No products in the cart.
Description
Treatment of a new coronavirus infection (COVID-19) of mild or moderate severity in adults, including those with an increased risk of disease progression to severe disease (see “Special Instructions”) and who do not require additional oxygen therapy.
Composition
Composition
For one tablet:
Active ingredients:
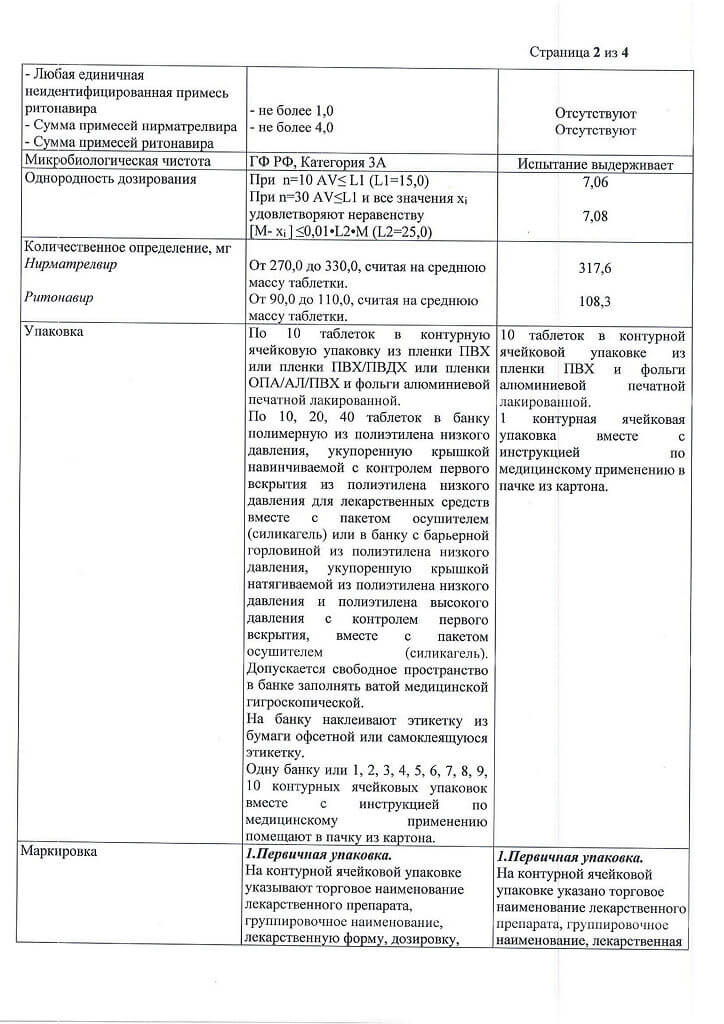
Nirmatrelvir – 300.0 mg
Ritonavir – 100.0 mg
Active ingredients:
Nirmatrelvir – 300.0 mg
Ritonavir – 100.0 mg
Excipients:
Lactose monohydrate – 143.0 mg
Microcrystalline cellulose – 100.0 mg
Croscarmellose sodium – 54.0 mg
Povidone – 40.0 mg
Copovidone – 40.0 mg
Sodium lauryl sulfate – 19.0 mg
Sodium stearyl fumarate – 8.0 mg
Film coating:
Hypromellose – 15.625 mg (62.50%)
Titanium dioxide (E171) – 7.522 mg (30.09%)
Macrogol (Polyethylene glycol) – 1.563 mg (6.25%)
Iron oxide yellow dye (E172) – 0.290 mg (1.16%) or similar finished film coating – 25.0 mg.
Pharmachologic effect
Pharmachologic effect
Pharmacotherapeutic group: systemic antiviral agents; direct acting antivirals; protease inhibitors. ATX code: J05AE. Pharmacodynamics Mechanism of Action Nirmatrelvir is a peptidomimetic inhibitor of SARS-CoV-2 major protease (Mpro), also called 3C-like protease (3CLpro) or nsp5 protease. Inhibition of Mpro by SARS-CoV-2 renders it unable to process polyprotein precursors, resulting in prevention of viral replication. The action of ritonavir as a pharmacokinetic enhancer is based on the activity of ritonavir as a potent inhibitor of metabolism mediated by the cytochrome CYP3A isoenzyme. In the combination medicinal product SKYVIRA, ritonavir, acting as a pharmacokinetic enhancer, inhibits the CYP3A-mediated metabolism of nirmatrelvir, thereby leading to an increase in plasma concentrations of nirmatrelvir. Antiviral activity Antiviral activity in vitro Nirmatrelvir exhibited antiviral activity against SARS-CoV-2 viral infection in dNHBE cell culture, a primary human lung alveolar epithelial cell line (with EC50 values of 61.8 nM and EC90 of 181 nM) after 3 days of exposure to the drug. Antiviral activity in vivo Nirmatrelvir had antiviral activity in cell culture (with EC50 values in the low nanomolar range ≤ 3-fold compared to isolate USA-WA1/2020) against SARS-CoV-2 Alpha (B.1.1.7) isolates ), Gamma (P.1), Delta (B.1.617.2), Lambda (C.37), Mu (B.1.621) and Omicron (B.1.1.529) variants. The beta (B.1.351) variant was the least susceptible variant tested, with an approximately 3.3-fold reduction in susceptibility compared to isolate USA-WA1/2020. Resistance There is currently no information on antiviral resistance of nirmatrelvir in the treatment of SARS-CoV-2. Studies assessing the resistance of SARS-CoV-2 to nirmatrelvir in cell culture and clinical trials have not been completed. Data are only available from an in vitro resistance study using mouse hepatitis virus (MHV)-M pro. The study showed a 4.4- to 5-fold decrease in the sensitivity of nirmatrelvir to mutant viruses with 5 mutations (Pro55Leu, Ser144Ala, Thr129Met, Thr50Lys, Pro15Ala) in MHV-Mpro after 10 passages in cell culture. The significance of this for SARS-CoV-2 is unknown.
Ritonavir-resistant HIV-1 isolates have been isolated from patients receiving therapeutic doses of ritonavir. The decrease in antiretroviral activity of ritonavir is primarily associated with protease mutations V82A/F/T/S and I84V. The accumulation of other mutations in the protease gene (including positions 20, 33, 36, 46, 54, 71, and 90) may also contribute to ritonavir resistance. As mutations associated with ritonavir resistance accumulate, susceptibility to other protease inhibitors may decrease due to cross-resistance.
Use during pregnancy and breastfeeding
Use during pregnancy and breastfeeding
Women of childbearing potential
There are no data on the use of nirmatrelvir/ritonavir in pregnant women to provide information on the risk of adverse pregnancy outcomes associated with SKYVIRA. Women of childbearing potential should avoid pregnancy during treatment and for 7 days after completion of treatment with SKYVIRA.
There are no data on the use of nirmatrelvir/ritonavir in pregnant women to provide information on the risk of adverse pregnancy outcomes associated with SKYVIRA. Women of childbearing potential should avoid pregnancy during treatment and for 7 days after completion of treatment with SKYVIRA.
When prescribing the drug SKYVIRA to women capable of childbearing (including those less than 2 years postmenopausal), it is necessary to confirm a negative pregnancy test result before starting treatment. A repeat pregnancy test should be performed after stopping the drug.
The use of ritonavir may reduce the effectiveness of combined hormonal contraceptives. Patients using combined hormonal contraceptives should be advised to use an effective alternative method of contraception or an additional barrier method of contraception during treatment and until the end of one full menstrual cycle after stopping SKYVIRA.
Pregnancy
There are no data on the use of the combination of nirmatrelvir and ritonavir in pregnant women.
Preclinical studies of nirmatrelvir in combination with ritonavir have not been conducted.
Nirmatrelvir
Preclinical studies with nirmatrelvir have shown no effect of different doses of the drug on fetal morphology or embryo-fetal viability in rats and rabbits, although lower fetal body weight was observed in rabbits with no toxicity in females. Systemic exposure (AUC24) in rabbits at the maximum dose of the drug was approximately 3 times higher than the exposure in humans at the recommended therapeutic dose of nirmatrelvir and ritonavir, without adverse effects on fetal body weight.
Ritonavir
A large body of data on the use of ritonavir in pregnant women suggests no increase in the incidence of congenital malformations compared with rates observed in the general population.
Data from animal studies treated with ritonavir showed reproductive toxicity. Developmental toxicity (embryonic lethality, decreased fetal weight, delayed ossification, and visceral changes including delayed testicular descent) has been observed in rats, primarily at dosages toxic to the female. Developmental toxicity (embryo lethality, reduced litter size, and decreased fetal weight) has been observed in rabbits at dosages toxic to the female.
Breastfeeding period
There is no reliable data on the use of the combination of nirmatrelvir and ritonavir during breastfeeding.
It is not known whether nirmatrelvir is excreted into breast milk in women or female animals. There are also no data on the effect of nirmatrelvir on newborns/infants and the effect on milk production.
Published data indicate that ritonavir is present in breast milk. There is no information on the effect of ritonavir on newborns/infants or on the effect of the drug on milk production. A risk to neonates/infants cannot be excluded.
Breastfeeding should be discontinued during treatment with SKYVIRA and for 7 days after taking the last dose of the drug.
Fertility
There are no data on the effects of the combination of nirmatrelvir and ritonavir, or of either drug alone, on fertility in humans.
Neither nirmatrelvir nor ritonavir had any effect on fertility in rats (see section “Pregnancy”).
Contraindications
Contraindications
- Hypersensitivity to nirmatrelvir, ritonavir or any other component of SKYVIRA
- Lactose intolerance, lactase deficiency, glucose-galactose malabsorption
- Severe liver failure (class C according to the Child-Pugh classification)
- Severe renal failure (eGFR < 30 ml/min)
- Moderate renal failure (eGFR from 30 to 60 ml/min), due to the impossibility of nirmatrelvir dose adjustment
- Pregnancy or planning pregnancy
- Breastfeeding period
- Children under 18 years of age
- Medicines whose clearance is highly dependent on CYP3A and for which elevated concentrations are associated with serious and/or life-threatening reactions
- Medicines that are strong CYP3A inducers that significantly reduce nirmatrelvir/ritonavir plasma concentrations, which may lead to loss of virological response and possible development of resistance
- SKYVIRA should not be started immediately after discontinuation of therapy with any of the following CYP3A inducers due to the long half-life of the recently discontinued drug (see Interactions with Other Drugs).
- The medications listed below are strictly contraindicated for use together with SKYVIRA. This list is mandatory but not exhaustive:
• alpha1-blockers – alfuzosin;
• analgesics – pethidine, piroxicam, propoxyphene;
• antianginal agents – ranolazine;
• antitumor agents – neratinib, venetoclax;
• antiarrhythmic drugs – amiodarone, bepridil, dronedarone, encainide, flecainide, propafenone, quinidine;
• antibiotics – fusidic acid, rifampicin;
• anticonvulsants – carbamazepine, phenobarbital, phenytoin;
• anti-gout drugs – colchicine;
• antihistamines – astemizole, terfenadine;
• antipsychotics/neuroleptics – lurasidone, pimozide, clozapine, quetiapine;
• derivatives of ergot alkaloids – dihydroergotamine, ergonovine, ergotamine, methylergonovine;
• stimulants of gastrointestinal motility – cisapride;
• herbal remedies – St. John’s wort (Hypéricum perforatum);
• lipid-modifying agents:
– HMG-CoA reductase inhibitors – lovastatin, simvastatin;
– Inhibitors of microsomal triglyceride transport protein – lomitapide;
• PDE5 inhibitors: avanafil, sildenafil, vardenafil;
• Sedative/hypnotics – clorazepate, diazepam, estazolam, flurazepam, oral midazolam and triazolam.
Carefully
- Increased liver transaminases
- Hepatitis
- Moderate liver failure (class B on the Child-Pugh scale)
- Patients with organic heart disease and pre-existing disorders of the cardiac conduction system or patients taking drugs that prolong the PR interval (such as verapamil or atazanavir, when using ritonavir as a pharmacokinetic enhancer during 5 days of therapy with SKYVIRA and at a dosage of 100 mg 2 times a day )
- Combined use with drugs is not recommended with drugs listed in the sections “Special instructions”, “Interaction with other drugs”, “Contraindications”.
Interaction
Interaction
SKYVIRA is an inhibitor of the CYP3A enzyme and may increase the concentrations of drugs whose main route of metabolism depends on the cytochrome CYP3A.
Drugs that are extensively metabolized by CYP3A and have high first-pass metabolism are most susceptible to significant increases in exposure when coadministered with nirmatrelvir/ritonavir.
Therefore, concomitant use of nirmatrelvir/ritonavir with drugs whose clearance is highly dependent on cytochrome CYP3A and whose elevated plasma concentrations are associated with serious and/or life-threatening events is contraindicated (see Table 3).
Ritonavir has high affinity for several cytochrome P450 (CYP) isoforms and can inhibit oxidation in the following order: CYP3A4 → CYP2D6.
Ritonavir also has high affinity for P-glycoprotein (P-gp) and can inhibit this transporter.
Ritonavir may induce cytochromes CYP1A2, CYP2C8, CYP2C9 CYP2C19, thereby increasing the biotransformation via glucuronidation and oxidation of some drugs that are metabolized in these ways, which may lead to a decrease in the systemic exposure of these drugs and reduce or reduce their therapeutic effect.
Concomitant administration of other CYP3A4 substrates that may result in potentially significant interactions with it (see Table 3) should only be considered if the benefits outweigh the risks.
Nirmatrelvir and ritonavir are substrates of CYP3A, therefore drugs that induce CYP3A may reduce plasma concentrations of nirmatrelvir and ritonavir and the therapeutic effect of SKYVIRA.
How to take, course of administration and dosage
How to take, course of administration and dosage
The drug SKYVIRA is taken orally regardless of meals.
The tablets should be swallowed whole, without chewing, breaking or crushing, and with a sufficient amount of liquid.
The use of SKYVIRA is possible only under the supervision of a physician.
Dosage regimen
For the treatment of new coronavirus infection (COVID-19) caused by the SARS-CoV-2 virus, the following dosage regimen is recommended in adults:
– 1 tablet orally (orally) twice a day (every 12 hours).
A single dose is 300 mg nirmatrelvir + 100 mg ritonavir. The daily dose is 600 mg nirmatrelvir + 200 mg ritonavir. The duration of the course of treatment is 5 days. Treatment with SKYVIRA should be started as soon as possible after the diagnosis of a new coronavirus infection (COVID-19) and/or within 5 days after the first symptoms of the disease appear. If the patient misses the scheduled time for taking the next dose of the drug, then the “missed dose” of the drug must be taken within 8 hours of the scheduled time of administration. If the patient has not taken the “missed dose” within 8 hours of the scheduled dosing time, then the missed dose does not need to be taken, but instead the next dose must be taken at the scheduled time. The patient should not double the dose to make up for a missed dose. Completion of the full 5-day course of treatment is recommended, even if after initiation of therapy the patient requires hospitalization due to progression of COVID-19 disease to severe treatment. If, after starting treatment, the patient requires hospitalization, he should consult with his attending physician about the need to complete the 5-day course of treatment. Special groups of patients Renal failure Patients with mild renal failure (eGFR 60 to 90 ml/min) do not require dose adjustment. Patients with moderate (see section “Special instructions” and “Contraindications”) (eGFR from 30 to 60 ml/min) and severe renal failure (eGFR < 30 ml/min, including patients with end-stage renal failure on hemodialysis ) taking the drug is contraindicated. Hepatic impairment No dosage adjustment of SKYVIRA is required for patients with mild (Child-Pugh class A) or moderate (Child-Pugh class B) hepatic impairment. SKYVIRA is contraindicated in patients with severe hepatic impairment (see “Special Instructions” and “Pharmacokinetic Properties”). Concomitant therapy with regimens containing ritonavir or cobicistat
No dose adjustment of SKYVIRA is required. Patients diagnosed with human immunodeficiency virus (HIV) or hepatitis C virus who are receiving medications containing ritonavir or cobicistat should continue treatment as prescribed.
Children
There are no data on the safety and effectiveness of the drug SKYVIRA in children under 18 years of age.
Additional information
| Weight | 0.040 kg |
|---|
Related products
Buy Skyvira 300 mg+100 mg 10 pcs with delivery to USA, UK, Europe and over 120 other countries.