No products in the cart.
Description
The drug has antiviral, antitumor, immunomodulatory activity.
Interferon alfa-2b human recombinant, which is the active substance of the preparation, is synthesized by bacterial cells of Escherichia coli strain SG-20050/pIF16, which have the gene of human interferon alfa-2b built into their genetic apparatus. It is a protein containing 165 amino acids and is identical in characteristics and properties to human leukocyte interferon alpha-2b.
The antiviral effect of interferon alpha-2b is manifested during the period of virus reproduction by its active inclusion in the metabolic processes of cells. Interferon, interacting with specific receptors on the surface of cells, initiates a number of intracellular changes, including synthesis of specific cytokines and enzymes (2-5-adenylate synthetase and protein kinase), the action of which inhibits formation of viral protein and viral ribonucleic acid in the cell.
The immunomodulatory action of interferon alfa-2b is manifested in the increase of phagocytic activity of macrophages, increase of specific cytotoxic action of lymphocytes on the target cells, changes in quantity and quality composition of secreted cytokines, changes in functional activity of immunocompetent cells, changes in production and secretion of intracellular proteins.
The antitumor action of the drug is realized due to suppression of proliferation of tumor cells and synthesis of some oncogenes which leads to inhibition of tumor growth.
Indications
Indications
- acute viral hepatitis B – moderate and severe forms at the beginning of jaundice period until the 5th day of jaundice (in later terms the drug is less effective; the drug is ineffective if hepatic coma develops and cholestatic course of the disease);
- in acute prolonged hepatitis B and C, chronic active hepatitis B and C, chronic hepatitis B with delta agent, without signs of cirrhosis and when signs of cirrhosis appear;
- in viral conjunctivitis, keratoconjunctivitis, keratitis, keratoiridocyclitis, keratouveitis;
In the complex therapy in children from 1 year old:
- in acute lymphoblastic leukemia in the period of remission after the end of induction chemotherapy (4-5 months of remission);
- in respiratory papillomatosis of the larynx, starting the day after removal of papillomas.
.
Active ingredient
Active ingredient
Interferon alfa-2b
Composition
Composition
One ampoule or one vial contains:
The active substance is interferon alpha 2b human recombinant 1 million IU;
Excipients:
albumin,
infusion solution 10% – 4.50 mg,
sodium chloride – 8.96 mg,<
Sodium hydrophosphate dodecahydrate – 2.86 mg,
Sodium dihydrophosphate dihydrate – 0.40 mg.
How to take, the dosage
How to take, the dosage
The drug is used intramuscularly, subcutaneously, in or under the lesion focus, subconjunctivally and locally. Immediately prior to use the contents of the ampoule or bottle shall be dissolved in water for injection or 0.9% sodium chlodir solution (1 ml for intramuscular, subcutaneous and nidus injection, 5 ml for subconjunctival and local injection). The drug solution shall be colorless, transparent or slightly opalescent, without sediment and foreign inclusions. The dissolution time should be about 3 min.
In intramuscular and subcutaneous administration
In acute viral hepatitis B the drug is administered by 1 mln IU 2 times per day for 5-6 days, then the dose is reduced to 1 mln IU per day and injected for another 5 days. If necessary (after control blood chemistry tests) the treatment course can be continued in dose of 1 million IU twice a week during two weeks. The course dose is 15-21 mln IU.
In chronic viral hepatitis B with delta agent without signs of liver cirrhosis the preparation is given by 500,000 -1 million IU twice a day 2 times a week during 1 month. Repeated course of treatment in 1-6 months.
In chronic viral hepatitis B with delta agent and signs of liver cirrhosis the preparation is given by 250-500 thousands IU 2 times a day 2 times a week for 1 month. If there are signs of decompensation, similar repeated courses with intervals of at least 2 months are carried out.
In case of acute prolonged and chronic active hepatitis C without signs of liver cirrhosis the preparation is given by 3 mln IU 3 times a week during 6-8 months. If there is no effect, treatment should be prolonged up to 12 months. The course of treatment is repeated in 3-6 months.
In case of kidney cancer the drug is administered 3 million IU daily for 10 days. Repeated courses of treatment (3-9 and more) are carried out at intervals of 3 weeks. The total amount of the drug is from 120 million IU to 300 million IU or more.
In case of hairy cell leukemia the drug is administered daily by 3-6 million IU for
2 months. After normalization of clinical blood test the daily dose of the drug is decreased to 1-2 million IU. Then maintenance therapy of 3 mln IU twice a week for 6-7 weeks is prescribed. The total amount of the drug is 420-600 million IU or more.
In case of acute lymphoblastic leukemia in children in the remission period after the end of induction chemotherapy (4-5 months of remission) – 1 million IU once a week during 6 months, then once every 2 weeks during 24 months. At the same time, maintenance chemotherapy is carried out.
In case of malignant lymphoma and Kaposi sarcoma the preparation is given by 3 million IU daily during 10 days in combination with cytostatics (prospidia chloride, cyclophosphamide) and glucocorticosteroids. In tumor stage of fungal mycosis, primary reticulosis and reticulosarcomatosis it is reasonable to alternate intramuscular administration of the drug by 3 million IU and intrafocal – by 2 million IU for 10 days.
In chronic myeloleukemia the drug is administered by 3 million IU daily or by 6 million IU every other day. The period of treatment is from 10 weeks to 6 months.
In histiocytosis from Langerhans cells the drug is administered 3 million IU daily for 1 month. Repeated courses at 1-2 month intervals for 1-3 years.
In subleukemic myelosis and essential thrombocythemia for correction of hyperthrombocytosis – 1 million IU daily or every 1 day for 20 days.
In case of respiratory papillomatosis of the larynx the preparation is given in the dosage of 100-150 thousand IU per kg of body weight daily for 45-50 days, and then in the same dosage 3 times a week for 1 month. The second and third course are carried out with an interval of 2-6 months.
In persons with high pyrogenic reaction (39 ° C and above) to the introduction of the drug it is recommended to use paracetamol or indomethacin at the same time.
Perifocal administration
In basal cell carcinoma, squamous cell carcinoma, keratoacanthoma, the drug is injected under the lesion 1 million IU 1 time daily for 10 days. In case of pronounced local inflammatory reactions, injection under the lesion site is carried out after 1-2 days. At the end of the course, if necessary, cryodestruction is performed.
Subconjunctival injection
In cases of stromal keratitis and kerato-iridocyclitis subconjunctival injections of the drug in a dose of 60,000 IU in 0.5 ml daily or every other day depending on the severity of the process are prescribed. The injections are performed under local anesthesia with 0.5% dicaine solution. The course of treatment is from 15 to 25 injections.
Local administration
For local use the contents of an ampoule of the drug is dissolved in 5.0 ml of 0.9% sodium chloride solution for injection. If the drug solution is stored, observing the rules of aseptics and antiseptics, transfer the contents of the ampoule into a sterile bottle and store the solution in refrigerator at 4-10 ° C for not more than 12 hours.
In case of conjunctivitis and superficial keratitis on the conjunctiva of the affected eye 2 drops of the solution are applied 6-8 times a day. As inflammation disappears the number of instillations is reduced to 3-4. The course of treatment is 2 weeks.
Interaction
Interaction
Interferon alfa-2b can decrease the activity of cytochrome P-450 isoenzymes and, therefore, affect the metabolism of cimetidine, phenytoin, curantil, theophylline, diazepam, propranolol, warfarin, some cytostatics. It may increase neurotoxic, myelotoxic or cardiotoxic effects of drugs prescribed earlier or simultaneously with it. Avoid co-administration with drugs depressing CNS, immunosuppressive drugs (including oral and parenteral forms of glucocorticosteroids).
Interferons may affect oxidative metabolic processes. This should be considered when concomitant use with drugs that are metabolized by oxidation (including xanthine derivatives – aminophylline and theophylline). When concomitant use of Referon-ES with theophylline it is necessary to monitor the concentration of theophylline in blood serum and adjust the dosing regimen if necessary.
The co-administration of Referon-ES and hydroxyurea may increase the incidence of cutaneous vasculitis.
Alcohol consumption during treatment is not recommended.
Special Instructions
Special Instructions
In order to timely detect abnormal laboratory values that may arise during therapy, general clinical blood tests should be repeated every 2 weeks, and biochemical tests every 4 weeks.
In case of platelet count decreasing to values less than 50-109/l, absolute neutrophil count to less than 0.75-109/l, it is recommended to decrease temporarily the dose by 2 times and repeat the analysis in 1-2 weeks. If the changes persist, it is recommended to stop the treatment.
If the platelet count decreases to less than 25-109/l, the absolute neutrophil count is less than 0.50-109/l, treatment should be discontinued.
In case of immediate hypersensitivity reactions (urticaria, angioedema, bronchospasm, anaphylaxis) the drug should be discontinued and appropriate medical therapy should be immediately administered. Transient skin rash does not require discontinuation of therapy.
In case of signs of liver dysfunction, the patient should be closely monitored. In case of progression of symptoms the drug should be discontinued.
In case of mild to moderate renal dysfunction, their functional status should be closely monitored.
Patients with severe chronic diseases, such as chronic obstructive pulmonary diseases, diabetes mellitus with predisposition to ketoacidosis, in patients with blood clotting disorders, expressed myelosuppression are treated with caution. Pneumonitis and pneumonia are rarely observed in patients receiving Reafferon-EC for a long time. Timely withdrawal of interferon alfa and administration of glucocorticosteroid therapy contribute to relief of pulmonary syndromes.
In patients with thyroid diseases it is necessary to determine the concentration of thyroid hormone before the beginning of treatment, it is recommended to control its level at least once in 6 months. In case of occurrence of thyroid function abnormalities or worsening of the course of existing diseases which cannot be adequately medicated, the drug should be discontinued.
In case of changes in the mental sphere and/or CNS, including the development of depression, monitoring by a psychiatrist is recommended during treatment as well as for 6 months after its completion. These disorders are usually rapidly reversible after discontinuation of therapy, but in some cases it takes up to 3 weeks for them to fully reverse. If mental health symptoms do not regress or worsen, suicidal ideation or aggressive behavior directed toward others appears, it is recommended to stop treatment with Reaferon-EC and provide a consultation with a psychiatrist. Suicidal thoughts and attempts are more common in pediatric patients, primarily adolescents (2.4%) than in adults (1%). If therapy with interferon alfa-2b is deemed necessary in adult patients with severe psychiatric disorders (including a history), it should only be initiated if appropriate individualized screening and therapy for the psychiatric disorder is provided. The use of interferon alfa-2b in children and adolescents with serious mental disorders (including a history of mental illness) is contraindicated.
In long-term use, usually after several months of treatment, visual disturbances may occur. Ophthalmologic examination is recommended before starting therapy. Immediate consultation with an ophthalmologist is necessary in case of any ophthalmologic complaints. Patients with diseases that may cause changes in the retina, such as diabetes mellitus or arterial hypertension, should undergo ophthalmologic examination at least once every 6 months. If visual impairment occurs or worsens, consideration should be given to discontinuing therapy with Reafferon EC.
In elderly patients receiving the drug in high doses, impaired consciousness, coma, seizures, encephalopathies are possible. If these disorders develop and dose reduction is ineffective, treatment should be discontinued.
Patients with cardiovascular disease and/or advanced cancer require close observation and ECG monitoring. If arterial hypotension develops, adequate hydration and appropriate therapy is recommended.
In patients after transplantation (e.g., kidney or bone marrow), medication-assisted immunosuppression may be less effective because interferon has a stimulatory effect on the immune system.
In long-term use, interferon alfa may cause individuals to develop antibodies to interferon. As a rule, antibody titers are not high, and their appearance does not lead to a decrease in the effectiveness of treatment.
With caution patients with predisposition to autoimmune diseases. If symptoms of autoimmune disease occur, a thorough examination should be performed and the possibility of continuation of interferon therapy should be evaluated. Interferon alfa therapy is rarely associated with the occurrence or exacerbation of psoriasis, sarcoidosis.
When using the drug, patients who experience fatigue, drowsiness or disorientation should refrain from engaging in potentially hazardous activities requiring increased concentration and rapid psychomotor reactions.
Influence on driving and operating ability
Patients who are tired, drowsy or disoriented during use should refrain from engaging in potentially hazardous activities requiring increased concentration and psychomotor responsiveness.
Contraindications
Contraindications
- Serious forms of allergic diseases;
- Serious diseases of the cardiovascular system – decompensated heart failure, recently suffered myocardial infarction, marked heart rhythm disorders;
- .Severe renal and/or hepatic failure, including that caused by the presence of metastases, chronic hepatitis with decompensated liver cirrhosis, autoimmune hepatitis;
- Epilepsy and other CNS disorders, mental illness and disorders in children and adolescents;
-
- The use of immunosuppressants after transplantation;
- Thyroid disease that cannot be controlled by conventional therapeutic methods;
- Creatinine clearance below 50 mL/min (when administered in combination with ribavirin), when used in combination with ribavirin, the contraindications in the instructions for ribavirin use should also be considered;
- Hypersensitivity
- Use in men whose partners are pregnant;
- Pregnancy and breastfeeding.
With caution
- Hepatic and/or renal failure, severe myelosuppression. Mental disorders, especially manifested by depression, suicidal thoughts and attempts in the history. Patients with psoriasis, sarcoidosis.
- Pregnancy and breastfeeding
- The drug is contraindicated in pregnancy and during breastfeeding
.
Side effects
Side effects
The incidence of adverse reactions is given according to the WHO classification: “very frequent” – 1/10, “frequent” – more than 1/100 but less than 1/10, “infrequent” – more than 1/1000 but less than 1/100, “rare” – more than 1/10000 but less than 1/1000 and “very rare” with incidence less than 1/10000, including individual reports.
The following adverse events have been observed with Reaferon-ES (in and out of clinical trials):
Often, a flu-like syndrome (chills, fever, asthenia, fatigue, fatigue, myalgias, arthralgias, headaches) is observed with parenteral administration of the drug, partially managed with paracetamol, indomethacin. As a rule, the flu-like syndrome appears at the beginning of treatment and decreases with continuation.
Cardiovascular system: rare – arrhythmias, transient reversible cardiomyopathy, very rare – arterial hypotension, myocardial infarction.
The digestive system: rare – dry mouth, nausea, abdominal pain, dyspepsia, appetite disorders, weight loss, vomiting, diarrhea, very rare – pancreatitis, hepatotoxicity.
The central nervous system: rarely – irritability, nervousness, depression, asthenia, insomnia, anxiety, impaired ability to concentrate, suicidal thoughts, aggressiveness, very rarely – neuropathies, psychosis.
Skin disorders: rare – skin rashes and itching, increased sweating, hair loss. When injected into or under the lesion, rarely a local inflammatory reaction. These side effects are usually not an obstacle to continued use of the drug.
Endocrine system disorders: against the background of long-term use of the drug, changes in the thyroid gland are possible. Very rarely – diabetes mellitus.
Laboratory disorders: during the use of the drug some laboratory function abnormalities may occur, manifested by leukopenia, lymphopenia, thrombocytopenia, anemia, increased alanine aminotransferase activity, alkaline phosphatase, concentration of creatinine, urea. As a rule, these changes are usually minor, asymptomatic and reversible.
Muscular system disorders: rare – rhabdomyolysis, leg cramps, back pain, myositis, myalgia.
Respiratory system: rarely – pharyngitis, cough, dyspnea, pneumonia.
Additional disorders of the urinary system: very rare – renal failure.
The immune system: rare – autoimmune pathology (vasculitis, rheumatoid arthritis, lupus-like syndrome), very rare – sarcoidosis, angioedema, anaphylaxis, facial edema.
In the organs of vision: during local administration of the drug on the ocular mucosa hyperemia, single follicles, edema of the lower vault conjunctiva may occur. Rarely – retinal hemorrhage, focal changes of the ocular fundus, thrombosis of retinal arteries and veins, decreased visual acuity, optic neuritis, edema of the optic disc.
Hearing organs: rare – hearing impairment.
In case of severe local and general adverse reactions, the drug should be discontinued.
Overdose
Overdose
There have been no cases of overdose. Taking into account that the active substance is interferon alfa-2b, in case of overdose it is possible to increase the severity of dose-dependent side effects.
Treatment: discontinuation of the drug; symptomatic therapy if necessary.
Similarities
Similarities
Infagel, Viferon, Grippferon, Reaferon-ES-Lypint, Laifferon
Additional information
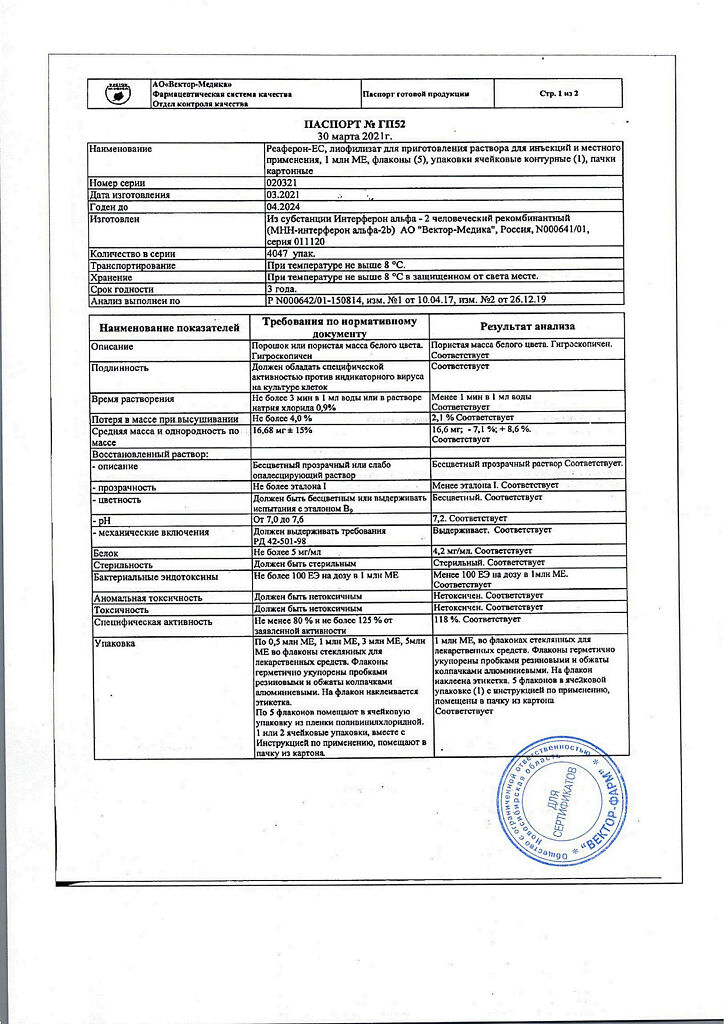
| Weight | 0.050 kg |
|---|---|
| Shelf life | 3 years. |
| Conditions of storage | Store in a dark place at a temperature not exceeding 8 ° C. |
| Manufacturer | Vector-Medica JSC, Russia |
| Medication form | lyophilizate |
| Brand | Vector-Medica JSC |
Related products
Buy Reafferon-ES, lyophilizate 1000000 me 5 pcs with delivery to USA, UK, Europe and over 120 other countries.