No products in the cart.
Pulmicort Turbukhaler, 200 mcg/dose 100 doses
€12.99 €10.83
Description
Pharmacotherapeutic group: glucocorticosteroid for topical use
ATC code: Pharmacological properties
Pharmacodynamics
Budesonide is a glucocorticosteroid with strong local anti-inflammatory effects.
The exact mechanism of action of glucocorticosteroids in the treatment of bronchial asthma is not fully understood. Anti-inflammatory effects, such as inhibition of release of inflammatory mediators and cytokine-mediated immune response, may be the most important. The affinity of budesonide for glucocorticosteroid receptors is 15 times greater than that of prednisolone.
The anti-inflammatory effect of budesonide is mediated by a reduction in the degree of airway obstruction during early and late allergic response. Budesonide reduces airway reactivity in response to histamine and methacholine inhalation.
The sooner budesonide treatment is initiated from the time of diagnosis of persistent bronchial asthma, the greater improvement in lung function should be expected.
A dose-dependent effect on plasma and urinary cortisol levels has been shown with Pulmicort Turbuhaler administration. At the recommended doses, the drug has significantly less effect on adrenal function than prednisone at 10 mg, as shown in ACTH tests.
Use of budesonide in doses up to 400 mcg daily in children over 3 years of age did not result in systemic effects. Biochemical signs of the drug systemic effect may be observed when using the drug in the dose from 400 to 800 mcg per day. When the dose of 800 mcg per day is exceeded, systemic effects of the drug are common.
The use of glucocorticosteroids for the treatment of bronchial asthma may cause growth retardation.
Initially small, usually transient growth retardation (about 1 cm) has been noted, usually during the first year of treatment. Long-term studies in clinical settings have shown that children and adolescents treated with inhaled budesonide achieve, on average, the estimated adult height. However, in a long-term double-blind study primarily without titration of the budesonide dose to the lowest effective dose, the growth of children and adolescents receiving inhaled budesonide was on average 1.2 cm shorter than that of the placebo group when they reached adulthood (see Dosage and Growth Control Guidelines in the Dosage and Administration and Special Instructions sections).
Therapy with inhaled budesonide once or twice daily has been shown to be effective in preventing physical effort asthma.
Pharmacokinetics
Absorption
Inhaled budesonide is rapidly absorbed. After inhalation with Turbuhaler, about 25-35% of the measured dose is absorbed into the lungs. Maximum plasma concentration is reached 30 minutes after inhalation. Systemic bioavailability of the drug is about 38% of the administered dose.
Metabolism and distribution
Binding to plasma proteins is on average 90%. The volume of distribution of budesonide is approximately 3 L/kg. After absorption budesonide undergoes intensive (over 90%) biotransformation in the liver to form metabolites with low glucocorticosteroid activity. The glucocorticosteroid activity of the major metabolites 6b-hydroxybudesonide and 16a-hydroxyprednisolone is less than 1% of the glucocorticosteroid activity of budesonide.
Evolution
Budesonide is metabolized mainly with participation of the CYP3A4 enzyme. Metabolites are excreted unchanged in the urine or in conjugated form. A small amount of unchanged budesonide is excreted in the urine. Budesonide has high systemic clearance (about 1.2 l/min). Pharmacokinetics of budesonide is proportional to the administered dose of the drug.
Pharmacokinetics of budesonide in children and patients with impaired renal function is unknown. In patients with liver disease the time of budesonide retention in the body may be prolonged.
Indications
Indications
Bronchitis, Chronic Obstructive Pulmonary Disease, Cough, Bronchial Asthma- Bronchial asthma requiring maintenance therapy with glucocorticosteroids to control inflammation.
– Chronic obstructive pulmonary disease (COPD).
Active ingredient
Active ingredient
Budesonide
Composition
Composition
One dose contains the active ingredient budesonide 100 mcg or 200 mcg.
How to take, the dosage
How to take, the dosage
The dose of Pulmicort Turbukhaler is selected individually.
Recommended doses of the drug in case of initiation of inhaled glucocorticosteroid therapy during severe exacerbations of
bronchial asthma, as well as against the background of dose reduction or withdrawal of oral glucocorticosteroids intake, are as follows:
Children over 6 years of age: 100-800 mcg/day (total daily dose of the drug may be divided into 2-4 inhalations). In case if the recommended dose does not exceed 400 mcg/day the whole dose may be taken at once (at one time).
In children conversion to single dose should be conducted under the supervision of a pediatrician.
Adults: the usual dose is 200-800 mcg/day (total daily dose of the drug may be divided into 2-4 inhalations). For treatment of severe exacerbation of bronchial asthma the daily dose may be increased up to 1600 mcg.
In case the recommended dose does not exceed 400 mcg/day the whole dose may be taken at once (one time).
When selecting the maintenance dose it is necessary to aim at prescribing the minimum effective dose.
The time of therapeutic effect after inhalation of a single dose of the drug is several hours. The maximum therapeutic effect is achieved 1-2 weeks after treatment. Pulmicort Turbukhaler has a prophylactic effect on the course of bronchial asthma and does not affect acute manifestations of the disease.
Better efficiency of budesonide has been demonstrated when using Turbukhaler compared to the same dose of budesonide in the form of dosed aerosol. If a stable patient is transferred from Pulmicort in aerosol form to Pulmicort Turbuhaler, a lower daily dose of budesonide should be considered.
An increased daily dose of Pulmicort Turbuhaler may be recommended instead of combining the drug with oral glucocorticosteroids due to the lower risk of systemic effects to enhance the therapeutic effect.
Patients receiving oral glucocorticosteroids
Discontinuation of oral glucocorticosteroids should be performed against a stable health background. For 10 days, it is recommended to take a high dose of Pulmicort in the background of oral glucocorticosteroids in a selected dose. Thereafter, the dose of oral glucocorticosteroids should be gradually reduced (e.g., 2.5 mg of prednisolone or its analog) to the lowest possible level. In many cases it is possible to completely discontinue oral glucocorticosteroids.
There are no data on the use of budesonide in patients with renal insufficiency or impaired liver function. Taking into account the excretion of budesonide due to biotransformation in the liver, an increase in the duration of drug action in patients with severe liver cirrhosis can be expected.
Interaction
Interaction
No interaction of budesonide with other drugs used in the treatment of bronchial asthma was observed.
Ketoconazole (200 mg once daily) increases the plasma concentration of oral budesonide (3 mg once daily) by an average of 6 times when used together.
When taking ketoconazole 12 hours after taking budesonide, the concentration of the latter in blood plasma increased on average by 3 times.
There is no information about this interaction when taking inhaled budesonide, but it is assumed that even in this case, an increase in plasma concentration of budesonide should be expected.
These drugs should not be prescribed concomitantly due to lack of data.
If ketoconazole and budesonide must be prescribed together, the time between doses should be increased to the maximum possible.
Decreasing the dose of budesonide should also be considered.
Other potential CYP3A4 enzyme inhibitors (e.g., itraconazole) also cause a significant increase in plasma concentration of budesonide.
Special Instructions
Special Instructions
To reduce the risk of fungal infection of the oropharyngeal mucosa, patients should be instructed to rinse their mouth thoroughly with water after each inhalation of the drug.
Co-prescribing budesonide with ketoconazole, itraconazole or other potential CYP3A4 inhibitors should be avoided. If budesonide and ketoconazole or itraconazole or other potential CYP3A4 inhibitors have been prescribed, the time between doses should be increased to the maximum possible.
Because of the possible risk of impaired pituitary-adrenal function, special attention should be paid to patients who are transferred from oral glucocorticosteroids to taking Pulmicort. Special attention should also be paid to patients who have taken high doses of glucocorticosteroids, or who have received the highest recommended doses of inhaled glucocorticosteroids for a long time. Such patients may exhibit signs and symptoms of adrenal insufficiency in stressful situations. Additional therapy with systemic glucocorticosteroids is recommended in stressful situations or in cases of surgical intervention.
Particular attention should be paid to patients who are transferred from systemic to inhaled glucocorticosteroids (Pulmicort Turbuhaler), or in cases where disruption of pituitary-adrenal function may be expected. In such patients, the dose of systemic glucocorticosteroids should be reduced with special care and adrenal hormonal function should be monitored. Patients may also need to be prescribed oral glucocorticosteroids during stressful situations such as trauma, surgery, etc.
When switching from oral glucocorticosteroids to Pulmicort Turbuhaler, patients may experience previously observed symptoms such as, muscle pain or joint pain. In such cases, a temporary increase in the dose of oral glucocorticosteroids may be necessary. Rarely, symptoms such as fatigue, headache, nausea and vomiting may occur, indicating systemic glucocorticosteroid insufficiency.
Substitution of oral glucocorticosteroids for inhaled ones sometimes leads to the manifestation of existing allergies, rhinitis and eczema, which were previously treated with systemic drugs.
In children and adolescents treated with glucocorticosteroids (regardless of mode of delivery) for a prolonged period, it is recommended that growth indicators be monitored regularly.
Patients should be instructed to contact the attending physician if the effectiveness of short-acting bronchodilators decreases, as an independent increase in the frequency of drug administration may delay the appointment of adequate treatment. In the case of a sudden worsening of the condition, oral glucocorticosteroids should be considered.
Clinical studies and meta-analyses have shown that the use of inhaled glucocorticosteroids in COPD maintenance therapy may lead to an increased risk of pneumonia. Physicians should be aware of the possibility of pneumonia in patients with COPD because clinical signs of pneumonia and exacerbations often coincide.
Effect on the ability to drive vehicles and mechanisms
Due to the possibility of developing neuropsychiatric symptoms, caution should be exercised when driving vehicles and mechanisms.
Synopsis
Synopsis
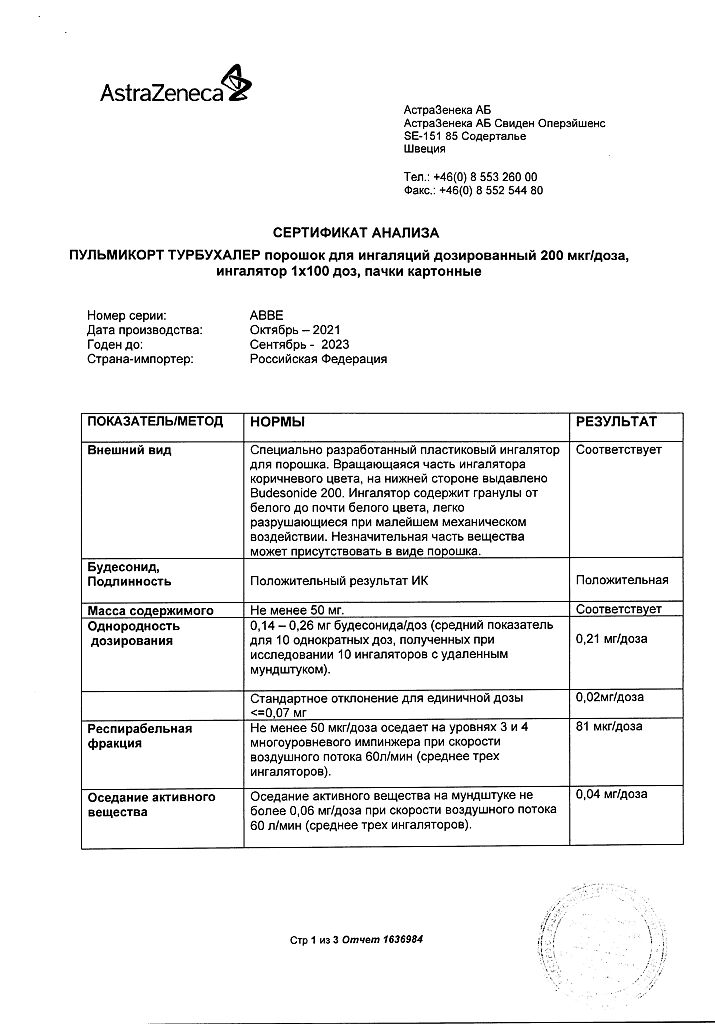
Plastic inhaler, for 100 mcg/dose: the rotating part of the inhaler is light brown, with Budesonide 100 printed on the underside; for 200 mcg/dose: the rotating part of the inhaler is brown, with Budesonide 200 printed on the underside. The inhaler contains round granules from white to almost white, easily disintegrated by the slightest mechanical action. A small part of the substance may be present as a powder.
Contraindications
Contraindications
Hypersensitivity to budesonide. Children under 6 years.
With caution: pulmonary tuberculosis (active or inactive form); fungal, viral or bacterial respiratory infections; liver cirrhosis; pregnancy; period of breastfeeding.
Side effects
Side effects
By frequency of occurrence, adverse effects are classified as follows: very frequently (> 1/10), frequently (> 1/100 to < 1/10), infrequently (> 1/1000 to < 1/100), rarely (> 1/10000 to < 1/1000), very rarely, including individual reports (< 1/10000), frequency not determined.
Patients taking the drug may experience the following side effects:
System-organ grade | . Frequency | Unwanted drug reaction | |
Infections and invasions | Often | Oropharyngeal candidiasis, pneumonia (in patients with COPD) | |
Immune system disorders | Rarely | Immediate and delayed-type hypersensitivity reactions, including rash, contact dermatitis, urticaria, anergic reaction/p> angioedema, bronchospasm, and anaphylactic reaction |
Mental disorders | Rarely | Nervousness, excitability, depression, behavioral disorders | ||
Respiratory system, chest organ disorders /p> and mediastinum | Often | Mild irritation of the pharyngeal mucosa, cough, hoarseness of the voice | ||
Rarely | Bronchospasm | |||
Skin and subcutaneous tissue disorders | Rarely | Bleeding |
Given the risk of oropharyngeal candidiasis, patients should rinse their mouth thoroughly with water after each inhalation of the drug.
In rare cases, symptoms due to systemic effects of glucocorticosteroids may occur, including adrenal hypofunction. Nausea, altered taste, hypercorticism, hypocorticism, cataracts, glaucoma, difficulty swallowing, stunted growth (in children and adolescents), and decreased bone density may also occur.
Overdose
Overdose
In case of overdose with Pulmicort Turbukhaler in doses significantly exceeding the recommended ones, there are no clinical manifestations. When prolonged use of the drug in doses significantly higher than recommended, a systemic glucocorticosteroid effect in the form of hypercorticism and suppression of adrenal function may develop.
Pregnancy use
Pregnancy use
Pregnancy
The results of animal studies have shown that glucocorticosteroids can cause fetal abnormalities, but these findings cannot be extrapolated to humans receiving glucocorticosteroids in the recommended doses.
The use of budesonide in pregnant women has not increased the risk of fetal abnormalities; however, the risk cannot be completely ruled out, and the lowest effective dose of budesonide should be used during pregnancy, keeping in mind the possibility of worsening the course of bronchial asthma.
When prescribing the drug, the expected benefit to the mother must be balanced against the potential risk to the baby.
Breastfeeding
Budesonide is excreted with breast milk, but no effect on the infant is expected with the use of Pulmicort Turbukhaler in therapeutic doses. When prescribing the drug, the expected benefit to the mother and the potential risk to the baby should be considered.
.
Similarities
Similarities
Pulmicort turbuhaler, Pulmicort, Budenofalk, Tafen nasal, Budenit Steri-Neb, Budesonide
Additional information
| Weight | 0.052 kg |
|---|---|
| Shelf life | 2 years. Do not use after the expiration date stated on the package. |
| Conditions of storage | Store at temperatures below 30 C, out of the reach of children. |
| Manufacturer | AstraZeneca AB, Sweden |
| Medication form | metered inhalation powder |
| Brand | AstraZeneca AB |
Related products
Buy Pulmicort Turbukhaler, 200 mcg/dose 100 doses with delivery to USA, UK, Europe and over 120 other countries.