredient
Insulin degludec, Liraglutide
Composition
Composition
1 ml contains:
Active ingredients:
Insulin degludec 100 IU (3.66 mg),
liraglutide 3.6 mg.
Excipients:
glycerol,
phenol,
zinc (as zinc acetate),
Hydrochloric acid/sodium hydroxide (for pH correction),
Water for injection,
pH solution 8.15.
How to take, the dosage
How to take, the dosage
The drug is intended to be administered by injection only.
The drug is administered once daily, at any time of day, preferably at the same time.
The drug should be injected into the hip, shoulder or abdomen. The injection sites within the same anatomical region should be changed frequently to reduce the risk of lipodystrophy.
The dose of the drug is set according to the individual patient’s needs. To optimize glycemic control, it is recommended to adjust the dose based on fasting plasma glucose concentration.
The maximum daily dose of the drug is 50 IU of insulin degludec and 1.8 mg of liraglutide.
When the drug is added to oral hypoglycemic medications, the recommended starting dose of the drug is 10 IU of insulin degludec and 0.36 mg of liraglutide.
When converting from GFP-1 receptor agonist therapy, the recommended starting dose is 16 IU of insulin degludec and 0.6 mg of liraglutide.
If transferring from basal insulin therapy, the recommended starting dose is 16 IU of insulin degludec and 0.6 mg of liraglutide.
Careful glycemic control is recommended during transfer and in subsequent weeks.
Dose adjustment is not required in patients with mild to moderate renal impairment (CKR 60-90 ml/min and 30-59 ml/min). The use of the drug in patients with severe renal impairment, including patients with end-stage renal failure, is contraindicated.
The use of the drug in patients with hepatic impairment is contraindicated.
The drug can be used in elderly patients. Enhanced glycemic control and individual dose adjustment are necessary.
The use of the drug in children and adolescents under 18 years of age is contraindicated due to lack of data on safety and efficacy.
Interaction
Interaction
A number of substances affect glucose metabolism and may require adjustment of the drug dose.
Hypoglycemic drugs, MAO inhibitors, non-selective beta-adrenoblockers, ACE inhibitors, salicylates, anabolic steroids and sulfonamides reduce drug requirements.
The need for the drug is increased by oral hormonal contraceptives, thiazides, GCS, thyroid hormone preparations, sympathomimetics, somatropin and danazol.
Beta-adrenoblockers may mask symptoms of hypoglycemia.
Octreotide/lanreotide can both increase and decrease the need for the drug.
Ethanol and ethanol-containing medications may both increase and decrease the hypoglycemic effect of the drug.
Liraglutide has shown very little pharmacokinetic interaction with other active ingredients with regard to metabolism in the cytochrome P450 (CYP) system and binding to plasma proteins.
A slight delay in gastric emptying with liraglugide may affect the absorption of concomitant oral medications. Drug interaction studies have not shown any clinically significant delay in absorption of these drugs.
A clinically significant interaction with active agents with low solubility or with a narrow therapeutic index, such as warfarin, cannot be excluded. After initiation of therapy with the combination drug, more frequent monitoring of INR is recommended in patients receiving warfarin or other coumarin derivatives.
Liraglutide did not alter total paracetamol exposure after a single dose of 1000 mg. Cmax paracetamol was reduced by 31% and median Tmax was increased by 15 minutes. No dose adjustment was required for concomitant use of paracetamol.
Liraglutide did not alter total atorvastatin exposure to a clinically significant extent after a single dose of atorvastatin 40 mg. Therefore, no dose adjustment of atorvastatin is required when used in combination with liraglutide. When liraglutide was used, Cmax of atorvastatin was reduced by 38% and median Tmax was increased from 1 h to 3 h.
Liraglutide did not alter total exposure to griseofulvin after a single dose of 500 mg. Cmax griseofulvin was increased by 37%, while median Tmax was unchanged. No dose adjustments were required for griseofulvin and other compounds with low solubility and high permeability.
Single administration of digoxin at a dose of 1 mg in combination with liraglutide resulted in a 16% reduction in the AUC of digoxin and a 31% reduction in Cmax. The median time to reach Cmax was increased from 1 h to 1.5 h. Given these results, no dose adjustment of digoxin is required.
Single administration of lisinopril at a dose of 20 mg in combination with liraglutide resulted in a 15% decrease in AUC of lisinopril, a 27% decrease in Cmax. The median Tmax of lisinopril was increased from 6 h to 8 h. Given these results, no dose adjustment of lisinopril is required.
After a single oral contraceptive administration, liraglutide resulted in a 12% and 13% reduction in Cmax of ethinylestradiol and levonorgestrel, respectively. The Tmax of both compounds was increased by 1.5 h with liraglutide. No clinically significant effect on the systemic exposure of ethinylestradiol or levonorgestrel was observed. Thus, no effect on the contraceptive effect is expected when oral hormonal contraceptives and liraglutide are used together.
Substances added to the combination drug may cause degradation of the active ingredients. The drug should not be added to infusion solutions and should not be mixed with other medicinal products.
Special Instructions
Special Instructions
Hypoglycemia may occur if a dose of the drug too large relative to the patient’s need is administered. Hypoglycemia may be caused by skipping meals or unplanned vigorous physical activity. When combined with sulfonylurea, the risk of hypoglycemia may be reduced by reducing the dose of sulfonylurea.
Concomitant renal, hepatic or diseases with adrenal, thyroid or pituitary gland involvement may require changes in the drug dose. Patients with significant improvement in glycemic control (e.g., with intensified therapy) may experience a change in the usual precursor symptoms of hypoglycemia, and they should be informed accordingly.
In patients with a long course of diabetes, the usual precursor symptoms of hypoglycemia may disappear. As with all drugs containing basal insulin, the prolonged effect of the combination drug may lead to a delayed recovery from hypoglycemia.
Injection of inadequate doses and/or discontinuation of hypoglycemic therapy may lead to the development of hyperglycemia and possibly to the development of ketoacidotic coma. In case of discontinuation of therapy with the drug, the instructions on initiation of alternative hypoglycemic therapy should be followed. In addition, comorbidities, especially infectious diseases, may lead to the development of hyperglycemia and thus cause an increased need for hypoglycemic therapy.
In general, the first symptoms of hyperglycemia develop gradually, over several hours or days. These include thirst, rapid urination, nausea, vomiting, drowsiness, hyperemia and dryness of the skin, dry mouth, loss of appetite, and the smell of acetone in the exhaled air. In a situation of severe hyperglycemia, short-acting insulin should be administered. If not treated, hyperglycemia eventually leads to the development of a hyperosmolar coma/diabetic ketoacidosis, which may lead to death. Intensification of insulin therapy (drug component) with dramatic improvement of glycemic control may be accompanied by temporary worsening of diabetic retinopathy manifestations, at the same time long-term improvement of glycemic control reduces the risk of progression of diabetic retinopathy.
The administration of the drug may lead to the formation of antibodies to insulin degludec and/or liraglutide. In rare cases of antibody formation, an adjustment of the drug dose may be required to prevent the development of hyperglycemia or hypoglycemia. In a very small number of patients, therapy with the drug may cause formation of specific antibodies to insulin degludec, antibodies cross-reacting with human insulin, or antibodies to liraglutide. Antibody formation is not associated with decreased efficacy of the drug.
The use of GFP-1 receptor agonists has been associated with a risk of acute pancreatitis. In clinical studies and in post-registration use, there have been reports of acute pancreatitis when using liraglutide, a component of the drug. Patients should be informed about the characteristic symptoms of acute pancreatitis.
If pancreatitis is suspected, the drug should be discontinued; if acute pancreatitis is confirmed, therapy with the drug should not be resumed. Caution should be exercised when using the drug in patients with a history of pancreatitis. In absence of other signs and symptoms of acute pancreatitis, increase of pancreatic enzyme activity is not prognostic factor of acute pancreatitis development.
In clinical trials with GFP-1 receptor agonists, including liraglutide, adverse thyroid reactions have been noted, including increased blood calcitonin concentrations, goiter and thyroid neoplasia, particularly in patients with pre-existing thyroid disease. Because of this, the drug should be used with caution in these patients.
There is no experience of using the drug in patients with inflammatory bowel disease and diabetic gastroparesis. Because of this, the use of the drug in such patients is contraindicated.
In clinical trials, signs and symptoms of dehydration, including renal dysfunction and acute renal failure, have been reported in patients receiving GFP-1 receptor agonists, including liraglutide. Patients receiving the combination drug should be informed about the potential risk of dehydration associated with gastrointestinal side effects and should take precautions to prevent hypovolemia.
Patients should be informed to always check the label on the syringe pen before injection to avoid accidentally injecting another injectable diabetes medication instead of the drug.
The effect of liraglutide on myocardial repolarization has been studied in a QTc interval study. Liraglutide at equilibrium concentration when used at daily doses up to 1.8 mg did not result in prolongation of the QTc interval. As for insulin degludec, there was no statistically significant difference between insulin degludec and the comparison drug with respect to changes in QTc interval duration compared with baseline based on ECG analysis in a 12-month clinical trial.
Impact on driving and operating ability
In hypoglycemia, patients’ ability to concentrate and reaction speed may be impaired. This may pose a risk in situations where these abilities are particularly necessary (e.g., driving or operating machinery). Patients should be recommended to take measures to prevent hypoglycemia when driving vehicles or working with mechanisms. This is especially important for patients with the absence or reduced severity of symptoms-predictors of hypoglycemia or with frequent episodes of hypoglycemia. In these cases, consideration should be given to the appropriateness of driving a vehicle or performing similar activities.
Contraindications
Contraindications
Diabetes mellitus type I, diabetic ketoacidosis; chronic heart failure III-IV classifications NYHA; liver dysfunction; severe renal dysfunction; inflammatory bowel disease and diabetic gastroparesis; age under 18 years; pregnancy, lactation; hypersensitivity to insulin degludec, liraglutide or any of the drug excipients.
The drug should be used with caution in patients with chronic heart failure I-II classifications NYHA, thyroid diseases, as well as in patients with chronic pancreatitis in anamnesis.
Side effects
Side effects
In the program of clinical trials of the drug, no increase in the incidence of specific adverse reactions has been demonstrated compared to the individual components of the drug: insulin degludec and liraglutide. Hypoglycemia and GI disorders were the most frequently reported adverse reactions during therapy with the drug.
The adverse reactions associated with the use of the drug are listed below according to organ system classification and frequency of occurrence. The frequency categories are defined as follows: very common (≥1/10); common (≥1/100 to < 1/10); infrequent (≥1/1000 to < 1/100); rare (≥1/10 000 to < 1/1000); very rare (< 1/10 000) and frequency unknown (frequency cannot be determined based on available data).
Allergic reactions: infrequent – urticaria, hypersensitivity; frequency unknown – anaphylactic reactions.
Metabolism: very common – hypoglycemia; infrequent – dehydration.
The digestive system: often – decreased appetite, nausea, diarrhea, vomiting, constipation, dyspepsia, gastritis, abdominal pain, gastroesophageal reflux, bloating; infrequent – belching, flatulence, cholelithiasis, cholecystitis; frequency unknown – pancreatitis (including necrotizing pancreatitis).
Skin and subcutaneous tissue: infrequent – rash, itching, acquired lipodystrophy.
Additional information
| Weight | 0.203 kg |
|---|---|
| Manufacturer | Novo Nordisk A/S, Denmark |
| Medication form | solution |
| Brand | Novo Nordisk A/S |
Related products
Buy Sultophy, 100 units/ml+3, 6 mg/ml 3 ml cartridges in syringe pens 5 pcs with delivery to USA, UK, Europe and over 120 other countries.